COMPANY ANNOUNCEMENT
Hospira Issues a Voluntary Nationwide Recall for Labetalol Hydrochloride Injection, USP, Due to the Potential of Cracked Glass at the Rim Surface of the Vials
This recall has been completed and FDA has terminated this recall.
When a company announces a recall, market withdrawal, or safety alert, the FDA posts the company's announcement as a public service. FDA does not endorse either the product or the company.
Read Announcement View Product PhotosSummary
- Company Announcement Date:
- FDA Publish Date:
- Product Type:
- Drugs
- Reason for Announcement:
-
Recall Reason DescriptionDue to Potential Of Cracked Glass At The Rim Surface Of The Vials
- Company Name:
- Hospira, Inc.
- Brand Name:
-
Brand Name(s)Hospira, Novaplus
- Product Description:
-
Product DescriptionLabetalol Hydrochloride Injection, USP
Company Announcement
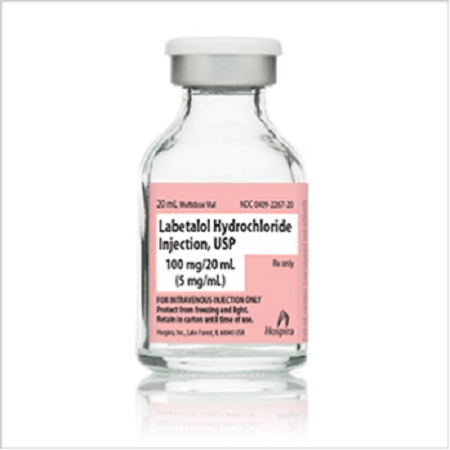
Hospira, Inc., a Pfizer company, is voluntarily recalling 3 lots of Labetalol Hydrochloride Injection, USP, 100 mg/20 mL Vial (NDC 0409-2267-20), and one lot of Labetalol Hydrochloride Injection, USP, Novaplus® (NDC 0409-2267-25) to the hospital/institution level. Hospira, Inc. initiated this recall due to the discovery of cracks on the rim surface of vials for these lots, which is covered by the stopper and crimp seal.
Cracked vials may result in a lack of sterility assurance for the product. Use of or exposure to a non-sterile product may be associated with adverse events such as fever, chills, sepsis or invasive systemic infections in patients. To date, Hospira, Inc. has not received any reports of adverse events associated with this issue for these lots.
Labetalol Hydrochloride is an adrenergic receptor blocking agent indicated for the control of blood pressure (BP) in severe hypertension. It is administered by repeated intravenous (IV) injections or by slow IV infusion.
Labetalol Hydrochloride Injection is packaged in a 20 mL Multiple Dose Fliptop Vial; packaged in 50 cartons per case. The affected lots include the following NDC, lot numbers, and expiry dates. For cartons, the NDC is located on the top and front label and Lot and Expiry Date information is located on the bottom. For individual vials, the NDC, Lot and Expiry Date information is located on the vial label.
Product was distributed nationwide to wholesalers / retailers / hospitals in the United States and Puerto Rico from April 2017 to August 2017.
|
NDC |
Lot Number |
Expiration Date |
Strength |
Configuration/Count |
|---|---|---|---|---|
|
0409-2267-20 |
74370DD |
1FEB2019 |
100 mg/20 mL |
1-20 mL Multiple Dose Fliptop Vial per carton; 50 cartons per case |
|
0409-2267-25 (Novaplus®) |
74230DD |
1FEB2019 |
100 mg/20 mL |
1-20 mL Multiple Dose Fliptop Vial per carton; 50 cartons per case |
Hospira, Inc. places the utmost emphasis on patient safety and product quality at every step in the manufacturing and supply chain process.
Hospira, Inc. has notified wholesalers/retailers/hospitals by recall letter to arrange for return of any recalled product.
Wholesalers/retailers/hospitals/institutions with an existing inventory of the lots subject to this recall should stop use and distribution of the remaining units and quarantine immediately. Healthcare Professionals in your organization should be informed of this recall. If you have further distributed the recalled product, to the wholesale or retail level, please notify any accounts or additional locations which may have received the recalled product from you. For additional assistance, call Stericycle at 1-800-805-3093 between the hours of 8 a.m. to 5 p.m. ET, Monday through Friday.
Retailers/ Healthcare Professionals with questions regarding this recall can contact Pfizer using the below information.
|
Contact |
Contact Information |
Areas of Support |
|---|---|---|
| Pfizer Medical Information | 1- 800-615-0187 , option 3 (8am to 7pm ET Monday through Friday) |
Medical inquiries |
| Pfizer Safety |
1-800-438-1985, option 1 (24 hours a day 7 days per week) |
To report adverse events or product complaints |
Adverse reactions or quality problems experienced with the use of this product may be reported to the FDA's MedWatch Adverse Event Reporting program either online, by regular mail or by fax.
- Complete and submit the report Online
- Regular Mail or Fax: Download form or call 1- 800-332-1088 to request a reporting form, then complete and return to the address on the pre-addressed form, or submit by fax to 1-800-FDA-0178
This recall is being conducted with the knowledge of the U.S. Food and Drug Administration.
Company Contact Information
- Consumers:
- 1-800-438-1985
- Media:
- 610-329-1340