Use of the "Healthy" Claim on Food Labeling
On This Page
Products that Will Now Qualify for “Healthy” under the Final Rule
Updated Criteria for Certain Food Groups and Sample Foods
Education Materials
Updated “Healthy” Claim
Factsheet (PDF)
Infographic (PDF)
Updated “Healthy” Claim & What it Means for You
Factsheet (PDF) en Español (PDF)
Infographic (PDF) en Español (PDF)
The FDA announced on December 19, 2024, a final rule to update the “healthy” claim that manufacturers can voluntarily use on food packages. The updated claim is consistent with current nutrition science and Federal dietary guidance.
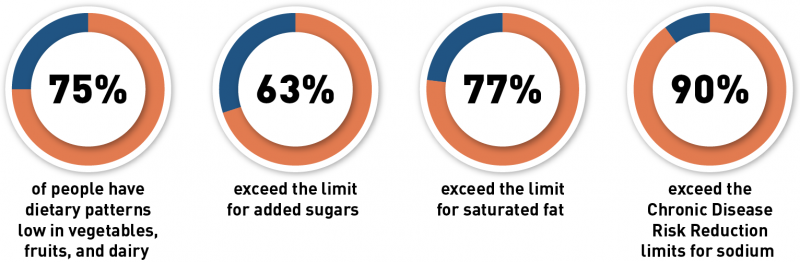
U.S. Dietary Intakes & Recommendations
Updated “Healthy” Claim
Claims like “healthy” on food labels can help consumers identify healthier food choices at a quick glance. Foods must meet specific criteria to use the “healthy” nutrient content claim. The updated criteria for the claim replace outdated criteria for “healthy” with criteria that are consistent with current nutrition science and Federal dietary guidance. For example, current U.S. dietary guidelines include a focus on the importance of healthy dietary patterns and the food groups that comprise them, the type of fat in the diet rather than the total amount of fat consumed, and the amount of sodium and added sugars in the diet. The updated criteria identify foods that help consumers build a healthy eating pattern.
To meet the updated criteria for the claim, a food product needs to
- contain a certain amount of food from at least one of the food groups or subgroups (such as fruit, vegetables, grains, fat-free and low-fat dairy and protein foods) recommended by the Dietary Guidelines for Americans, and
- meet specific limits for added sugars, saturated fat and sodium.
The criteria for how much food from a particular food group is required (called food group equivalents) and the specific limits for the three individual nutrients vary for individual food products, mixed products (which contain certain amounts of more than one food group), main dishes and meals, and are based on a Reference Amount Customarily Consumed, which is the basis for determining a serving size.
Products that Will Now Qualify for “Healthy” under the Final Rule
Under the updated ”healthy” claim, nutrient-dense foods that are encouraged by the Dietary Guidelines – vegetables, fruits, whole grains, fat-free and low-fat dairy, lean game meat, seafood, eggs, beans, peas, lentils, nuts, and seeds – with no added ingredients except for water, automatically qualify for the “healthy” claim because of their nutrient profile and positive contribution to an overall healthy diet. Under the updated claim, foods such as water, avocados, nuts and seeds, higher fat fish, such as salmon, and olive oil will now qualify to use the “healthy” claim. These include many varieties of these foods that fit into a range of budgets, for example, fruits, vegetables, and fish that are either fresh, frozen, or canned. Examples of products that qualified for “healthy” under the original claim that do not under the updated claim include fortified white bread, highly sweetened yogurt, and highly sweetened cereal.
Updated Criteria for Certain Food Groups and Sample Foods
Per Reference Amount Customarily Consumed
oz = ounce
g = grams
mg = milligrams
DV = Daily Value
| Food Groups | Food Group Equivalent Minimum | Added Sugar Limit | Sodium Limit | Saturated Fat Limit |
|---|---|---|---|---|
| Grains product | 3/4 oz whole-grain equivalent | 10% DV (5 g) | 10% DV (230 mg) | 5% DV (1 g) |
| Dairy product | 2/3 cup equivalent | 5% DV (2.5 g) | 10% DV (230 mg) | 10% DV (2 g) |
| Vegetable product | 1/2 cup equivalent | 2% DV (1 g) | 10% DV (230 mg) | 5% DV (1 g) |
| Fruit product | 1/2 cup equivalent | 2% DV (1 g) | 10% DV (230 mg) | 5% DV (1 g) |
| Protein Foods | ||||
| Game meat | 1 ½ oz equivalent | 2% DV (1 g) | 10% DV (230 mg) | 10% DV (2 g) |
| Seafood | 1 oz equivalent | 2% DV (1 g) | 10% DV (230 mg) | 5% DV (1 g)* |
| Egg | 1 egg | 2% DV (1 g) | 10% DV (230 mg) | 10% DV (2 g) |
| Beans, peas, and lentils | 1 oz equivalent | 2% DV (1 g) | 10% DV (230 mg) | 5% DV (1 g) |
| Nuts, seeds, and soy products | 1 oz equivalent | 2% DV (1 g) | 10% DV (230 mg) | 5% DV (1 g)* |
| Oils | ||||
| 100% Oil | N/A | 0% DV | 0% DV | 20% of total fat |
| Oil-based Spreads | N/A | 0% DV | 10% DV (230 mg) | 20% of total fat |
| Oil-based Dressing** | N/A | 2% DV (1 g) | 10% DV (230 mg) | 20% of total fat |
| * Excluding saturated fat inherent in nuts, seeds, soy products, and seafood ** Must contain at least 30% oil | ||||
| Sample Foods | |||
|---|---|---|---|
Individual food | Mixed product | Meal | |
| Amount of food groups required | 1 food group equivalent* 2/3 cup yogurt | 1 total food group equivalent with at least 1/4 food group equivalent from 2 or more food groups For example, 1/8 cup dried fruit and 1/4 oz nuts | 3 total food group equivalents with at least 1/2 food group equivalent from 3 or more food groups For example, 1 oz salmon, 1/2 cup green beans, 3/4 oz brown rice |
| Nutrients to Limit (no more than)** | 2 g saturated fat 230 mg sodium 2.5 g added sugar | 2 g saturated fat*** 345 mg sodium 5 g added sugar | 4 g saturated fat*** 690 mg sodium 10 g added sugar |
| * A food group equivalent is the minimum amount of a food group required ** Amounts based on percentage of the Daily Value for that nutrient *** Saturated fat inherent in nuts, seeds, soy products, and seafood does not contribute to limit | |||
Previous Actions
On September 29, 2022, the FDA issued a proposed rule to update the nutrient content claim “healthy,” which was set in 1994. The previous definition had limits for total fat, saturated fat, cholesterol and sodium and to qualify, foods also had to provide at least 10% of the Daily Value (DV) for one or more of the following nutrients: vitamin A, vitamin C, calcium, iron, protein, and fiber. The proposed rule was informed in part by a public meeting the FDA held on March 9, 2017 to discuss use of the term “healthy” in food labeling.
During the FDA’s consideration of how to update the "healthy” claim, in September 2016, the FDA issued a guidance that states the agency’s intent to exercise enforcement discretion for products labeled with a “healthy” claim that meet certain nutrient requirements.
Also in September 2016, the FDA issued a request for information and comments related to the use of the term “healthy” in food labeling.
“Healthy” Symbol
On a separate but related track, the FDA is also continuing to explore development of a symbol that manufacturers could use on food labeling to show that a product meets the definition of “healthy.” Having a standardized graphic to show that a food meets the criteria for the “healthy” claim would further support the FDA’s goal of helping consumers to identify food products that can be the foundation of healthy eating patterns.
The FDA issued two procedural notices for public comment when seeking Office of Management and Budget approval for the “healthy” symbol’s preliminary quantitative consumer research. The first notice was issued in May 2021 and the second notice was issued in March 2022, and the FDA’s research was approved. The FDA continues to work on development of the symbol.
FDA Nutrition Goals
The U.S. faces an ever-growing epidemic of preventable diet-related chronic diseases such as cardiovascular disease, type 2 diabetes and obesity. Improving nutrition is one of the best public health interventions for reducing these and other chronic illnesses and premature death. The FDA promotes nutritious eating patterns, in part, by providing information so that consumers can make healthier food choices, and by making changes that can result in a healthier food supply.