Hearing Aids and Personal Sound Amplification Products: What to Know
Learn about hearing aids, including OTC hearing aids, and personal sound amplification products.
Are you or a loved one having a hard time hearing? Perhaps you considered getting a hearing aid in the past?
Hearing aid technology keeps evolving, which means there’s a growing variety of styles and features to consider.
“People who already use a hearing aid know that selecting one is not a simple decision,” says Eric Mann, M.D., Ph.D., Senior Advisor in the U.S. Food and Drug Administration office responsible for hearing aids. “Hearing loss affects people in different ways. So, it’s important to choose a hearing aid that’s appropriate for your condition and fits your lifestyle.”
The FDA regulates hearing aids to make sure they provide reasonable assurance of safety and effectiveness. If you’re considering hearing aids, this article highlights some common technologies and terms you may encounter, including information about the FDA’s requirements for hearing aids sold over-the-counter (OTC), which are intended for people 18 years of age and older who have perceived mild to moderate hearing loss.
Hearing Aids and How They Work
People may be born with hearing loss. Or they may develop it later in life — often because the inner ear can wear out as we age or be damaged by years of exposure to loud sounds.
In some cases, hearing loss is temporary and can be restored with medical help. In other cases, it’s permanent but can be improved with hearing aids.
Hearing aids are medical devices worn behind or in the ear. They can improve hearing by making sounds louder. However, hearing aids usually won’t restore your hearing to normal levels or quality in the way that eyeglasses can often restore vision to 20/20.
Air-Conduction vs. Bone-Conduction Hearing Aids
Most hearing aids work through air conduction. They bring amplified sound into the ear canal. Sound then moves through the eardrum and three tiny bones in the middle ear to reach the inner ear, where it’s processed and sent to the brain.
For people who have problems with their outer or middle ear, those areas can be bypassed with bone-conduction hearing aids. They send sound through the skull to reach the inner ear.
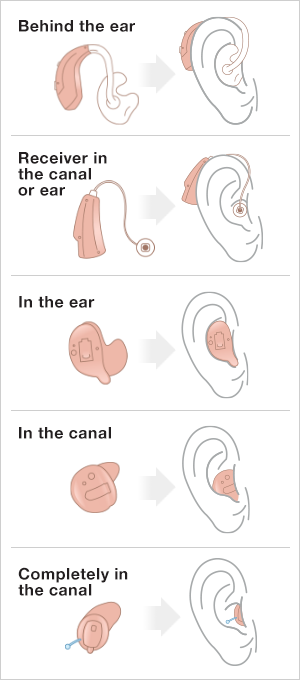
Styles of Hearing Aids
Behind-the-ear (BTE) aids: BTE hearing aids are generally the largest hearing aid style. A plastic case containing most of the electronics sits behind the ear and is connected to an earmold that fits in the ear canal. BTE hearing aids can be used by people of all ages. The style is often chosen for young children because it can be adapted as they grow.
Receiver-in-the-canal (RIC) aids: RIC (or mini receiver-in-the-ear; mini RITE) hearing aids sit behind the ear but are typically smaller than a BTE. The RIC hearing aid is attached to a tube housing a small wire with a dome-shaped tip at the end that rests in the ear canal (in some cases, earmolds are used). The RIC design allows more of the ear canal to remain open and is less visible than the BTE style.
In-the-ear (ITE) aids: This hearing aid sits completely in the outer ear (the “bowl” of the ear). All the hearing aid electronics are housed in a custom-fit shell.
In-the-canal (ITC) aids and completely-in-the-canal (CIC) aids: These are the smallest hearing aids currently available. The electronics are contained in a small custom-fit shell that fits partly or completely into the ear canal. Some people may like them because they are less noticeable while other people may find them harder to handle.
For more information, please see the FDA webpage, Types of Hearing Aids
Getting Hearing Aids
Medical Evaluation for Children (Younger Than 18 Years of Age)
While hearing loss in adults is often caused by aging or noise exposure, the reasons for hearing loss in children are more varied and may be associated with other medical conditions that should be medically evaluated before prescribing hearing aids. OTC hearing aids are not intended for and must not be sold to people younger than 18 years of age. Hearing aids intended for people younger than 18 years of age are prescription hearing aids.
Prescription hearing aids are sold by audiologists; ear, nose, and throat doctors; or sellers licensed to dispense hearing aids, such as instrument specialists.
Over-the-Counter (OTC) Hearing Aids
To broaden access to hearing aids, the FDA established a new category of OTC hearing aids so you will be able to buy one in the store or online without seeing a physician for an exam or an audiologist for help with fitting. The new FDA regulation, which implements a law from Congress, also aims to stimulate competition, providing consumers with improved access to devices that meet their needs and are less expensive than current options.
The OTC final rule applies to certain air-conduction hearing aids intended for people 18 years of age and older who have perceived mild to moderate hearing loss. A person with mild hearing loss may have difficulty hearing some speech sounds but not others. A person with moderate hearing loss may have increased difficulty hearing some speech sounds, even in a quiet room with someone talking at a normal level.
“We want hearing aids to be more readily available and accessible, especially as our population ages,” Mann explains. “It’s also important for people to recognize that hearing loss could be a sign of an easily treatable problem like built-up earwax or a more serious problem like a benign tumor on the hearing nerve. See a doctor when things don’t feel right, when your hearing loss is progressing, or if you are having associated symptoms like dizziness, ear pain, or drainage from the ear canal.”
For more information on OTC hearing aids, please see the FDA webpage, OTC Hearing Aids: What You Should Know.
Hearing Aids vs. Personal Sound Amplification Products
You may have seen products in stores or online that are known as personal sound amplification products (PSAPs). These are not alternatives to hearing aids.
While hearing aids and PSAPs both amplify sound for the user, the products have different intended uses. Hearing aids are intended to make up for impaired hearing. PSAPs, in contrast, are intended for people with normal hearing to amplify sounds in certain situations, such as recreational activities like birdwatching or hunting.
Because such PSAPs are regulated as consumer electronics and not medical devices, they may be more variable in terms of product quality compared to hearing aids.
|
|
Over-the-Counter |
Prescription |
Personal Sound Amplification Products |
|
Type of Product |
Medical device Electronic product |
Medical device Electronic product |
Electronic product |
|
Intended Users |
|
|
|
|
Conditions for Sale |
|
|
No applicable FDA requirements regarding conditions for sale |
Reporting Problems with Hearing Aids
To report a problem involving your hearing aid, submit information to the FDA as soon as possible after the problem. The FDA calls them “adverse events,” and they might include: skin irritation in your ear, injury from the device (like cuts or scratches, or burns from an overheated battery), pieces of the device getting stuck in your ear, suddenly worsening hearing loss from the device, etc. Reports can be made voluntarily to MedWatch: The FDA Safety Information and Adverse Event Reporting Program online or by calling 1-800-FDA-1088
Additional Resources
- Hearing Aids, FDA
- Cochlear Implants: A Different Kind of ‘Hearing’, FDA Consumer Update
- Hearing, Ear Infections and Deafness, National Institute on Deafness and Other Communications Disorders
- Establishing Over-the-Counter Hearing Aids - Final Rule
- Regulatory Requirements for Hearing Aid Devices and Personal Sound Amplification Products - Final Guidance for Industry and Food and Drug Administration Staff