COMPANY ANNOUNCEMENT
Mylan Initiates Voluntary Nationwide Recall of One Lot of Daptomycin for Injection, Due to The Presence of Particulate
This recall has been completed and FDA has terminated this recall.
When a company announces a recall, market withdrawal, or safety alert, the FDA posts the company's announcement as a public service. FDA does not endorse either the product or the company.
Read Announcement View Product PhotosSummary
- Company Announcement Date:
- FDA Publish Date:
- Product Type:
- Drugs
- Reason for Announcement:
-
Recall Reason DescriptionPresence of particulate matter
- Company Name:
- Mylan Institutional LLC
- Brand Name:
-
Brand Name(s)Mylan
- Product Description:
-
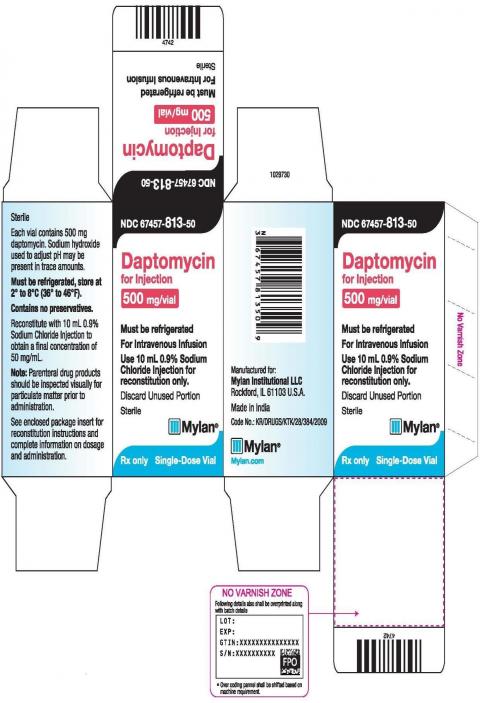
Product DescriptionDaptomycin for Injection
Company Announcement
Mylan N.V. (NASDAQ: MYL) today announced that its U.S.-based Mylan Institutional LLC business is conducting a voluntary nationwide recall to the consumer level of one lot of Daptomycin for Injection, 500 mg/vial due to the presence of particulate matter found in one single-dose vial manufactured by Mylan Laboratories Limited’s Specialty Formulation Facility. To date, Mylan has not received any reports of adverse events related to this recall.
Intravenous administration of a solution containing visible particulates could lead to serious adverse events including, but not limited to, local irritation, vasculitis/phlebitis, antigenic or allergic reactions, and microvascular obstruction, including pulmonary embolism.
This batch was distributed nationwide to wholesalers and retail pharmacies between April 2020 and May 2020. The recalled batch is as follows:
| NDC # | Material Description | Strength | Size | Lot No | Expiry |
|---|---|---|---|---|---|
| 67457-813-50 | Daptomycin for Injection | 500 mg/vial | 20 mL vial | 7605112 | October 2021 |
Daptomycin for injection is an injectable antibacterial indicated for the treatment of complicated skin and skin structure infections (cSSSI) and staphylococcus aureus bloodstream infections (bacteremia) in adult patients.
Mylan is notifying its distributors and customers by letter and is arranging for return of all recalled products. Consumers/distributors/retailers that have product which is being recalled should stop use/further distribution or dispensing. Wholesalers, retailers and consumers that are in possession of recalled product should contact Stericycle at 1-888-641-9736 for the return of the recalled product. Normal business hours are Monday through Friday 8 a.m. to 5 p.m. EST.
Consumers with questions regarding this recall can contact Mylan Customer Relations at 800.796.9526 or customer.service@mylan.com, Monday through Friday from 8 a.m. – 5 p.m. EST. Consumers should contact their physician or healthcare provider if they have experienced any problems that may be related to using these drug products.
Adverse reactions or quality problems experienced with the use of this product may be reported to the FDA's MedWatch Adverse Event Reporting program either online, by regular mail or by fax.
- Complete and submit the report Online
- Regular Mail or Fax: Download form or call 1- 800-332-1088 to request a reporting form, then complete and return to the address on the pre-addressed form, or submit by fax to 1-800-FDA-0178
This recall is being conducted with the knowledge of the U.S. Food and Drug Administration.
About Mylan
Mylan is a global pharmaceutical company committed to setting new standards in healthcare. Working together around the world to provide 7 billion people access to high quality medicine, we innovate to satisfy unmet needs; make reliability and service excellence a habit; do what's right, not what's easy; and impact the future through passionate global leadership. We offer a portfolio of more than 7,500 marketed products around the world, including antiretroviral therapies on which approximately 40% of people being treated for HIV/AIDS globally depend. We market our products in more than 165 countries and territories. We are one of the world's largest producers of active pharmaceutical ingredients. Every member of our approximately 35,000-strong workforce is dedicated to creating better health for a better world, one person at a time. Learn more at Mylan.com. We routinely post information that may be important to investors on our website at investor.mylan.com.
Company Contact Information
- Consumers:
- Mylan Customer Relations
- 800.796.9526
- customer.service@mylan.com
- Media:
- Christine Waller
- 724.514.1968