CORE 2024 Annual Report
Investigations of Foodborne Outbreaks and Adverse Events in FDA-Regulated Foods

Report Contents
Introduction
This report includes information on incidents investigated by the United States Food & Drug Administration’s (FDA) Office of Coordinated Outbreak Response, Evaluation, & Emergency Preparedness (CORE+EP), within FDA’s Human Foods Program (HFP), that were linked to FDA-regulated human food in the 2024 calendar year. One of CORE+EP’s missions is to find, stop, and aid in the prevention of foodborne illness outbreaks. These investigations were conducted in partnership with the United States Centers for Disease Control and Prevention (CDC) and state and local partners. In addition to handling outbreaks of foodborne illness and, in some cases, series of adverse events, as of October 2024, CORE+EP has increased responsibilities including food defense, traceability, and supply chain management activities. This report also includes work that was conducted by HFP, CORE+EP, and other respective FDA offices.
CORE+EP has a publicly available Investigation Table that is updated weekly with information about foodborne illness outbreak and adverse event investigations that occur across the United States. This tool provides information and transparency around outbreak investigations, even in their early stages, allowing for early awareness of developing multistate outbreaks of foodborne illnesses. This table is limited to information about incidents managed by CORE+EP and does not include shellfish-related illnesses and outbreaks or incidents related to animal or pet food. The FDA Division of Seafood Safety investigates, tracks, and monitors bivalve molluscan shellfish-related illnesses, fish-related intoxications, and their respective outbreaks. Illnesses or adverse events related to animal/pet food and feed are monitored and investigated by FDA’s Center for Veterinary Medicine (CVM).
Information on outbreaks linked to products regulated by the United States Department of Agriculture Food Safety and Inspection Service (USDA FSIS) can be found on their Foodborne Illness and Disease page. CDC also publishes annual summaries, which include information on both FDA- and USDA-regulated products linked to outbreaks.
Note from the CORE+EP Director
In 2024, FDA’s Center for Food Safety & Applied Nutrition was part of the single largest reorganization in FDA’s modern history and became known as the Human Foods Program. With this reorganization, CORE expanded to become CORE+EP. While the CORE teams continue to safeguard public health by ensuring the safety and integrity of the nation's food supply through continued outbreak detection, response, and aiding in prevention efforts, the EP side is focused on the management of food defense, traceability, and supply chain programs.
We managed several high-profile investigations throughout 2024, including a large outbreak of E. coli linked to carrots, a historical outbreak of illnesses associated with cheese that had stumped investigators since 2018, and a novel adverse events series linked to Diamond Shruumz-Brand Chocolate bars, cones, and gummies.
Beyond responding to outbreaks and certain adverse events, the information included in this report highlights just a few of the activities CORE+EP engages in outside of outbreak and adverse event investigations. I’m pleased to share with you some of our many achievements in this 2024 annual report.
Dr. Stic Harris, CORE+EP Director
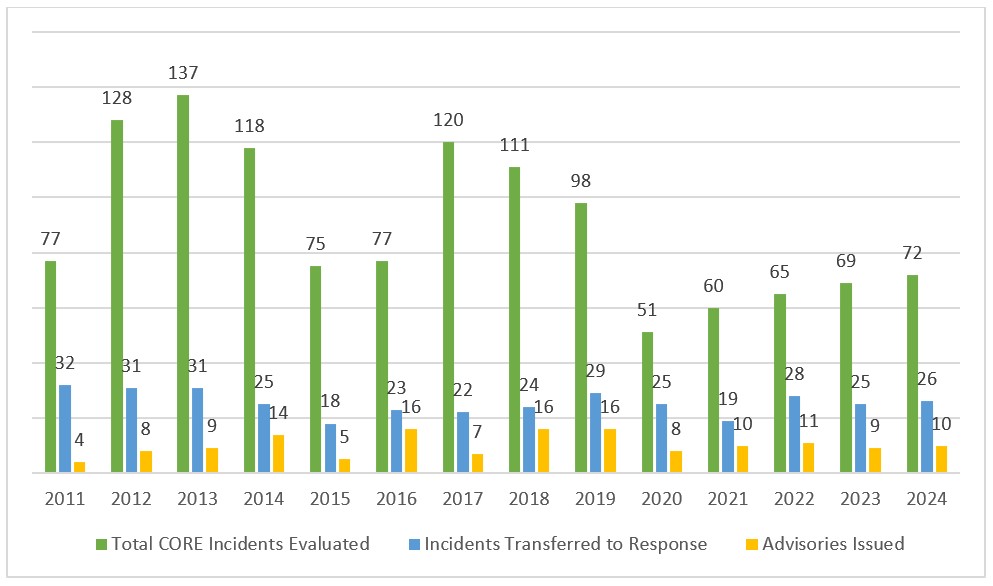
2024 At A Glance
72 Incidents Evaluated:
Incidents evaluated included potential outbreaks, confirmed outbreaks, and adverse events that were analyzed by the CORE Signals and Surveillance (Signals) Team. Not all incidents evaluated by Signals are transferred to one of four CORE Response Teams for follow up.
26 Responses Initiated:
When an outbreak appears to be caused by an FDA-regulated food, information related to the incident is transferred to a Response Team to coordinate FDA’s response efforts.
10 Advisories Issued:
Public health advisories provide consumers with actionable advice to stay safe when an FDA-regulated food is linked to an outbreak.
Note: These categories above represent the number of activities that occurred during 2024. The lifespan of a single incident may include activities spanning multiple years.
CORE Activities, by Year, 2011 – 2024
Note: These categories are not direct subsets of each other but rather represent the number of activities in each category that occurred during 2024. The lifespan of a single incident may include activities spanning multiple years.
The Signals Team evaluates emerging outbreaks and disease surveillance trends in collaboration with CDC, other FDA offices, and state agencies. The team reviews data from food firms, including past inspections, sampling results, product distribution, and sourcing information. It also considers previous incidents involving similar pathogen and food pairs. This information can provide clues to understand emerging outbreaks. When an outbreak appears to be caused by an FDA-regulated human food, information related to the incident is transferred to a Response Team to coordinate FDA’s response efforts. The coordinated efforts often lead a Response Team to identify a specific ingredient or products that can be linked to illnesses and support preventative public health actions.
Not all evaluations conducted by Signals result in a transfer. For example, outbreaks that are linked to non-FDA regulated products, such as those regulated by USDA, are not transferred to a Response Team but are assigned to the proper regulatory agency for further coordination. Additionally, not all incidents transferred to a Response Team result in an advisory being issued. In most cases, this is because a vehicle is not identified while the outbreak is ongoing or at the end of an outbreak investigation.
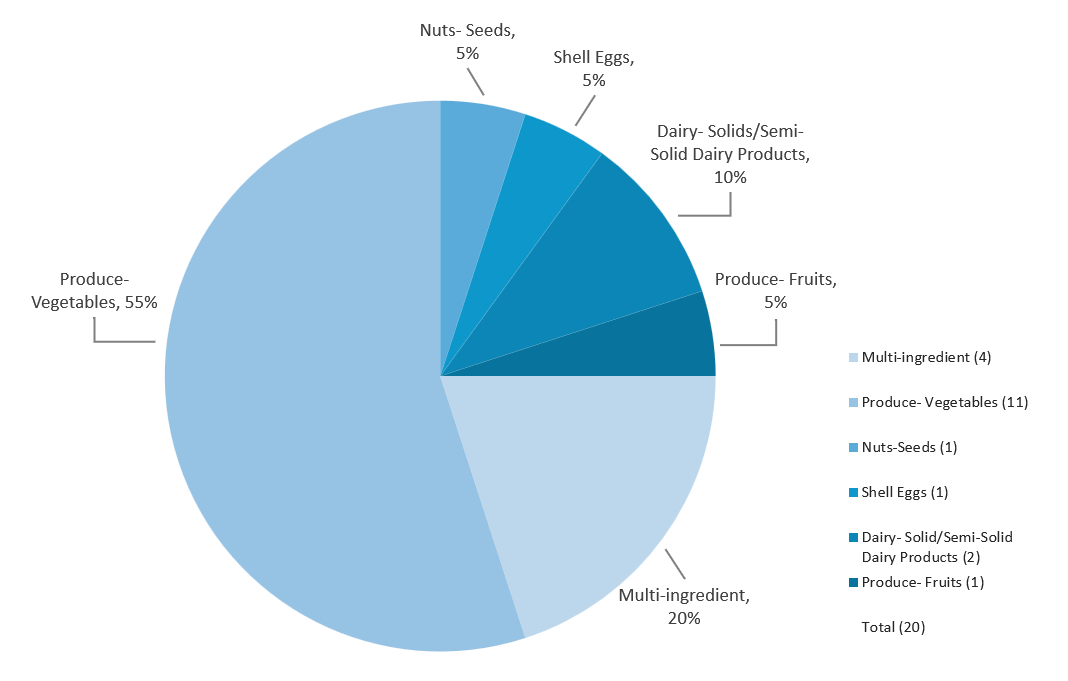
Responses with Identified Product(s)* Linked to Illnesses, by Associated Food Category, 2024
Multi-ingredient: Frozen Shakes, Shrimp Salad**, Chocolates, Gummies & Cones, Bagged salad mix**
Nuts-Seeds: Organic Walnuts
Shell Eggs: Eggs
Dairy- Solid/Semi-Solid Dairy Products: Raw Cheddar Cheese, Queso Fresco and Cotija Cheese
Produce- Fruits: Mangoes**
Produce- Vegetables: Romaine Lettuce**, Spinach**, Imported Cucumbers, Domestic Cucumbers, Jalapeno Pepper**, Organic Carrots, Onions, Sprouts**, Alfalfa Sprouts**, Organic Fresh Basil, Imported Parsley**
*Products were categorized using Interagency Food Safety Analytics Collaboration (IFSCAC) criteria.
**Responses where a product(s) was linked to illnesses, but no Advisory was issued – this is typically due to a lack of specific actionable advice for consumers, including an identified product that is no longer available to consumers at the time of identification. Although an Advisory is not issued in these instances, confirmed vehicles are reported on the CORE Investigation Table.
Public Health Actions
The work conducted by CORE during and following investigations of foodborne illness outbreaks and certain adverse events culminate in actions that can drive the prevention of additional illnesses and future outbreaks. When investigators find the food source of a multistate foodborne illness outbreak, they can take public health actions, such as issuing a public health advisory or recommending that companies voluntarily recall products confirmed to be associated with an outbreak, which are powerful steps to protect public health. In some cases, FDA’s Office of Compliance & Enforcement can pursue additional compliance actions to further protect consumers from unsafe food, such as issuance of warning letters, seizure, injunction, and addition of firms to import alerts.
In 2024, FDA issued ten public health advisories: nine were related to multistate outbreaks of foodborne illnesses and one corresponded to an adverse event series.
Investigations coordinated by CORE also inform follow-up activities carried out by other offices and divisions of FDA. These include but are not limited to follow-up inspections, continued risk assessments, and the development of prevention strategies.
FDA Actions Related to Outbreaks in 2024*
| Pathogen or Cause of Illness | Product(s) Linked to Illnesses | FDA Advisories | Recalls | Legal and Other Action |
|---|---|---|---|---|
| Listeria monocytogenes | Queso Fresco and Cotija Cheese | Outbreak Advisory |
| |
| E. coli O157:H7 | Raw Cheddar Cheese | Outbreak Advisory | ||
| Salmonella Typhimurium | Organic Fresh Basil | Outbreak Advisory |
| |
| E. coli O157:H7 | Organic Walnuts | Outbreak Advisory | Recall Announcement | |
| Salmonella Africana & Braenderup | Cucumbers | Outbreak Advisory | Recall Announcement | Warning Letter |
| Not Identified | Chocolate, Gummies, Cones | Advisory | Recall Announcement | |
| Salmonella Enteritidis | Eggs | Outbreak Advisory | Recall Announcement | Warning Letter |
| E. coli O157:H7 | Onions | Outbreak Advisory | Recall (Announcement in Advisory) | |
| E. coli O121:H19 | Organic Carrots | Outbreak Advisory |
| |
| Salmonella Typhimurium | Imported Cucumbers | Outbreak Advisory | Recall Announcement | Country-wide Import Alert IA # 99-35 |
*Note: Some investigations listed in this table may have been initiated in 2023, but an advisory was not issued until 2024. This is not an exhaustive list of all FDA activities related to these incidents and additional activities may be ongoing.
New Trends in 2024
Food Traceability Rule
In 2024, CORE+EP played a central role in numerous activities involving the Food Traceability Rule (FTR), which implements Section 204(d) of the FDA Food Safety Modernization Act. CORE+EP led or assisted in:
- Development of training for both industry and regulators
- Deployment of FDA’s internal Product Tracing System, within FDA’s IT environment, to test and prepare the system for operational use during active investigations
- Execution of substantial education and outreach efforts.
The training, technology, and outreach efforts conducted in 2024 are important steps to implementing the FTR and will help CORE rapidly trace contaminated food through the supply chain, identify the source, and remove that product from the market before more people get sick.
Noteworthy Outbreaks
In mid-January 2024, FDA and CDC reopened a multi-year historical Listeria monocytogenes investigation, which FDA had previously investigated in 2018 and 2021. Epidemiologic evidence, including information from cases dating back to 2014, identified queso fresco and other similar cheeses as a potential source of those previous outbreaks; however, despite thorough investigations, there was not enough information at the time to identify a specific brand.
Interest in reopening this historical investigation was in part sparked by a sample of Rizo Bros Aged Cotija-branded cheese that tested positive for Listeria monocytogenes during sampling conducted by the Hawaii State Department of Health in early January 2024. The state sampled and tested the product as part of the Laboratory Flexible Funding Model, which is a program where FDA assists partner laboratories with additional resources to increase sample throughput capacity within their state. Rizo-López Foods, Inc., recalled one lot of Aged Cotija Mexican Grating Cheese after the state’s findings. However, a genetic relationship was found that connected the sample of cheese that tested positive for Listeria to a 2014 clinical isolate from the historical investigation. This connection, along with new illnesses being reported to CDC at the time, prompted further investigation by FDA.
On January 24, 2024, FDA initiated an on-site inspection at Rizo-López Foods, Inc., of Modesto, California. In addition to cotija cheese, the firm manufactured queso fresco cheese and a variety of dairy products. State and local public health officials interviewed people about the foods that they ate in the month before they got sick. Of the 22 people interviewed, 16 (73%) reported eating queso fresco, cotija, or other similar cheeses. In response to FDA’s outbreak investigation, the firm temporarily ceased the production and distribution of all dairy products while the investigation was ongoing. The firm took action to stop product from reaching consumers at the time of FDA’s investigation. But two environmental samples collected during the on-site inspection tested positive for Listeria monocytogenes. Whole genome sequencing (WGS) analysis of each sample showed they contained the same strain of Listeria that caused illnesses in the outbreak. Results from WGS analysis prompted the firm to recall all sell by dates of its dairy products including cheese, yogurt, and sour cream, which impacted 19 brands. Collection of product and environmental samples during the 2024 investigation, as well as the analysis of those samples, helped to protect public health and ultimately provided the information needed to solve the multi-year investigation.
On October 8, 2024, the U.S. District Court for the Eastern District of California entered a consent decree of permanent injunction against Rizo-López Foods, Inc. Among other things, this permanent injunction requires the company to make corrective actions and receive FDA approval before they may resume operations.
In June 2024, FDA and CDC quickly responded to a series of illnesses associated with eating Diamond Shruumz-brand Chocolate Bars, Cones, and Gummies. These products were available nationwide including at smoke, or vape, shops and retailers that sell hemp-derived products such as cannabidiol (CBD) or delta-8 tetrahydrocannabinol (delta-8 THC). With the assistance of America’s Poison Centers as well as state and local partners, FDA reviewed reports of consumer complaints and adverse event submissions to FDA to determine appropriate steps to protect public health.
At the onset of FDA’s adverse event investigation, samples of Diamond Shruumz-branded products were collected for analysis because initial reports from consumers showed a variety of severe symptoms after eating the products including, but not limited to, seizures, central nervous system depression (loss of consciousness, confusion, sleepiness), and abnormal heart rates. FDA swiftly conducted traceback efforts to narrow down the multiple manufacturers that were identified as possible production or distribution centers and initiated investigations at key firms.
Part of this investigation focused on the analysis of results from products that were collected and sampled, which provided an additional opportunity for FDA and CDC medical officers to collaborate on understanding the health effects and risks associated with such products. While the effects of ingesting individual ingredients contained in the products are understood, more research is needed to determine the effects of these compounds when they are ingested together.
This investigation is noteworthy for several reasons, including the speed with which FDA and CDC, in collaboration with other federal, state, and local partners, were able to issue public warnings and work with industry to remove product from the market. Although adverse event reports are a common means by which consumers and healthcare professionals inform federal agencies of issues or concerns with products, adverse event reports that potentially involve foodborne illnesses or injuries are a less common source of information leading to investigations by CORE. Unlike traditional outbreaks of foodborne illnesses that are genetically linked to microbial pathogens, CORE had to rely on self-reported information submitted by healthcare providers, consumers, and some state partners as an initial step in determining if a product is a potential shared source of exposure among complainants, making it more complicated to establish a causal relationship.
FDA is now focused on post-incident response actions and activities, which may include additional surveillance as well as prevention and compliance activities.
In November 2024, FDA and CDC collaborated with state and local partners to investigate cases of Shiga toxin-producing E. coli O121:H19 infections spanning nationwide. This investigation involved a unique hypothesis for the potential food source causing the illnesses – carrots. The hypothesis would lead CORE+EP to confirm this pathogen-commodity pair for the first time.
FDA’s traceback efforts pinpointed a common carrot supplier, leading to inspections with environmental sampling at Grimmway Farms in California. Two samples collected during the course of the investigation tested positive for different strains of Shiga toxin-producing E. coli (STEC). While both strains of E. coli detected in the samples are capable of causing human illness, neither matched the strain of E. coli that caused illnesses in this outbreak. Ultimately, epidemiologic and traceback evidence showed that recalled organic carrots were the likely source of illnesses in this outbreak though sample analysis did not identify the specific strain of E. coli that caused illness in the environmental samples.
In response to this investigation, Grimmway Farms recalled product, prompting recalls from 20 different brands. Traceback efforts also uncovered that the recalled carrots may have been distributed and available internationally. FDA included information in this outbreak advisory that indicated the recalled products were also distributed to customers in Canada, Colombia, Hong Kong, and the United Arab Emirates. Grimmway Farms also directly notified international customers who received recalled products, which further amplified the efforts to prevent additional illnesses. Both the wide range of products and the potential for the products to be in homes of consumers even after the recommended “Best if used by” date made alerting the public a priority. FDA and CDC quickly aligned on advice to share with consumers so that additional illnesses could be prevented.
CORE Publications
One way CORE guides and supports outbreak prevention efforts is through sharing CORE’s outbreak investigations and analytic summaries with internal and external stakeholders through publications and presentations. CORE publishes articles on FDA.gov, in peer-reviewed scientific journals, and in other food safety and public health related periodicals. CORE staff also present at conferences and scientific meetings. In 2019, the Outbreak Analytics Team was created to support this mission and has since successfully published an increased number of articles since inception, compared to previous years.
For details on publications, see CORE Publications.
CORE Resources
The CORE Investigation Table is updated weekly with information about foodborne illness outbreak and certain adverse event investigations that occur across the United States. This tool provides information and transparency around outbreak investigations, even in their early stages, which allows for early awareness of developing multistate outbreaks of foodborne illnesses. This table is limited to incidents managed by FDA’s CORE and does not include shellfish-related illnesses and outbreaks or incidents related to animal or pet food. The FDA Division of Seafood Safety investigates, tracks, and monitors bivalve molluscan shellfish-related illnesses, fish-related intoxications, and their respective outbreaks. Illnesses or adverse events related to animal/pet food and feed are monitored and investigated by FDA’s Center for Veterinary Medicine (CVM). Information on outbreaks linked to products regulated by USDA FSIS can be found on their Foodborne Illness and Disease page. CDC also publishes annual summaries, which include information on both FDA- and USDA-regulated products linked to outbreaks.
Investigations of Foodborne Illness Outbreaks
Public Health Advisories from Investigations of Foodborne Illness Outbreaks
Acknowledgments
CORE’s work to find, stop, and aid in the prevention of foodborne illness outbreaks would not be possible without all our partners across the U.S. and internationally. Our Office would like to extend our greatest appreciation for the continued collaborations with local, state, tribal, and territorial health and agriculture departments; the Centers for Disease Control and Prevention; the U.S. Department of Agriculture; and internal partners at FDA.