Counterfeit Version of Botox Found in the United States
[4-16-2015] FDA is alerting health care practitioners and the public that a counterfeit version of Botox was found in the United States and may have been sold to doctors’ offices and medical clinics nationwide. The product was sold by an unlicensed supplier who is not authorized to ship or distribute drug products in the United States.
The counterfeit products are considered unsafe and should not be used. FDA cannot confirm that the manufacture, quality, storage, and handling of these suspect products follow U.S. standards.
There are some similarities between the counterfeit Botox products and the FDA-approved Botox for injection (100 units/vial), manufactured by Allergan. The FDA-approved Botox displays the active ingredient as “OnabotulinumtoxinA” on the outer carton and vial. Currently, there is no indication that Allergan’s FDA-approved version is at risk, and the genuine product should be considered safe and effective for its intended and approved uses.
Both the outer carton and vial on the suspect product are counterfeit. The counterfeit product can be identified by one or more of the following:
- the vial is missing the lot number
- the outer carton does not have any entries next to the LOT: MFG: EXP:
- the outer carton and vial display the active ingredient as “Botulinum Toxin Type A” instead of “OnabotulinumtoxinA”
|
Authentic Vial |
Counterfeit Vial | Counterfeit Package |
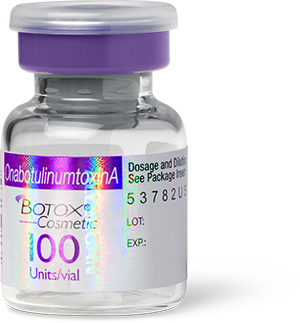 |
  |
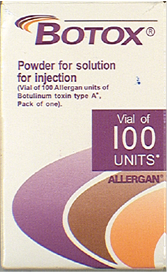  |
Health practitioners should check with Allergan to make sure that the distributor that they purchase from is authorized to distribute Botox. To access a list of authorized Botox suppliers, visit Allergan’s website.
Medications purchased from foreign or unlicensed sources may be misbranded, adulterated, counterfeit, contaminated, improperly stored and transported, ineffective, and/or unsafe. Medical practices that purchase and administer counterfeit, illegal, and unapproved medications from unlicensed or foreign sources are putting patients’ health at risk, as patients may not be getting proper treatment. Wholesale drug distributers must be licensed in the states where they conduct business.
FDA is asking the public to report suspect Botox products to FDA:
- Call FDA’s Office of Criminal Investigations (OCI) at 800-551-3989, or
- Report to OCI at www.accessdata.fda.gov/scripts/email/oc/oci/contact.cfm, or
- Email - DrugSupplyChainIntegrity@fda.hhs.gov
FDA is not aware of any adverse events associated with the counterfeit version of Botox at this time.
FDA encourages health care professionals and patients to report adverse events related to the use of any suspect medications to FDA’s MedWatch Safety Information and Adverse Event Reporting Program either online, by regular mail, by fax, or by phone. Health care professionals and consumers can either:
- Complete and submit the report online: www.fda.gov/MedWatch/report.htm, or
- Download form or call 1-800-332-1088 to request a reporting form, then complete and return to the address on the pre-addressed form, or submit by fax to 1-800-FDA-0178. FDA has issued a series of alerts and letters to practitioners about the risk of buying medicines from foreign or unlicensed sources.
The Food and Drug Administration is committed to protecting public health by securing the drug supply chain against counterfeit and unapproved medications that enter the United States through fraudulent sources. Visit FDA’s website to access information about how to safely purchase prescription drugs for your patients: Know Your Source: Protecting Patients from Unsafe Drugs.

