COMPANY ANNOUNCEMENT
Dibar Nutricional S. de R.L. De C.V. Issues Voluntary Nationwide Expanded Recall of Dibar Labs Hand Sanitizer Due to the Presence of Methanol (Wood Alcohol)
When a company announces a recall, market withdrawal, or safety alert, the FDA posts the company's announcement as a public service. FDA does not endorse either the product or the company.
Read Announcement View Product PhotosSummary
- Company Announcement Date:
- FDA Publish Date:
- Product Type:
- Drugs
- Reason for Announcement:
-
Recall Reason DescriptionPresence of methanol
- Company Name:
- Dibar Nutricional S. de R.L. de C.V.
- Brand Name:
-
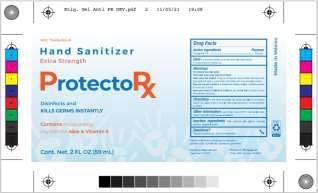
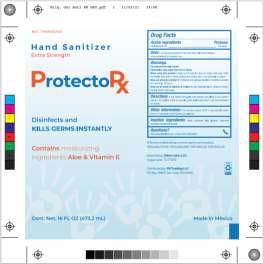
Brand Name(s)DIBAR Labs, ProtectoRx
- Product Description:
-
Product DescriptionHand Sanitizer
Company Announcement
Morelia, Michoacán, Mexico,Dibar Nutricional S. de R.L. De C.V. (“Dibar”) is voluntarily recalling all Dibar Labs Hand Sanitizer packaged in 8oz and 16 oz bottles, respectively, to the consumer level (see Label 1 below). Dibar is also voluntarily recalling ProtectoRx Hand Sanitizer packaged in 2oz and 16oz fluid ounce bottles, respectively, to the consumer level (see Label 2 below), as well as all Advance Hand Sanitizer packaged in 16 oz bottles, likewise, to the consumer level (see Label 3 below). These products are being recalled after testing conducted by our firm revealed the presence of methanol.
Risk Statement: Substantial methanol exposure can result in nausea, vomiting, headache, blurred vision, permanent blindness, seizures, coma, permanent damage to the nervous system, or death. Although all persons using these products on their hands are at risk, young children who accidentally ingest the products and adolescents and adults who drink the products as an alcohol (ethanol) substitute are most at risk for methanol poisoning. To date, the company has not received any reports of adverse reactions related to this recall.
These products are used as hand sanitizers and marketed to help decrease bacteria on the skin when soap and water are not available. The affected bottles of hand sanitizer include a twelve-digit lot code printed on the bottle near the base. The products can be identified by the label image provided at the end of this release. Products labeled as Dibar Labs Hand Sanitizer were distributed nationwide in the USA to wholesale companies who further distributed through their customers. Products labeled as ProtectoRx Hand Sanitizer were distributed in Puerto Rico to wholesale companies who further distributed through their customers. Products labeled as Advance Hand Sanitizer were distributed in the USA to wholesale companies who further distributed through their customers.
Dibar Nutricional S. de R.L. De C.V., has notified its direct distributors by letter and asked that they remove the recalled products from commerce immediately if they still possess inventory.
Consumers, distributors and retailers that have the hand sanitizers which are being recalled should stop using, distributing and/or selling them and return them to the place of purchase.
Consumers with questions regarding this recall can contact our Commercial Offices, +52 443-314-5369, M-F, during business hours 9:00 a.m. – 6:00 p.m. ET and/or email us to QA@dibarlabs.com. Consumers should contact their physician or healthcare provider if they have experienced any problems that may be related to taking or using this product.
Adverse reactions or quality problems experienced with the use of this product may be reported to the FDA's MedWatch Adverse Event Reporting program either online by regular mail or by fax.
- Complete and submit the report Online
- Regular Mail or Fax: Download form or call 1- 800-332-1088 to request a reporting form, then complete and return to the address on the pre-addressed form, or submit by fax to 1-800-FDA-0178
This recall is being conducted with the knowledge of the U.S. Food and Drug Administration.
Link to Original Recall
Company Contact Information
- Consumers:
- Commercial Offices, Barvara Palomino
- +52 443-314-5369