Drug Trials Snapshots: REBLOZYL
HOW TO USE THIS SNAPSHOT
The information provided in Snapshots highlights who participated in the clinical trials that supported the FDA approval of this drug, and whether there were differences among sex, race and age groups. The “MORE INFO” bar shows more detailed, technical content for each section. The Snapshot is intended as one tool for consumers to use when discussing the risks and benefits of the drugs.
LIMITATIONS OF THIS SNAPSHOT:
Do not rely on Snapshots to make decisions regarding medical care. Always speak to your health provider about the risks and benefits of a drug. Refer to the REBLOZYL Package Insert for complete information.
REBLOZYL ( luspatercept-aamt)
REB-loh-zil
Celgene Corporation
Approval date: November 8, 2019
DRUG TRIALS SNAPSHOT SUMMARY:
What is the drug for?
REBLOZYL is a drug for the treatment of anemia in adults with beta thalassemia who require regular red blood cell transfusions.
Beta thalassemia is an inherited disorder of hemoglobin leading to anemia and other medical problems. Some patients may have severe anemia and need regular blood transfusions.
How is this drug used?
REBLOZYL is an injection given under the skin (subcutaneously) every three weeks.
What are the benefits of this drug?
REBLOZYL was better than placebo in decreasing the need for red blood cell transfusions. In the trial 21% of patients receiving REBLOZYL achieved a 33% reduction in RBC transfusion in comparison to 5% of patients who received placebo.
What are the benefits of this drug (results of trials used to assess efficacy)?
The table below presents efficacy results from the trial based upon the proportion of patients achieving RBC transfusion burden reduction (≥33% reduction from baseline) with a reduction of at least 2 units from week 13 to week 24.
Table 2. Efficacy Results in Beta Thalassemia
|
Endpoint |
REBLOZYL |
Placebo |
Risk Difference |
p-value |
|
≥33% Reduction from baseline in RBC transfusion burden with a reduction of at least 2 units for 12 consecutive weeks |
|
|
|
|
|
Primary endpoint – Week 13 to Week 24 |
48 (21.4) |
5 (4.5) |
17.0 |
<0.0001 |
|
Week 37 to Week 48 |
44 (19.6) |
4 (3.6) |
16. |
<0.0001 |
Adapted from Prescribing Information
Were there any differences in how well the drug worked in clinical trials among sex, race and age?
- Sex: REBLOZYL worked similarly in men and women.
- Race: REBLOZYL worked imilarly in all tested race groups.
- Age: REBLOZYL worked similarly in all tested age groups. There were not enough patients older than 65 years to determine whether there was any difference between older and younger patients.
Were there any differences in how well the drug worked in clinical trials among sex, race, and age groups?
The table below presents subgroup analyses based on primary endpoint.
Table 4. Summary of Subgroup Analyses by Sex, Race and Age in Trial 1 (ITT population)
|
|
Responders/N |
ORR (95% CI) |
||
|
REBLOZYL |
Placebo |
REBLOZYL |
Placebo |
|
|
Primary Analysis |
48 / 224 |
5 / 112 |
21.4 (16.2, 27.4) |
4.5 (1.5, 10.1) |
|
Sex |
||||
|
Male |
13 / 92 |
1 / 49 |
14.1 (7.7, 23.0) |
2.0 (0.1, 10.9) |
|
Female |
35 / 132 |
4 / 63 |
26.5 (19.2, 34.9) |
6.3 (1.8, 15.5) |
|
Race |
||||
|
White |
30 / 122 |
2 / 60 |
24.6 (17.2, 33.2) |
3.3 (0.4, 11.5) |
|
Asian |
12 / 81 |
2 / 36 |
14.8 (7.9, 24.4) |
5.6 (0.7, 18.7) |
|
Other Race |
6 / 21 |
1 / 16 |
28.6 (11.3, 52.2) |
6.3 (0.2, 30.2) |
|
Age (years) |
||||
|
Age ≤ 32 |
22 / 129 |
4 / 63 |
17.1 (11.0, 24.7) |
6.3 (1.8, 15.5) |
|
Age > 32 |
26 / 95 |
1 / 49 |
27.4 (18.7, 37.5) |
2.0 (0.1, 10.9) |
|
Region |
||||
|
North America and Europe |
23 / 100 |
1 / 51 |
23.0 (15.2, 32.5) |
2.0 (0.0, 10.4) |
|
Asia-Pacific |
14 / 72 |
2 / 35 |
19.4 (11.1, 30.5) |
5.7 (0.7, 19.2) |
|
Middle East & North Africa |
11 / 52 |
2 / 26 |
21.2 (11.1, 34.7) |
7.7 (0.9, 25.1) |
Adapted from FDA Review
What are the possible side effects?
REBLOZYL can cause serious side effects including increased risk for blood clots and high blood pressure. REBLOZYL may cause harm to unborn babies.
The most common side effects of REBLOZYL are headache, bone pain and joint arthralgia, fatigue, cough, abdominal pain, diarrhea, and dizziness.
What are the possible side effects (results of trials used to assess safety)?
The table below presents adverse reactions occurring at a rate of > 5% in the REBLOZYL treated group, and for which the rate for REBLOZYL exceeds the rate for placebo for at least 1%.
Table 6. Adverse Drug Reactions (>5%) in Patients Receiving REBLOZYL with a Difference Between Arms of 1% in the Trial
|
Body System |
REBLOZYL |
Placebo |
||
|
All Grades |
Grades ≥3 a |
All Grades |
Grades ≥3 |
|
|
Musculoskeletal and connective tissue disorders |
||||
|
Bone Pain |
44 (20) |
3 (1) |
9 (8) |
0 (0) |
|
Arthralgia |
43 (19) |
0 (0) |
13 (12) |
0 (0) |
|
Infections and infestation |
||||
|
Influenza |
19 (9) |
0 (0) |
6 (6) |
0 (0) |
|
Viral Upper Respiratory Infection |
14 (6) |
1 (0.4) |
2 (2) |
0 (0) |
|
Nervous system disorders |
||||
|
Headache |
58 (26) |
1 (<1) |
26 (24) |
1 (1) |
|
Dizziness |
25 (11) |
0 (0) |
5 (5) |
0 (0) |
|
General disorders and administration site conditions |
||||
|
Fatigue |
30 (14) |
0 (0) |
14 (13) |
0 (0) |
|
Gastrointestinal disorders |
||||
|
Abdominal Pain b |
31 (14) |
0 (0) |
13 (12) |
0 (0) |
|
Diarrhea |
27 (12) |
1 (<1) |
11 (10) |
0 (0) |
|
Nausea |
20 (9) |
0 (0) |
6 (6) |
0 (0) |
|
Vascular disorders |
||||
|
Hypertension c |
18 (8) |
4 (2) |
3 (3) |
0 (0) |
|
Metabolism and nutrition disorders |
||||
|
Hyperuricemia |
16 (7) |
6 (3) |
0 (0) |
0 (0) |
|
Respiratory, thoracic and mediastinal disorders |
||||
|
Cough |
32 (14) |
0 (0) |
12 (11) |
0 (0) |
Prescribing Information
Were there any differences in side effects among sex, race and age?
- Sex: The occurrence of side effects was similar in men and women.
- Race: The occurrence of side effects was similar in all tested race groups.
- Age: The occurrence of side effects was similar in all tested age groups. There were not enough patients older than 65 years to determine whether there was any difference between older and younger patients.
Were there any differences in side effects of the clinical trials among sex, race, and age groups?
Subgroup analyses of adverse events are presented in Tables 7-9.
Table 7. Adverse Events by Sex Subgroup
|
Adverse Events |
REBLOZYL (N=223) |
Placebo (N=109) |
||
|
Men |
Women |
Men |
Women |
|
|
Any TEAE, x (%) |
87 (95.6) |
127 (96.2) |
44 (91.7) |
57 (93.4) |
|
SAE, x (%) |
16 (17.6) |
18 (13.6) |
2 (4.2) |
4 (6.6) |
Table 8. Adverse Events by Race Subgroup
|
Adverse Events |
REBLOZYL (N=223) |
Placebo (N=109) |
||
|
White (n=121) |
Other |
White |
Other |
|
|
Any TEAE, x (%) |
116 (95.6) |
93 (95.9) |
55 (94.8) |
41 (89.1) |
|
SAE, x (%) |
19 (15.7) |
14 (14.4) |
3 (5.2) |
3 (6.5) |
Table 9. Adverse Events by Age Subgroup
|
Adverse Events |
REBLOZYL (N=223) |
Placebo (N=109) |
||
|
≤32 years |
>32 years |
≤32 years |
>32 years |
|
|
Any TEAE, x (%) |
121 (93.8) |
93 (98.9) |
57 (90.5) |
44 (95.7) |
|
SAE, x (%) |
18 (14) |
16 (17) |
5 (7.9) |
1 (2.2) |
TEAE=treatment -emergent adverse events; SAE=serious adverse events
Clinical Trial Data
WHO WAS IN THE CLINICAL TRIALS?
Who participated in the clinical trials?
The FDA approved REBLOZYL based primarily on evidence from one clinical trial (Trial 1/ NCT02604433) of 336 adult patients 18-66 years old with beta thalassemia. The trial was conducted at 65 sites in 15 countries (Australia, Malaysia, Taiwan, Thailand, Israel, Lebanon, Tunisia, Turkey, Bulgaria, Canada, France, Greece, Italy, the United Kingdom, and the United States).
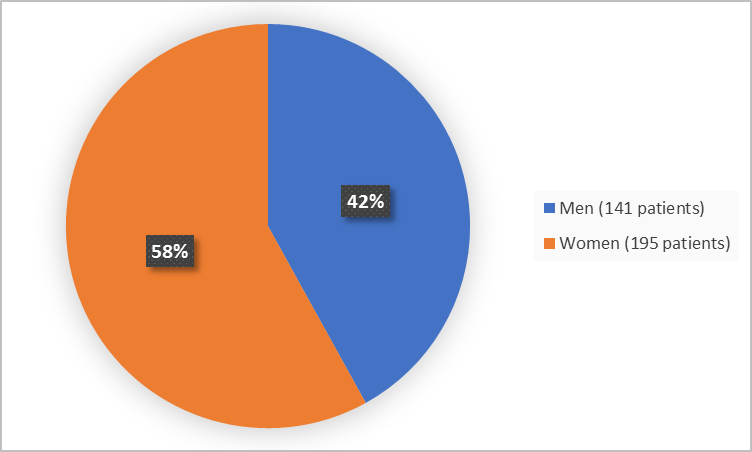
Figure 1 summarizes how many men and women were in the clinical trial.
Figure 1. Demographics by Sex
FDA Review
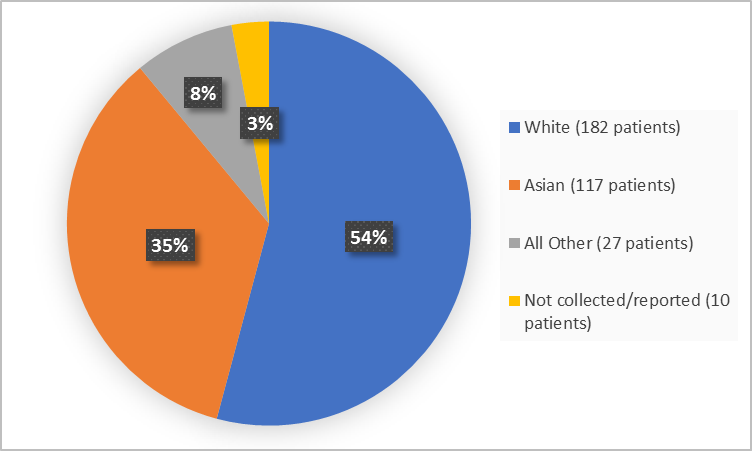
Figure 2 summarizes the percentage of patients by race in the clinical trial.
Figure 2. Demographics by Race
FDA Review
Table 1. Demographics by Race
|
Race |
Number of Patients |
Percentage |
|
White |
182 |
54 |
|
Black or African American |
1 |
Less than 1 |
|
Asian |
117 |
35 |
|
Other |
26 |
8 |
|
Not collected or not reported |
10 |
3 |
FDA Review
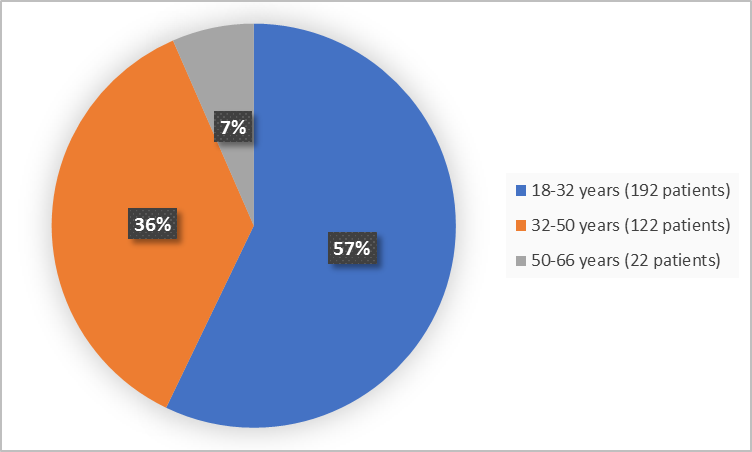
Figure 3 summarizes the percentage of patients by age group in the clinical trial.
Figure 3. Demographics by Age
Clinical Trial Data
Who participated in the trials
Demographics data from Trial 1 representing the efficacy population is presented below.
Table 10. Trial Demographics
|
Demographic Characteristic |
REBLOZYL |
Placebo |
Total |
|
Sex |
|
|
|
|
Women |
132 (58.9) |
63 (56.3) |
195 (58) |
|
Men |
92 (41.1) |
49 (43.8) |
141 (42) |
|
Race |
|
|
|
|
Asian |
81 (36.2) |
36 (32.1) |
117(34.8) |
|
Black or African |
1 (0.4) |
0 (0.0) |
1 (0.3) |
|
White |
122 (54.5) |
60 (53.6) |
182 (54.2) |
|
Other |
15 (6.7) |
11 (9.8) |
26 (7.7) |
|
Not Collected or |
5 (2.2) |
5 (4.5) |
10 (2.9) |
|
Age (years) |
|
|
|
|
Mean (SD) |
32.2 (10.7) |
31.9 (9.9) |
32.1 (10.40) |
|
Median (Range) |
30 (18, 66) |
30 (18, 59) |
30 (18,66) |
|
Age Group (years) |
|
|
|
|
≤32 |
129 (57.6) |
63 (56.3) |
192 (57.1) |
|
>32 - ≤50 |
78 (34.8) |
44 (39.3) |
122 (36.3) |
|
>50 |
17 (7.6) |
5 (4.5) |
22 (6.5) |
|
Ethnicity |
|
|
|
|
Hispanic or Latino |
5 (2.2) |
2 (1.8) |
7 (2.1) |
|
Not Hispanic or Latino |
218 (97.3) |
107 (95.5) |
325 (96.7) |
|
Missing |
1 (0.4) |
3 (2.7) |
4 (1.2) |
|
Region |
|
|
|
|
Asia-Pacific |
72 (32.1%) |
35 (31.3%) |
107 (31.8) |
|
Middle East and |
52 (23.2%) |
26 (23.2%) |
78 (23.2) |
|
North America and |
100 (44.6%) |
51 (45.5%) |
151 (44.9) |
|
US only |
18 (8) |
9 (8) |
27 (8) |
Adapted from FDA Review
How were the trials designed?
REBLOZYL was evaluated in one clinical trial of 336 adult patients with beta thalassemia who required regular red blood cell transfusions (6-20 red blood cell units per 24 weeks).
In the trial, patients were randomly assigned to receive either REBLOZYL or a matched placebo every three weeks subcutaneously as long as they required less transfusions or unacceptable toxicity occurred. Neither the patients nor the investigators knew which treatment was given until the end of the trial.
The benefit of REBLOZYL was assessed by proportion of patients whose need for transfusion was reduced at least 33% and comparing it to the proportion of placebo- treated patients who also achieved at least 33% transfusion reduction.
How were the trials designed?
REBLOZYL was evaluated in a multicenter, randomized, double-blind, placebo-controlled trial which enrolled patients with beta thalassemia requiring regular red blood cell transfusions (6-20 RBC units per 24 weeks) with no transfusion-free period greater than 35 days. REBLOZYL was administered subcutaneously once every 3 weeks as long as a reduction in transfusion requirement was observed or until unacceptable toxicity. All patients were eligible to receive best supportive care.
The major efficacy outcome was the proportion of patients achieving RBC transfusion burden reduction (≥33% reduction from baseline) with a reduction of at least 2 units from week 13 to week 24.
GLOSSARY
CLINICAL TRIAL: Voluntary research studies conducted in people and designed to answer specific questions about the safety or effectiveness of drugs, vaccines, other therapies, or new ways of using existing treatments.
COMPARATOR: A previously available treatment or placebo used in clinical trials that is compared to the actual drug being tested.
EFFICACY: How well the drug achieves the desired response when it is taken as described in a controlled clinical setting, such as during a clinical trial.
PLACEBO: An inactive substance or “sugar pill” that looks the same as, and is given the same way as, an active drug or treatment being tested. The effects of the active drug or treatment are compared to the effects of the placebo.
SUBGROUP: A subset of the population studied in a clinical trial. Demographic subsets include sex, race, and age groups.