New Approach Methodologies (NAMs)
Introduction | Definitions | FDA’s NAMs Activities
Selected Guidance | Other Resources | Contact Information
FDA Achieves Year 1 Goals in Reducing Animal Testing in Drug Development
April 20, 2026
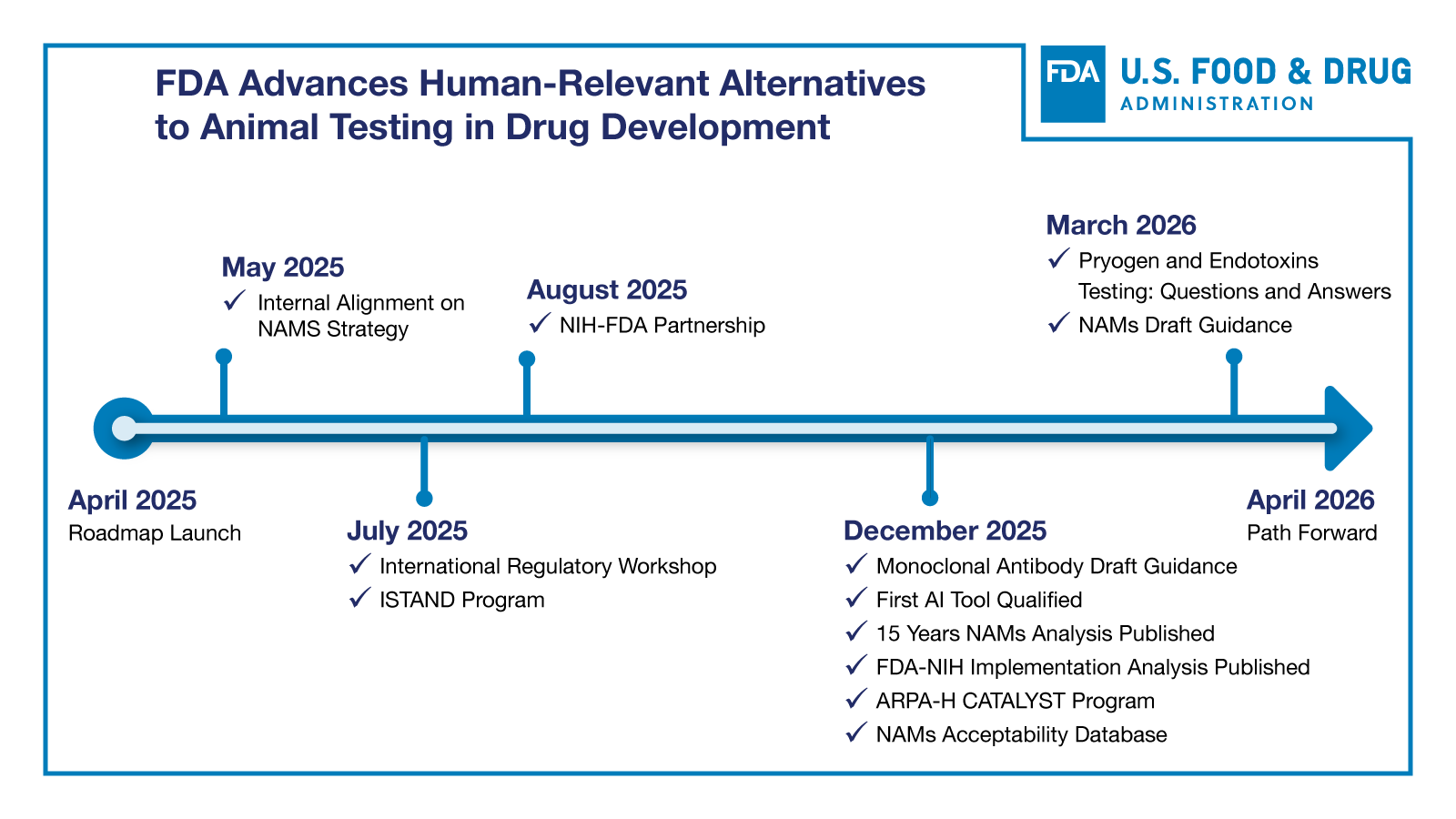
Since publishing a roadmap last April, the FDA has successfully launched several key initiatives to replace animal testing with better alternatives. The agency released a follow-up report, Reducing Animal Testing in Nonclinical Studies: Year One Progress and the Path Forward, summarizing the agency’s progress in implementing the roadmap and clarifying its next steps.
The U.S. Food and Drug Administration is leading a transformative shift in drug safety evaluation—reducing, replacing or refining animal testing with advanced, human-relevant methods that better predict how medicines work in people. Over 90% of drugs safe in animals fail in humans, yet some human-safe medications like aspirin might never have passed animal tests. Through New Approach Methodologies (NAMs)—including AI-powered models, human organ-on-a-chip systems, and real-world data—we're accelerating safer treatments to patients faster while reducing costs and sparing thousands of laboratory animals annually. This represents a win for patients, innovation, and ethics.
Streamlines Non-Clinical Studies and Acceptable NAMs
Innovative Science and Technology Approaches for New Drugs (ISTAND) Program
Critical Path Innovation Meetings (CPIM)
Definitions
New Approach Methodologies (NAMs) are innovative testing methods and strategies used to assess the safety, efficacy, and quality of drugs, biologics, and other FDA-regulated products. NAMs encompass in vitro human-based systems, in silico modeling, and other innovative platforms that can collectively evaluate immunogenicity, toxicity, and pharmacodynamics in humans and provide an opportunity to improve the predictive relevance of nonclinical drug testing while reducing or replacing animal use.
FDA’s NAMs Activities
Reducing Animal Testing in Nonclinical Studies: Year One Progress and the Path Forward. Released in April 2025, this landmark document outlines a strategic, stepwise approach for transitioning from animal-based testing to validated New Approach Methodologies (NAMs) that better predict human responses. Beginning with monoclonal antibodies—where animal models have proven particularly unreliable—and potentially expanding to other biological molecules and eventually new chemical entities and medical countermeasures, the roadmap establishes clear timelines, validation frameworks, and success metrics for this historic shift.
FDA-NIH Workshop: Reducing Animal Testing. On Monday, July 7, 2025, the FDA hosted a hybrid workshop on reducing animal testing. The agenda focused on implementing novel methodologies and other strategies to reduce the use of animal testing. Featured speakers from the FDA, NIH and representatives from international regulatory bodies discussed successes, challenges and ideas for implementation.
Selected Guidance
Oncology Pharmaceuticals: Streamlined Nonclinical Safety Studies for Biologics and Conjugated Products (Draft, OCE 2026)
The purpose of this guidance is to assist sponsors in implementing streamlined approaches for nonclinical safety assessments of certain oncology pharmaceuticals. This guidance is intended to facilitate drug development for biologics and conjugated products for the treatment of cancer while avoiding unnecessary animal use.
General Considerations for the Use of New Approach Methodologies in Drug Development (Draft, CDER, 2026)
Provides a validation framework and regulatory expectations for using non-animal methods (e.g., in vitro, in silico) in drug development submissions to replace or reduce animal toxicology studies
Monoclonal Antibodies: Streamlined Nonclinical Safety Studies (Draft Guidance for Industry, CDER, 2025)
Provides recommendations to eliminate or reduce long-term primate toxicity studies for certain monoclonal antibodies using alternative data sources such as computational models and human-relevant systems with a weight-of-evidence (WoE) assessment.
Pyrogen and Endotoxins Testing – Questions and Answers (Updated Guidance for Industry, 2026)
Supports replacement of animal-based pyrogen tests (e.g., rabbit test) with non-animal methods such as recombinant reagent-based endotoxin assays in manufacturing quality control.
Assessing the Credibility of Computational Modeling and Simulation in Medical Device Submissions (Guidance for Industry, 2023)
Describes risk-based framework, including context of use examples, that can be used in the credibility assessment of computational modeling and simulation (CM&S) used in medical device regulatory submissions.
S5(R3) Detection of Reproductive and Developmental Toxicity for Human Pharmaceuticals (Guidance for Industry, 2021)
Description of testing strategies utilizing alternative assays for the assessment of malformations and embryofetal lethality, and the qualification process for these alternative assays.
Oncology Pharmaceuticals: Reproductive Toxicity Testing and Labeling Recommendations (Guidance for Industry, 2019)
Potential use of alternative assays, such as fit-for-purpose in vitro or ex vivo, or nonmammalian in vivo assays for assessment of reproductive toxicity.
S3A Guidance: Note for Guidance on Toxicokinetics: The Assessment of Systemic Exposure in Toxicity Studies: Focus on Microsampling Questions and Answers (Guidance for Industry, 2018)
Use of microsampling in toxicity studies.
M7(R1) Assessment and Control of DNA Reactive (Mutagenic) Impurities in Pharmaceuticals to Limit Potential Carcinogenic Risk (Guidance for Industry, 2018)
Use of computational approaches for the assessment of mutagenic potential of drug impurities.
S10 Photosafety Evaluation of Pharmaceuticals (Guidance for Industry, 2015)
Use of in chemico and in vitro approaches to assess phototoxicity potential.
OECD Test Guideline No. 437
Reconstructed human cornea-like epithelium model replaced rabbit tests for eye irritation for pharmaceuticals
OECD Test Guideline No. 439
A 3D reconstructed human epidermis model is accepted for human pharmaceuticals, when warranted, to assess primary dermal irritation
Other Resources
Roadmap to Reducing Animal Testing in Preclinical Safety Studies
Outlines agency-wide strategy to phase down animal testing by promoting New Approach Methodologies (NAMs), including AI models, organ-on-chip systems, and cell-based assays
CDER/Office of New Drugs Streamlined Nonclinical Studies and Acceptable New Approach Methodologies (NAMs)
Contains an inventory of drug development contexts for which CDER is open to a streamlined nonclinical program. This includes but is not limited to reduced sample size, reduced number of species tested, and NAMs use.
A CDER perspective: Landscape of New Approach Methodologies (NAMs) submitted in drug development programs
This study highlights one of CDER's activities aimed at better understanding the current usage of NAMs in drug development, while providing evidence to support areas of focus and prioritization of resources towards the validation of NAMs with significant regulatory impact potential.
FDA/CDER/OND Experience With New Approach Methodologies (NAMs)
This study demonstrates how FDA/CDER has historically incorporated NAMs into standard nonclinical assessments, describing how specific tests became validated and internationally accepted alternatives to animal testing for regulatory decision-making.
Liver Toxicity Knowledge Base (LTKB)
TKB is a project at the FDA's National Center for Toxicological Research to study drug-induced liver injury (DILI). The project involves the collection of diverse data (e.g., DILI mechanisms, drug metabolism, histopathology, therapeutic use, targets, side effects, etc.) associated with individual drugs and the use of systems biology analysis to integrate these data for DILI assessment and prediction.
Collection of Alternative Methods for Regulatory Application (CAMERA)
CAMERA is an interactive database and user interface providing online access to validated alternative methods for U.S. regulatory and other contexts of use.
NIH Complement Animal Research In Experimentation (Complement-ARIE) Program
The NIH Common Fund’s Complement Animal Research In Experimentation (Complement-ARIE) program speeds the development, standardization, validation, and use of human-based New Approach Methodologies (NAMs).
Contact Information
For more information on NAMs, please visit the individual Center or program pages, or contact alternatives@FDA.hhs.gov.