
Highly Pathogenic Avian Influenza (HPAI)
FDA’s Response to Ensure Continued Effectiveness of Federal-State Milk Safety System

FDA’s Response to Ensure Continued Effectiveness of Federal-State Milk Safety System

You can help. Get the facts from the FDA. Stop the spread of false rumors, and share the facts with your loved ones.
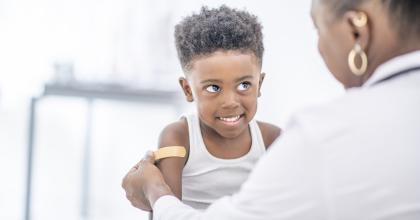
FDA-approved vaccines to prevent measles are safe and effective. Talk to your health care provider and learn more about how childhood vaccines can protect your children.
Alerting you to the latest recalls of potentially unsafe FDA-regulated products gathered from press releases and other public notices. Not all recalls have press releases or are posted on this website.
Sep 20
Food & Beverages
Sep 20
Food & Beverages
Sep 19
Food & Beverages

FDA will hold the first National Hispanic/Latino Family Cancer Awareness Week to increase cancer awareness within the Hispanic/Latino population.

FDA attended an NC roundtable with members of the REACH Consortium, researchers, patients, community members, and LGBTQIA+ health professionals and advocates.

The trajectory of men’s health in the U.S. is headed in a negative direction. Let’s examine the potential underlying reasons for this decline.
Sep 23
Biologics, Medical Devices
Sep 23
Biologics, Medical Devices
Sep 23
Biologics, Medical Devices