Office of Global Operations Also referred to as: OGO
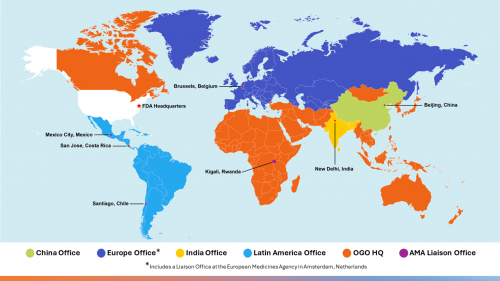
The Office of Global Operations (OGO) manages the Office of Global Policy and Strategy's foreign offices located in strategic locations around the globe including Asia (China and India); Europe (Belgium); and Latin America (Chile, Costa Rica, and Mexico), as well as the African Medicines Agency Liaison Office (AMALO) in Rwanda. One member of the Europe Office is embedded with the European Medicines Agency in Amsterdam, Netherlands. Interactions with other locations where the FDA does not have a physical foreign presence are managed by OGO's dedicated bilateral engagement staff located at White Oak. The office also includes the Global Support Team (GST) and Program and Policy Team (PPT).
OGO’s programmatic priorities include:
• Increasing the FDA's knowledge about the country or region for better decisions and actions.
• Conducting in-country/in-region inspections in cooperation with the FDA's larger foreign inspection program.
• Engaging with regulatory counterparts and regional and international organizations to foster regulatory convergence and harmonization, and to advance best practices.
• Expanding foreign governments’ and industry’s awareness and understanding of FDA regulations through outreach and training.
• Managing the deployment and support of overseas staff.
Each foreign office (excluding AMALO) includes a director, a deputy director, locally employed staff, translators, and international relations specialists. Consumer safety officers, who conduct inspections of food and medical product facilities, can be found in China, India, and Latin America Offices.
Focus on OGO
- Looking Back at the Founding of the FDA Foreign Offices, (November 17, 2023)
- China Office
- Europe Office
- India Office
- Latin America Office
- African Medicines Agency Liaison Office
- FDA Overseas Office Directors
Recalls and Alerts
- FDA Recall Information on Twitter
- MedWatch: The FDA Safety Information and Adverse Event Reporting Program
- Recalls, Market Withdrawals, & Safety Alerts
- Warning Letters
Resources for You
- Contact Information: OGPS Offices
- Countries and Regions Covered by OGPS Offices
- FDA Globalization
- Imports and Exports
- International Arrangements
- International Programs and Activities
- Partnerships and Collaboration
International Publications, Speeches, and News
Contact Us
- Contact OGPS
-
Hours Available

