COMPANY ANNOUNCEMENT
Jubilant Cadista Pharmaceuticals Inc. Issues Voluntary Nationwide Recall of Drospirenone and Ethinyl Estradiol Tablets, USP, Due to Out-of-Specification Dissolution Test Results
This recall has been completed and FDA has terminated this recall.
When a company announces a recall, market withdrawal, or safety alert, the FDA posts the company's announcement as a public service. FDA does not endorse either the product or the company.
Read Announcement View Product PhotosSummary
- Company Announcement Date:
- FDA Publish Date:
- Product Type:
- Drugs
- Reason for Announcement:
-
Recall Reason DescriptionDue to out of specification (OOS) dissolution results at the 3-month stability time point
- Company Name:
- Jubilant Cadista Pharmaceuticals Inc.
- Brand Name:
-
Brand Name(s)Cadista
- Product Description:
-
Product DescriptionDrospirenone and Ethinyl Estradiol Tablets, USP, 3 mg/ 0.02mg
Company Announcement
Salisbury, Maryland, Jubilant Cadista Pharmaceuticals Inc. is voluntarily recalling one lot of Drospirenone and Ethinyl Estradiol Tablets, USP, 3 mg/ 0.02mg, 28x3 Blister Pack/Carton to the consumer level. The affected product is being recalled due to out-of-specification (OOS) dissolution results at the 3-month stability time point. The affected product is manufactured by Cyndea Pharma, S.L., Olvega (Soria), 42110 Spain under contract from Jubilant Cadista Pharmaceuticals Inc., 207 Kiley Drive Salisbury, Maryland 21801.
As a result of the OOS dissolution results, product efficacy may be decreased due to incomplete absorption of the active ingredients. To date, Jubilant Cadista Pharmaceuticals Inc. has not received any reports of adverse events related to this recall.
Product Description: Drospirenone and Ethinyl Estradiol tablets are an estrogen/progestin combination oral contraceptive, indicated for use by women to:
- Prevent pregnancy
- Treat symptoms of premenstrual dysphoric disorder (PMDD) for women who choose to use an oral contraceptive for contraception
- Treat moderate acne for women at least 14 years old and/or if the patient desires an oral contraceptive for birth control
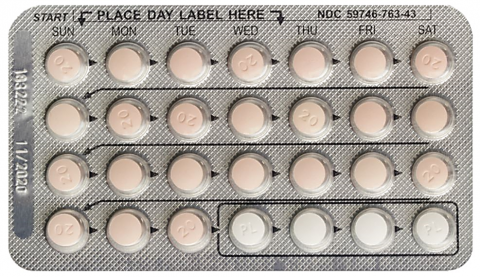
Drospirenone and Ethinyl Estradiol tablets are packaged into a carton containing 3 blister cards. Each blister card contains 28-film coated, biconvex tablets, in the following order: 24 active pink-color round, unscored, film-coated tablets debossed with a “20” on one side, each containing 3 mg Drospirenone and 0.02 mg Ethinyl Estradiol, and four (4) inert white-color round, unscored, film-coated tablets debossed with a “PL”.
The affected Drospirenone and Ethinyl Estradiol Tablets, USP, has been identified as Lot number 183222, with NDC number 59746-763-43 and expiration date of 11/2020.
Drospirenone and Ethinyl Estradiol Tablets, USP, 3 mg/0.02 mg, lot 183222 was distributed Nationwide to wholesalers, distributors, and retailers.
Jubilant Cadista Pharmaceuticals Inc. is notifying its customers by emailing a recall notification letter and response form and is arranging for return of all recalled product.
Patients that have used the affected lot of Drospirenone and Ethinyl Estradiol Tablets, USP, 3 mg/0.02 mg should consult their healthcare provider. Patients may return the affected lot to their place of purchase.
Wholesalers, distributors, and retailers should immediately examine their inventory for the affected lot. All inventory of the affected lot should be quarantined to prevent further distribution to patients. Customers who purchased the impacted product directly from Jubilant Cadista Pharmaceuticals Inc. can call Inmar at 1-855-205-9246 (9:00 a.m. – 5:00 p.m. EDT, Monday – Friday) to arrange for their return.
Consumers with additional questions regarding the recall may contact Jubilant Cadista by phone at 1-800-308-3985 (9:00 a.m. – 6:00 p.m. EDT, Monday – Friday). Consumers should contact their physician or healthcare provider if they have experienced any problems that may be related to taking or using this drug product.
Adverse reactions or quality problems experienced with the use of this product may be reported to the FDA's MedWatch Adverse Event Reporting program either online, by regular mail or by fax.
This recall is being conducted with the knowledge of the U.S. Food and Drug Administration.
About Jubilant Cadista Pharmaceuticals Inc.
Jubilant Cadista Pharmaceuticals Inc. is a rapidly growing generic pharmaceutical company in the United States devoted to discovery, manufacture, sale, and distribution of prescription generic pharmaceutical products. To learn more about Jubilant Cadista, please visit our website: www.cadista.com
- Complete and submit the report Online
- Regular Mail or Fax: Download form or call 1- 800-332-1088 to request a reporting form, then complete and return to the address on the pre-addressed form, or submit by fax to 1-800-FDA-0178
Company Contact Information
- Consumers:
- Jubilant Cadista Pharmaceuticals Inc.
- 1-855-205-9246
- Media:
- Kevin J. Fortier
- 1-800-308-3985
- kevin.fortier@cadista.com