FDA ANNOUNCEMENT
Aurobindo Pharma USA, Inc. Initiates Voluntary Nationwide Consumer Level Recall of 38 Lots of Ranitidine Tablets 150mg, Ranitidine Capsules 150mg, Ranitidine Capsules 300mg and Ranitidine Syrup 15mg/mL Due to the Detection of NDMA (Nitrosodimethylamine) Impurity
Summary
- Company Announcement Date:
- FDA Publish Date:
- Product Type:
- Drugs
- Reason for Announcement:
-
Recall Reason DescriptionNDMA (Nitrosodimethylamine)
- Company Name:
- Aurobindo Pharma USA, Inc.
- Brand Name:
-
Brand Name(s)Aurobindo & DG Health
- Product Description:
-
Product DescriptionRanitidine
FDA Announcement
Aurobindo Pharma USA, Inc. is conducting a voluntary recall of 1 lot of Ranitidine Tablets 150mg to the retail level and 37 lots of Ranitidine Capsules 150mg, Ranitidine Capsules 300mg and Ranitidine Syrup 15mg/mL to the consumer level due to the detection of NDMA (Nitrosodimethylamine) Impurity in the finished product. The impurity detected is N-nitrosodimethylamine (NDMA), which is a substance that occurs naturally in certain foods, drinking water, air pollution, and industrial processes, and has been classified as a probable human carcinogen as per International Agency for Research on Cancer (IARC) classification. To date, Aurobindo Pharma USA, Inc. has not received any reports of adverse events related to this recall.
Ranitidine is a competitive, reversible inhibitor of the action of histamine at the histamine H2 receptors found in gastric parietal cells. This results in decreased gastric acid secretion and gastric volume, and reduced hydrogen ion concentration. Uses are:
- Relieves heartburn associated with acid indigestion and sour stomach.
- Prevents heartburn associated with acid indigestion and sour stomach brought on by eating or drinking certain foods and beverages.
Patients who prescribed or are taking Ranitidine Tablets 150mg, Ranitidine Capsules 150mg, Ranitidine Capsules 300mg and Ranitidine Syrup 15mg/mL should continue taking their medication. Patients should contact their pharmacist or physician who can advise them about an alternative treatment prior to returning their medication.
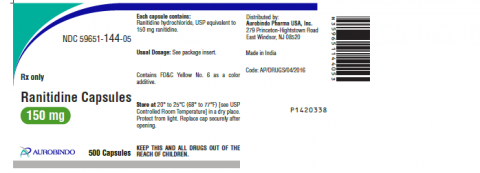
The products subject to recall are listed below and are packaged in bottles. The product can be identified by checking the product name, manufacturer details and batch or lot number on the bottle containing these products.
| Product Name | NDC | Batch Number | Exp. Date |
|---|---|---|---|
| Ranitidine Tablets 150mg | 55910-092-79 | NBSB19001DA3 | Feb-2021 |
| Ranitidine Capsules 150mg | 59651-144-60 | RA1518001-A | Jul-2020 |
| RA1518002-A | Jul-2020 | ||
| 59651-144-05 | RA1518002-B | Jul-2020 | |
| RA1518003-A | Jul-2020 | ||
| RA1518004-A | Aug-2020 | ||
| RA1518005-A | Aug-2020 | ||
| 59651-144-60 | RA1518005-B | Aug-2020 | |
| RA1518006-A | Aug-2020 | ||
| 59651-144-05 | RA1518007-A | Sep 2020 | |
| RA1518008-A | Sep 2020 | ||
| RA1518009-A | Sep 2020 | ||
| RA1518010-A | Oct 2020 | ||
| RA1518011-A | Nov 2020 | ||
| RA1518012-A | Nov 2020 | ||
| RA1518013-A | Nov 2020 | ||
| RA1518014-A | Nov 2020 | ||
| RA1518015-A | Nov 2020 | ||
| 59651-144-60 | RA1519003-A | May-2021 | |
| 59651-144-05 | RA1519003-B | May 2021 | |
| RA1519004-A | May 2021 | ||
| Ranitidine Capsules 300mg | 59651-145-30 | RA3018001-A | Jul-2020 |
| RA3018002-A | Jul-2020 | ||
| RA3018003-A | Jul-2020 | ||
| RA3018004-A | Aug-2020 | ||
| RA3018005-A | Aug-2020 | ||
| RA3018006-A | Aug-2020 | ||
| RA3018007-A | Sep-2020 | ||
| RA3018008-A | Sep-2020 | ||
| RA3018009-A | Sep-2020 | ||
| RA3018010-A | Oct-2020 | ||
| RA3019001-A | Jan 2021 | ||
| RA3019002-A | Jan 2021 | ||
| RA3019003-A | May-2021 | ||
| Ranitidine Syrup (Ranitidine Oral Solution, USP) 15 mg/mL (75 mg/5 mL) | 65862-431-74 | UI1519001-A | May-2021 |
| UI1519002-A | May-2021 | ||
| UI1519003-A | May-2021 | ||
| UI1519004-A | May-2021 |
Ranitidine Tablets 150mg, Ranitidine Capsules 150mg, Ranitidine Capsules 300mg and Ranitidine Syrup 15mg/mL were distributed nationwide to Aurobindo Pharma USA, Inc. and AuroHealth wholesale and distributor customers 28 September 2018 through 19 September 2019. Qualanex, on behalf of Aurobindo Pharma USA, Inc. will be notifying its distributors and customers by phone and in writing to immediately discontinue distribution of the specific lots being recalled and to notify their sub-accounts. Aurobindo Pharma USA, Inc. is arranging for return of all recalled products to Qualanex. Instructions for returning recalled products are given in the recall letter.
Consumers with medical questions regarding this recall or to report an adverse event can contact Aurobindo Pharma USA, Inc. at:
- 1-866-850-2876 Option 2
- pvg@aurobindousa.com
Consumers should also contact their physician or healthcare provider if they have experienced any problems that may be related to taking or using this drug product.
Any general questions regarding the return of this product please contact Qualanex at 1-888-504-2014 or email recall@qualanex.com (live calls received 7:00 am to 4:00 pm M-F CST).
Adverse reactions or quality problems experienced with the use of this product may be reported to the FDA's MedWatch Adverse Event Reporting program either online, by regular mail or by fax.
- Complete and submit the report Online
- Regular Mail or Fax: Download form or call 1- 800-332-1088 to request a reporting form, then complete and return to the address on the pre-addressed form, or submit by fax to 1-800-FDA-0178
This recall is being conducted with the knowledge of the U.S. Food and Drug Administration.