Import Offices and Ports of Entry
Contacting FDA Import Teams and Offices
The FDA's Import Program manages the review and oversight of all FDA-regulated products entering the United States.
How to Contact FDA Import Teams
- Entry Review: If your shipment is under initial review or documents have been requested, use ITACS to find status and provide additional information.
- See the FDA's Import Trade Communications Guide for instructions on how to contact the Entry Review Team.
- Field Examination/Sample: If your shipment is set up for examination or sample collection, provide locations for exam/sample via ITACS. If you need additional assistance, use the interactive map to contact the FDA import office that covers your port of entry.
- Compliance Review: If your shipment is under compliance review, contact the Compliance Officer listed on the Notice of FDA Action. You can obtain Notices of FDA Action via ITACS. See ITACS - How to Receive Electronic Notices of FDA Action.
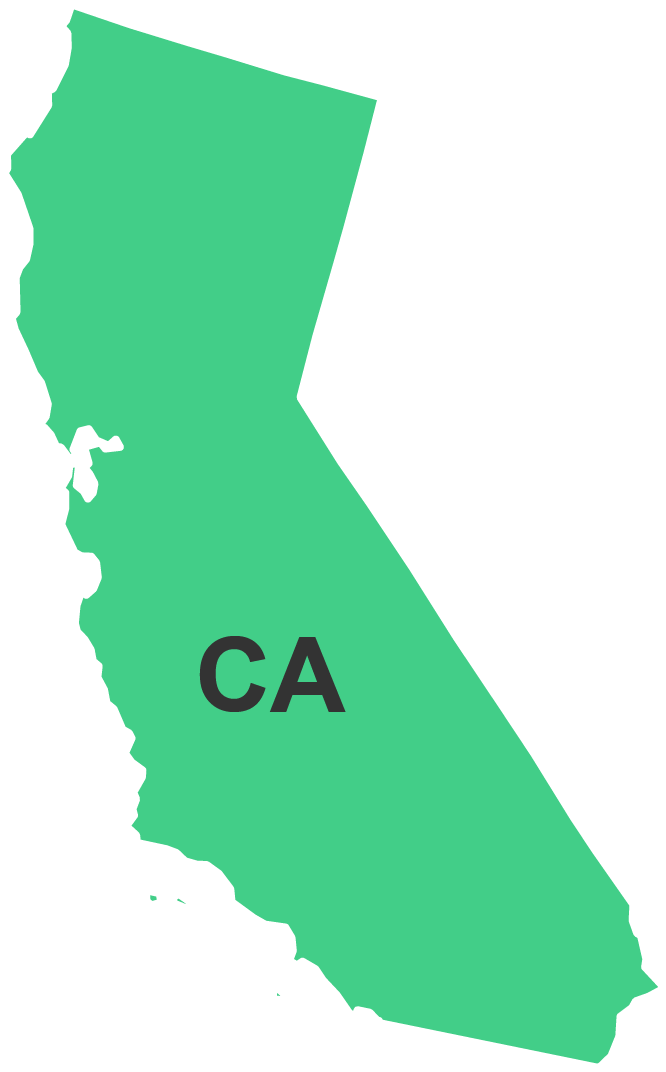
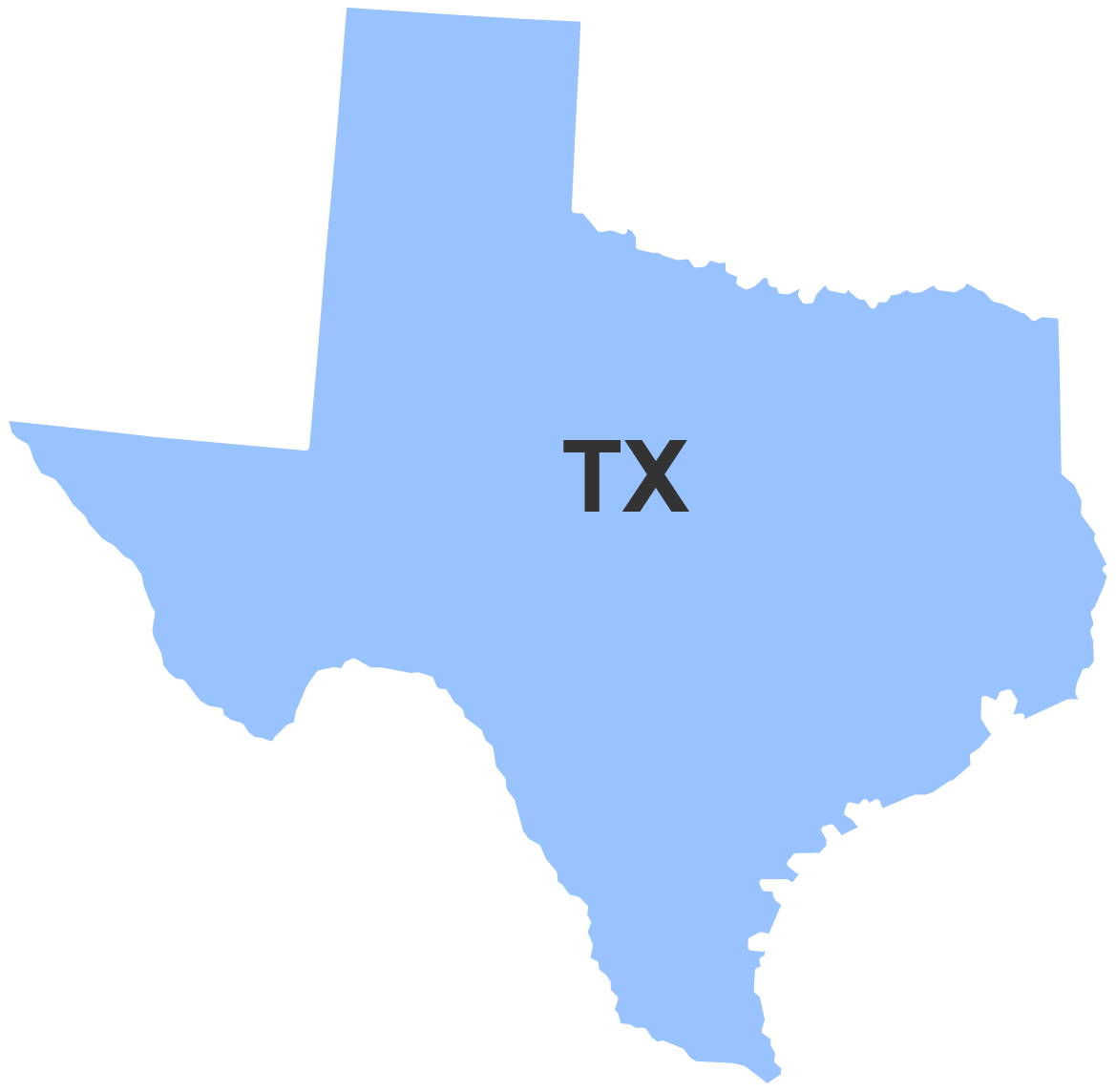
Interactive Contact Map
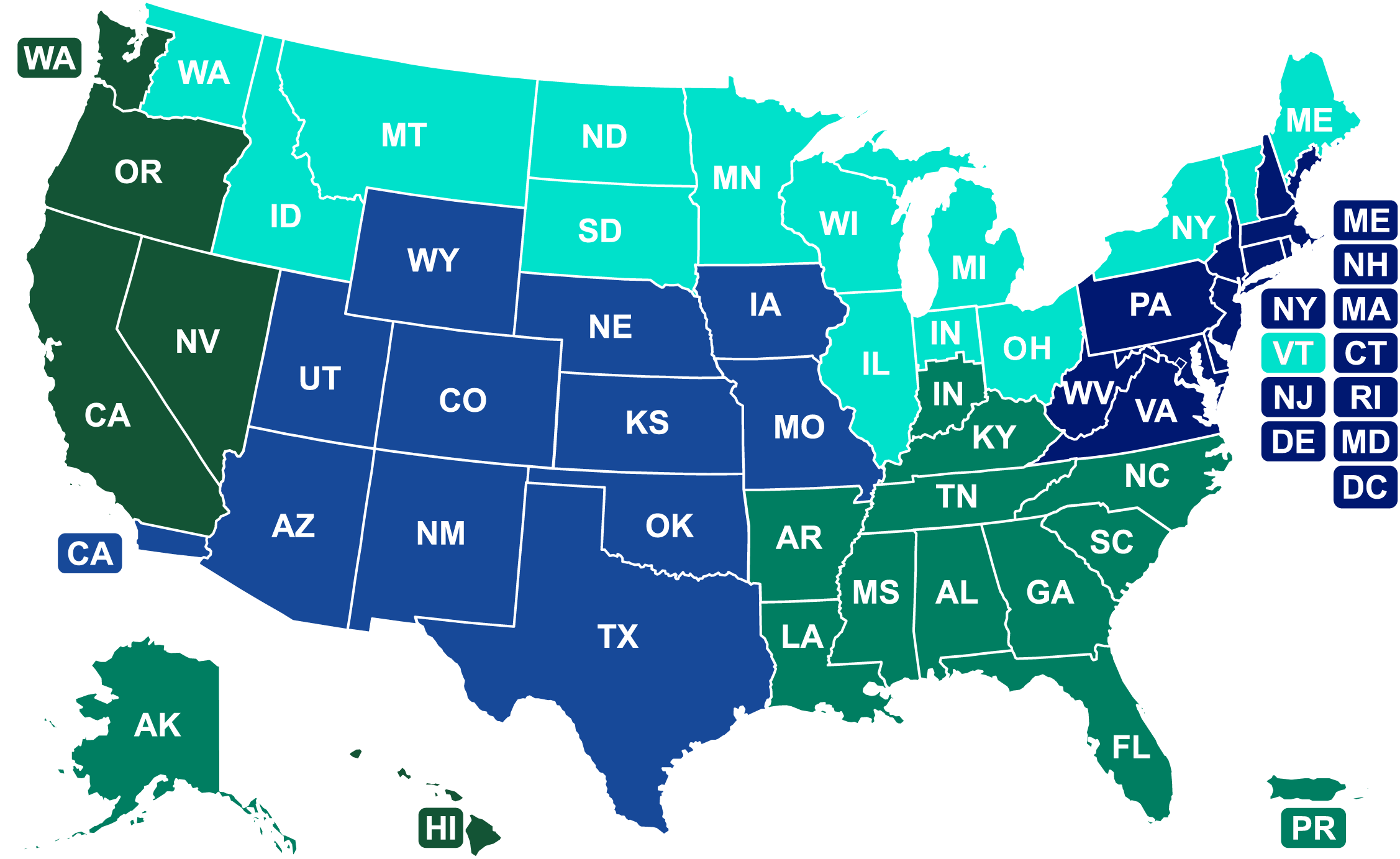

Division of Northeast Imports (DNEI)
Division of Northern Border Imports (DNBI)
Division of Southeast Imports (DSEI)
Division of Southwest Imports (DSWI)
Division of West Coast Imports (DWCI)
- FDA Import Page
- FDA Import Trade Communications Guide
- For general import operation or policy questions: imports@fda.hhs.gov
- For ACE related inquiries contact:
- For Industry FSVP Planning, use the FDA Data Dashboard
- For Industry VQIP Inquiries: fsmavqip@fda.hhs.gov
- OII Leadership Contacts - Import Program
- Product Code Builder