Mammography: What You Need to Know
Learn about mammography and the FDA's certification of mammography facilities.
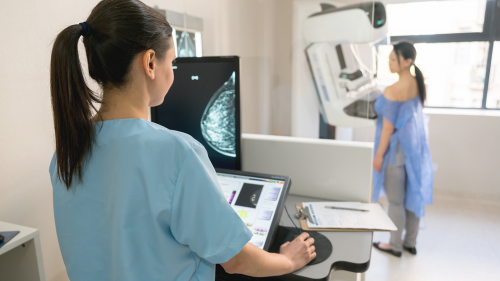
Mammograms continue to be the best primary tool for breast cancer screening. The U.S. Food and Drug Administration, along with some FDA-approved state certifying agencies, certify facilities to perform mammography. The FDA also clears, approves, and authorizes new mammography devices for sale in the U.S.
Congress enacted the Mammography Quality Standards Act (MQSA) in 1992 to ensure all women have access to quality mammography for the detection of breast cancer in its early, most treatable stages. Always look for the MQSA certificate at the mammography facility, which is required to be displayed and indicates that the facility meets the national baseline standards for mammography.
To continue to protect women's health, the FDA has updated mammography regulations to, among other things, reflect advances in mammography technology and processes since the current regulations were published.
How Does a Mammogram Work?
A mammogram is a series of low-dose X-ray images of the breasts. Getting a regular mammogram is the best primary screening method to find breast cancer early, because it can show growths in the breast or other signs of breast cancer when they are too small for you or your health care provider to feel them.
Thermograms and nipple aspirate tests are not substitutes for mammograms. Regular screening mammograms are important, and the risk of developing breast cancers varies from person to person, so it's a good idea to ask your health care provider when and how often you should schedule a mammogram.
To get a mammogram, you will need to take off your shirt and bra. While standing in front of the mammogram unit, a technologist will position your breast on a small platform. A clear plastic plate will press down on your breast while a mammogram image is acquired. This compression of the breast helps spread out the breast tissue so that it doesn't overlap as much, allowing for a clearer look at the breast tissue.
If you’re worried about how the procedure feels, you should know that most women do not find it painful. Some women may find the pressure on the breast uncomfortable, but it usually lasts for only a few seconds.
The FDA’s regulations already require that facilities provide patients an easy-to-understand summary of their mammography results within 30 days after the mammogram, and that they make reasonable attempts to communicate the results as soon as possible if indications of potential cancer are found.
The FDA has amended the regulations to require that facilities also provide you with information about whether your breast tissue is dense or not dense. Dense breasts have a higher proportion of fibroglandular tissue compared to fatty tissue. This is important, because dense breast tissue can make cancers more difficult to find using a mammogram; and it is also now known to be an independent risk factor for developing breast cancer.
In addition, the FDA is requiring that facilities advise you to talk to your health care provider about breast density, risks for breast cancer, and your individual situation. The idea is to provide information you can discuss with your provider to make better informed decisions, including if you need to consider any next steps.
If you do not receive your mammography summary report, call the provider who sent you for the mammogram to get your results, and contact the facility where you had the mammogram performed to get the report resent to you.
Call your health care provider if you notice any change in either of your breasts. A lump, thickening of or leakage from the nipple, or changes in how the nipple or skin looks can signal a potential problem.
Why Is Facility Certification Important?
Under the MQSA, mammography facilities must be certified by the FDA, or an FDA-approved state certifying agency, to provide mammography services. Certification is important because it indicates that a facility has met the MQSA requirements for practicing quality mammography. A high-quality mammogram can help detect breast cancer in its earliest, most treatable stages.
Each mammography facility is inspected every year. During the inspection, an FDA or FDA-trained inspector checks the facility’s equipment, staff training qualifications, and quality control records. Each facility also undergoes an in-depth accreditation process every three years to be eligible for an MQSA certificate.
The certificate, which is required to be prominently displayed, shows that the facility has met the MQSA quality standards and may legally perform mammography. When you arrive for your mammogram, look for the certificate. If you don’t see it, ask for where the certificate is in the facility.
What Are 3D Mammograms?
New breast imaging equipment must receive the FDA’s approval, clearance, or authorization before being sold in the U.S. The FDA has approved digital breast tomosynthesis (known as DBT or 3D) advanced mammography devices that can create multiple cross-sectional images of the breast from X-rays taken from multiple angles. These devices provide informative images of the breast tissue and may be helpful in evaluating dense breast tissue.
Before granting approval, the FDA determined there was a reasonable assurance that the new 3D devices were safe and effective for their intended use. This determination was based on a review of clinical studies involving multiple radiologists and hundreds of cases. The FDA also sought input on the safety and effectiveness of the devices from a panel of non-FDA clinical and technical experts.
Where Is the Closest Certified Facility?
There are more than 8,600 MQSA-certified facilities across the country. To find a certified mammography facility in your area, you can search the list on the FDA’s website by your zip code. The list is updated weekly.
How Will the Changes to the MQSA Regulations Help You?
The updates to the MQSA regulations would, among additional changes:
- Better inform you and your health care provider about your mammography results by providing specific information on breast density, an important risk factor for breast cancer.
- Strengthen the FDA's ability to suspend or revoke the certificates of facilities that are noncompliant with the regulations.
- Require facilities to provide mammography personnel access to their own records of MQSA-qualifying training and experience upon their request, so they may continue to provide mammography services.

