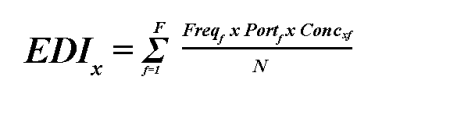GUIDANCE DOCUMENT
Guidance for Industry: Estimating Dietary Intake of Substances in Food August 2006
For questions regarding this document, please contact Hyoung Lee, 240-402-1316 at the Center for Food Safety and Applied Nutrition (CFSAN).
I. INTRODUCTION
This guidance, one in a series of Chemistry Guidance documents,[2] provides general recommendations for calculating and submitting estimates of dietary intake to support the documentation of the safety of substances introduced into food either intentionally to accomplish a technical effect, adventitiously as a component of an added substance, or inadvertently through contamination resulting from processing.
The U.S. Food and Drug Administration's (FDA's) pre-market approval processes for food and color additives require an estimate of the probable consumer intake of the additive to determine whether its use or presence in a food at a given concentration is safe. The basis for this requirement is expressed in the Federal Food, Drug and Cosmetic Act (hereinafter referred to as "the Act") as amended in 1958 (Section 409 - food additives) and 1960 (Section 721 - color additives). Furthermore, FDA also considers consumer intake of contaminants that may be present in foods. Under Section 402(a)(1)-adulterated food, a food shall be deemed to be adulterated if it bears or contains any poisonous or deleterious substance which may render it injurious to health. The intent of this document is to provide an understanding of the databases and methodologies used by the Office of Food Additive Safety (OFAS) in FDA's Center for Food Safety and Applied Nutrition (CFSAN) to estimate consumer intakes, not only of food and color additives, but of ingredients generally recognized as safe (GRAS), and other substances (e.g., chemical contaminants, including naturally-occurring toxicants) found in the diet. This document is primarily directed at petitioners for food and color additive regulations and notifiers for GRAS substances, and includes illustrative examples of calculations performed by OFAS reviewers to obtain an estimate of probable intake for substances in the diet.
The key determinant in the safety evaluation of a substance found in or added to the diet is the relation of its probable human intake to the level at which adverse effects are observed in toxicological studies.[3] Simply, "the dose makes the poison."[4] The implications of this adage as it pertains to food can be illustrated with two examples. While "pure" water can be viewed as the safest of foods, excessive intake can lead to a potentially fatal electrolyte imbalance. Conversely, pure concentrated sulfuric acid can destroy human tissue, but FDA has affirmed it as GRAS[5] for controlling pH during the processing of alcoholic beverages or cheeses. Clearly, conditions of use and dose (i.e., intake) are considered jointly when discussing the safety of a component of food.
The quality of estimated intakes for food ingredients and other food constituents depends on the quality of the intake data, either from food consumption surveys or other food-use sources (e.g., poundage data), and the substance concentration data used for calculating intake. In general, the quality of data can be highly variable, method dependent, and lacking in precision. From the point of view of food safety, estimated intakes are conservative, but reasonable, and any assumptions and novel approaches used in the calculations are transparent.
FDA's guidance documents, including this guidance, do not establish legally enforceable responsibilities. Instead, guidance describes the Agency's current thinking on a topic and should be viewed only as recommendations, unless specific regulatory or statutory requirements are cited. The use of the word should in Agency guidances means that something is suggested or recommended, but not necessarily required.
II. BACKGROUND
A. Ingredients
For purposes of this document, the term "ingredient" will be used to refer generically to substances intentionally added to food, including food additives, GRAS ingredients, color additives, and prior-sanctioned substances.[6] The procedures described for estimating intake of food ingredients are equally applicable to all of these substances.
Food additives, as defined in section 201(s) of the Act, are substances whose intended use results directly or indirectly in the substance becoming a component of food. Direct food additives are substances intentionally added to a food to accomplish an intended technical effect (21 CFR 170.3(o)). Regulations for direct food additives are listed in 21 CFR Part 172.
GRAS ingredients are substances which have been shown, through scientific procedures or history of safe use prior to 1958, to be Generally Recognized As Safe. Partial lists of GRAS ingredients may be found in 21 CFR Parts 182 and 184. CFSAN's Internet site also contains a list of substances that have been the subject of a GRAS notice submitted to FDA under the voluntary GRAS notification program. [7] Color additives are defined in Section 201(t)(1) of the Act. Regulations for color additives are listed in 21 CFR Parts 70-74 and Parts 80-82.
B. Impurities
Impurities are substances that are unavoidably or unintentionally present in food. They can be present as a result of the manufacturing process of a food ingredient (e.g., residues of the solvent dimethyl sulfoxide in sucrose fatty acid esters) or a finished food (e.g., chloropropanols in certain soy sauces), or they can arise from natural or environmental sources (e.g., aflatoxins in grains). The procedures used to estimate intakes of impurities in food are essentially the same as those used for estimating intakes of ingredients.
The practical difference between estimating intakes of impurities and ingredients is the derivation of the concentrations of these substances in foods. Impurity concentrations vary and are determined experimentally, while ingredient concentrations or use levels are proposed by a petitioner or notifier[8] for specified uses.
The different procedures used for estimating intake of ingredients and impurities will be discussed in the remainder of this document, with detailed examples in the Appendix.
III. SOURCES OF DATA FOR ESTIMATING INTAKE
A. Overview
Estimates of intake of an ingredient or chemical constituent of food require two key pieces of information: (1) the concentration of the substance in a food, and (2) consumer intake of foods that will or might contain the substance.
1. Substance Concentration Data
In dietary intake assessments, the concentration data used will depend on the nature of the specific intake assessment. The concentration of an ingredient or chemical constituent in food can be obtained from[9]
- the intended use levels of the substance in target foods (typical, recommended, or maximum use level);
- the measured concentration in food as consumed, accounting for processing and storage losses of ingredient;
- the limit of detection (LOD) or limit of quantification (LOQ) of the analytical method, as appropriate, if the concentration in the food is non-detectable or non-quantifiable at the LOD or LOQ;
- established limits for the substance (e.g., specifications in the CFR or the Food Chemical Codex (FCC)[10] for impurities and contaminants in food ingredients, or maximum levels for contaminants in foods adopted by a recognized standards-setting body, such as the Codex Alimentarius Commission).
When estimating intakes of ingredients, OFAS typically uses the maximum intended use levels proposed by a petitioner or notifier. However, in some cases, analytically-determined residues in the food may be used. For example, for a color additive used in feed for food fish, the intake of the color by consumers is estimated from the amount of color additive measured in the edible flesh of the fish.
For estimating intake of constituents in food ingredients,[11] OFAS often relies on specification limits (e.g., for appropriate heavy metals, such as lead, arsenic, and mercury) either cited directly in a regulation for the ingredient or incorporated by reference to a specification monograph published in the FCC. When limits are not specified in the FCC, OFAS may rely on documented specifications provided by the petitioner or notifier to estimate the potential intake of a constituent from the intended use of a specific ingredient.
The concentrations of constituents in foods will generally fall on a distribution curve. The values falling below an analytical LOD are typically reported as "non-detects." Concentrations of zero, one half the LOD, the LOD, the LOQ, or some other derived distribution of values have been reported as non-detects.[12] OFAS has used all of these options, selecting the most appropriate in each case on the basis of the quality and quantity of data available. To gauge the effect of the selection of a non-detect value on an intake assessment, the intake estimate may be performed twice: once using zero as the non-detect level to determine the low end of the estimate range and again using the LOD as the non-detect level to determine the high end of the estimate range. The spread in this range is useful as a guide in assessing the importance of non-detect concentration values for a given intake estimate.
In evaluating the suitability of experimentally-determined concentration data for use in an intake assessment, OFAS considers the following: analytical sampling methodology, the precision and sensitivity of the analytical method, and method validation procedures.[13] At times, a concentration of a constituent in food might have been determined on the basis of an entire organism (e.g., a plant, fish), rather than on the edible portion of the organism. Therefore, recognizing that a contaminant is generally not distributed uniformly in a living organism can be critical for an intake estimate. That is, its concentration in plant roots or animal flesh may differ significantly from that in the leaves of the plant or the skin of the animal. OFAS estimates or determines the concentration of the contaminant in the edible portion(s) of the organism as prepared for consumption, for use in its intake estimate.
2. Food Consumption Data
There are a number of sources of data available to OFAS for use in estimating intake of substances in the diet,[14] including:
- Food consumption surveys
- Food/ingredient disappearance figures
- Total Diet Study
- Body burden/excretion measurements: "Biomarkers"
Each source has advantages and limitations. For a number of reasons, including cost and availability, breadth of data, and ease of data manipulation, OFAS relies primarily on data taken from food consumption surveys. To date, OFAS has not used data from body burden/excretion measurements for estimating intake, although such data may be useful for validating intake estimates. Duplicate diet studies are another option for estimating intakes. Although OFAS has not used such data, this method was recently used by researchers in Switzerland to quantify acrylamide intake.[15] In the following sections, OFAS's use of the data from consumption surveys, disappearance data, and FDA's total diet study are outlined.
B. Food Consumption Surveys
Food consumption data may be collected at the national, household, or individual level. OFAS regularly uses food consumption surveys conducted nationwide at the individual level to estimate the dietary intake of substances. Consumption surveys at the level of the individual provide information on mean food intakes and the distribution of food intakes within sub-populations of individuals defined by demographic factors (e.g., age, sex) and health status (e.g., pregnancy, lactation). These surveys measure food intake by one or more methods: i.e., food records or diaries, 24-hour recalls, food frequency questionnaires (FFQ), and diet history.[16] In the first two methods, participants record or recall (with a trained interviewer) the amounts and types of each food eaten during the day, both at home and away from home. For the food frequency methods, participants record or recall only the number of occasions each food was consumed over a specified period of time that may vary from one day to more than one year. These eating-occasion frequencies are multiplied by an appropriate food portion size (based on age-sex considerations) to obtain semi-quantitative estimates of the daily food intake.[17]
Another food consumption survey approach is the diet history method which is designed to assess usual individual food consumption (intake and patterns) over a specified period of time. For example, the National Cancer Institute's (NCI's) Risk Factor Monitoring and Methods Branch developed a food frequency questionnaire called the "diet history questionnaire" (DHQ)[18] that consists of 124 food items and includes questions on portion size and dietary supplement use. The DHQ has been used to provide estimates of long-term nutrient intake for purposes of investigating diet-health hypotheses.
Data derived from dietary surveys usually include the amount and type of food consumed, the percentage of consumers (or "eaters") of each food type, and the intake of each food type by the "average" and the "high-level" consumer. OFAS typically uses an eaters-only intake (i.e., the total of the amounts of food consumed per day averaged over the number of days in the survey period by individuals consuming the food at least once during the survey period). In cases where the percent eaters of a food is relatively high (e.g., for a commonly-consumed food), the eaters-only intake will be comparable to the per capita (or total-sample) intake which captures both eaters and non-eaters. At 100% eaters, the eaters-only intake equals the per capita intake. The per capita (or total-sample) intake includes both eaters and non-eaters of the food (see discussion below in "Food Intake Distributions").
To estimate consumption of particular foods, OFAS has generally consulted, since the early 1990s, the USDA Continuing Survey of Food Intakes by Individuals (CSFII). OFAS has most recently used the 2-day survey data from CSFII 1994-96 and the 1998 Supplemental Children's Survey. For information about the intake of certain nutrients in the diet, OFAS has consulted nutrient intake data obtained through the National Health and Nutrition Examination Surveys (NHANES), most recently NHANES III (1988-1994).[19] The CSFII and NHANES studies, previously independent, were combined by the USDA's ARS (Agricultural Research Service) and the CDC's NCHS (National Center for Health Statistics) into a single, continuous, population-based national nutrition survey beginning in January 2002. The data on dietary intakes and nutritional status collected during the first CSFII/NHANES (2003-04) will soon be available. [Updated Information: CSFII/NHANES (2003-04) was released in September 2006.] The CSFII, NHANES, and integrated CSFII/NHANES are further described below. [20]
For many years, for most of its intake estimates, OFAS also used a commercially-contracted food intake database developed from the 1982-87 Market Research Corporation of America (MRCA) Menu Census 14-day frequency of food consumption survey. The MRCA 14-day-average intake data, although not the most current data available, are sometimes consulted by OFAS for estimating intakes of ingredients and contaminants in infrequently consumed foods or verifying estimates of intake obtained from other surveys, such as the 1994-96, 1998 CSFII, that are limited to collection of two 24-hour recalls. The MRCA survey is described in further detail below.
1. Continuing Survey of Food Intake by Individuals (CSFII)
The USDA has surveyed food use by U.S. households since 1936. In the 1960s, the surveys were expanded to include food intake by individuals. Overviews of USDA food composition surveys are available in the published literature and at the website of the Food Surveys Research Group (FSRG) of USDA/ARS.[21] Traditionally, these surveys were developed to appraise the nutritional adequacy of American diets rather than the safety of food with respect to additives or other components of food. However, the information on food intakes in the USDA surveys also is now frequently used to assess intake of additives and other food constituents.
The 1994-96 and 1998 CSFII are the most recent in a series of USDA surveys designed to measure the kinds and amounts of foods eaten by Americans. The surveys are popularly known as the "What We Eat in America" Survey.[22] Intake data from this survey has been released in two database forms: (1) food consumption expressed as food and nutrient intakes, and (2) food consumption expressed in equivalent amounts of basic food commodities.[23] The commodity data have also been released in the form of a Food Commodity Intake Database that enables assessment of intakes for specific commodities consumed as such or as a component of a food mixture. The USDA's food coding database is linked to food commodities and nutrient values to convert the survey data (reported food consumption) into relevant outputs for the respective databases.
In each of the 3 survey years of the 1994-96 CSFII, a nationally representative sample of approximately 5,000 non-institutionalized individuals residing in the U.S. provided, through in-person interviewing using a 1-day dietary intake questionnaire, food intakes on 2 non-consecutive days (3 to 10 days apart) and health-related information. Additionally, the survey was bolstered by the CSFII 1998 Supplemental Children's Survey with 2-day food and nutrient intake data for approximately 5,300 children under the age of 10. Data collection methods, instruments, and procedures were the same in all years of the survey (1994-96 and 1998), resulting in a combined survey containing 2-day food and nutrient intake data for approximately 20,000 individuals.[24]
2. National Health and Nutrition Examination Survey (NHANES)
NCHS's NHANES studies link information on food intake with respondent health information obtained by a physical examination, anthropometric measures, and laboratory analyses of various blood and urine parameters. Three NHANES studies were conducted between 1971 and 1994 (NHANES I, II, and III). Information on vitamin, mineral and dietary supplement use was collected in those surveys. The food intake data for NHANES were previously based on one 24-hour recall that was supplemented with portion size information obtained using detailed measurement guides. NHANES sampled a larger number of individuals than CSFII (approximately 30,000 respondents in NHANES III), and included information on the monthly frequency of dietary supplement use by respondents. NHANES nutrient intake data have been used to a limited extent by OFAS, but are a valuable source of information on the distribution of usual intakes of nutrients in the U.S. diet. Means and percentiles of usual intake of certain nutrients, based on food or supplement intake data from NHANES III, are compiled in summary tables in Institute of Medicine, Food and Nutrition Board reports[25] on Dietary Reference Intakes for various nutrients.
NHANES became a continuous program in 1999, with approximately 5,000 individuals surveyed each year. NCHS released data sets to the public in two-year cycles (NHANES 1999-2000, NHANES 2001-2002).[26] These dietary data are released in two files: a total nutrient intakes file and an individual food file (with detailed records of gram weights and nutrient values). Dietary supplement data are available in separate files. Since 1999, targeted food frequency questions have been included in the dietary data collected for NHANES, including consumption frequency for the 30 days preceding the reporting for foods, such as fish and shellfish (both species-specific), milk, legumes, and alcohol and for the preceding year for green leafy vegetables.
3. The Integrated CSFII/NHANES
The CSFII and NHANES surveys were combined by ARS and NCHS into a single, continuous, population-based national nutrition survey beginning in January 2002. While NHANES studies have historically included a one-day recall, beginning in 2002, a second non-consecutive 24-hour recall was added. NCI has developed a new Food Propensity Questionnaire (FPQ) that has been included in the NHANES study since 2003. The FPQ, a modified version of NCI's diet history questionnaire, is designed to enable assessment of daily lifetime (i.e., chronic) food intake by different populations by combining FFQ and 24-hour recall data. But rather than inquiring about the portion size of a specific food, the FPQ is used to measure the "propensity" to consume a specific food (i.e., the probability that a given person will consume a specific food on any given day of the year). The propensity is multiplied by the average amount of food consumed per day (on a day the food is eaten) to calculate usual intake. Results from the FPQ component of the 2003-04 CSFII/NHANES are expected to become available by the end of 2006.
4. The MRCA 14-Day Survey
Marketing research groups in the U.S., such as the MRCA, have also surveyed the food consumption patterns of individuals and households for the food industry. The MRCA's 1982-87 14-day food-frequency survey, which included over 25,000 participants, was also conducted in a manner to avoid geographical and seasonal eating biases. The survey, which relies on reporting of the number of eating occasions for a given food per day rather than the amount of food consumed, provided a means to determine 14-day-average food intakes. The survey data were recorded by a single member of each surveyed household, usually the female head. For each member of the household, the recorder submitted the age, sex, weight, and number of eating occasions for each type of food listed on the survey form, for both home and away-from-home consumption of food and drink (water consumption was not considered in this survey). MRCA linked the frequencies of eating occasions to portion size data from USDA surveys to obtain the food consumption data necessary for estimating intakes of substances in food (see discussion below in Modeling Intake Analysis).
Longer duration "food-frequency" surveys, such as those conducted by the MRCA, provide more reasonable estimates of chronic consumption of foods or of intakes of food ingredients and contaminants than do food-consumption surveys of shorter duration (2-3 days), which tend to exaggerate intake of infrequently consumed foods. The MRCA's 1982-87 14-day food-frequency survey had until recently been a primary source of food intake information for OFAS. Although dated, the MRCA data remain of value to OFAS for comparison purposes (i.e., to verify that reasonable intake estimates for individual foods are produced from the more recent CSFII two-day food-consumption data). Survey data over periods as long as 14 days are not commonly available throughout the rest of the world, although 7-day surveys have been conducted in some European countries.[27] The World Health Organization (WHO) has emphasized the need for such longer term data.[28]
C. Food/Ingredient Disappearance Data
In the U.S., information on the poundage of many food commodities entering commerce is available annually from the Economic Research Service (ERS) of the USDA.[29] An online database of poundage-based per capita food consumption is also available from the ERS,[30] which permits customized searches of commodities (e.g., meat) and sub-commodities (e.g., red meat, poultry, fishery products) by year. Annual amounts of major U.S. commodities entering into the US food supply are measured by subtracting data on exports, year-end inventory, and non-food use from data on production, imports, and beginning inventories. These data are sometimes referred to as "disappearance" data because they represent the disappearance of food into the marketing system. Annual disappearance figures for a food commodity can be divided by the national population and by 365 days to obtain a "per capita" estimate of the food that is available for consumption per day expressed as grams per person per day (g/p/d). Disappearance data cannot be used to estimate intake for targeted sub-populations (e.g., young children, diabetics, or specific age-sex groups).
The food industry also measures the disappearance of specific products, including ingredients, into the market. These data are usually gathered to observe marketing trends and lack the specificity needed for use by regulatory bodies. However, when the data reflect disappearance of all of an ingredient being monitored, a crude estimate of per capita intake of the ingredient can be made.
Food disappearance data may overestimate actual per capita consumption because they include spoilage and waste accumulated in the marketing system and in the home, and food that is not available for human consumption, such as turkey parts used in pet foods.[31] For example, in 1987-88, the estimate of food energy available for consumption per capita per day in the U.S. was about twice the estimate of mean food energy intake by the U.S. population based on reported food intake in a national food consumption survey.[32]
Poundage data for the production of food ingredients have also been collected. The National Academy of Sciences (NAS) has, on several occasions (the most recent being 1987), conducted annual surveys of the food industry to acquire such data. These data were used to calculate the per capita intake for each substance. Because these NAS surveys enjoyed only a 60% response, the poundage data were divided by a factor of 0.6 to account for the underreporting.[33]
Because of the limitations inherent in disappearance data and poundage data, OFAS restricts its use of these types of data to comparisons, i.e., for intakes obtained using data from different sources (primarily to check the reasonableness of an estimate), and, when no other data on a substance are available to estimate per capita intake, if one can infer that the substance is widely distributed in the food supply.
D. Total Diet Study
FDA's Total Diet Study (TDS; also called the market basket study) is an ongoing program through which the Agency monitors and analyzes a wide range of foods to determine levels of selected substances (e.g., nutrients, pesticide residues, and industrial contaminants). Estimates of dietary intake of those substances can be obtained by combining the analytical results with amounts of food consumed reported in national food consumption surveys.[34]
The TDS has been conducted continuously in the U.S. since the early 1960s. Although the program has been revised many times over the years, it currently uses four regional sample collections (or market baskets) each year. For each market basket, three samples of approximately 280 different foods are collected at retail food stores and fast-food restaurants. The foods sampled in the TDS are representative of those commonly consumed in the U.S. as reported in national food consumption surveys conducted by USDA or DHHS (as described in the previous sections). FDA updates the list of foods to be surveyed about every 10 years to reflect changes in food consumption patterns.
Following sample collection, FDA prepares the foods as they would be consumed (i.e., table-ready), composites the three samples per food item, and analyzes each of the 280 food composites for about 300 different contaminants and nutrients. The analytical results provide a rich source of data on levels of contaminants and nutrients in foods for two reasons: (1) the TDS food list is comprehensive, including a broad range of foods and beverages that comprise the typical American diet; and (2) because the foods are prepared table-ready prior to analysis, the analytical results reflect the effects of preparation and cooking on the analyte concentrations. On the other hand, the number of composite samples of a given food analyzed each year has been restricted to four, which limits the usefulness of the TDS in evaluating the distribution of the substance in foods and in the diet. Furthermore, the TDS food list can exclude foods that are not widely consumed, but which could be significant sources of particular contaminants or nutrients (e.g., some seasonal fruits or particular species of fish or shellfish).
Dietary intakes of substances measured in the TDS can be estimated by combining TDS analytical results and food consumption data. There are two main approaches to calculating intakes using TDS data, depending on whether detailed data from national food consumption surveys or the TDS diets are used as the food consumption database. The first approach is identical to the method used by OFAS for deriving intake estimates for which concentration data (in this case, analytical results for one or more of the TDS foods) are linked to consumption data for a population subgroup (e.g., pregnant women) and calculated on a specific consumption basis (e.g., eaters-only). In the second approach, the TDS analytical results are linked to a static set of per capita consumption amounts derived for each TDS food (referred to as the TDS diets). The TDS diets are compiled from the same national food consumption data used by OFAS (i.e., from USDA or DHHS), but they represent the total per capita consumption of all of the approximately 6,000 foods reported in the survey (hence the name "total diet study"). To derive the TDS diets, per capita consumption is first calculated for each survey food and each food is then assigned to the TDS food to which it is most similar (e.g., cooked apple products reported in the survey, such as applesauce, stewed apples, and cooked apples, are assigned to the TDS food "applesauce"). Finally, the per capita consumption for all survey foods assigned to a given TDS food are summed to obtain the TDS consumption amount (e.g., the sum of per capita consumption of the cooked apple products equals the consumption amount for the TDS food "applesauce"). Diets are compiled in this way for the total U.S. population as well as for 14 age-sex subgroups. Like the TDS food list, the TDS diets are updated about every 10 years.[35]
The TDS intake model is useful for providing estimates of average background intakes of TDS analytes owing to the breadth of foods analyzed, analysis of table-ready foods, and the fact that the TDS diets account for consumption of all foods in the diet. On the other hand, TDS diets do not allow estimation of eaters-only acute or upper percentile intakes, although the analytical results from the TDS can be combined with other food consumption data to derive such estimates.
Total diet studies are conducted by many other countries, and data from these studies are very useful in comparing dietary intakes worldwide. The WHO has co-sponsored several regional and international total diet study workshops to promote undertaking of reliable and comparable studies worldwide. WHO also maintains a database of total diet study results from many countries through its Global Environmental Monitoring System/Food Contamination Monitoring and Assessment (GEMS/Food) Programme.[36]
E. Biomarkers
Although not typically used by OFAS, biomarkers can be of value in the assessment of exposure to nutrients, food contaminants, and other food constituents.[37] The biomarker methodology requires demonstration of a quantitative relationship between intake of a substance and the amount of the substance or a metabolite in the body tissue or fluid of humans, e.g., in blood, adipose tissue, urine, or breast milk. The methodology also requires an understanding of dietary and endogenous factors that might influence the concentration (i.e., uptake, clearance, excretion) of the biomarker in different body tissues. At the present time, biomarker methodology has limited application in the assessment of human exposure to constituents of foods. The use of biomarkers for estimating intake has been investigated for methyl mercury,[38] fumonisins,[39] phthalate diesters, [40] and several nutrients (e.g., fat, protein, minerals, and vitamins).[41] The use of biomarkers in exposure assessment methods is the subject of ongoing research.[42]
IV. MODELING INTAKE ANALYSIS
A. Background
Two factors are required for estimating intake of a food substance. The first is the daily intake of the foods in which the substance is used or can be found. The second is the concentration or use level of the substance in each food. Simple examples of intake estimates are provided under "Single Use Additives," in the Appendix. In the early 1970s, data on food intake by individuals were available from only two national food consumption surveys: the 1965 USDA Nationwide Food Consumption Survey (NFCS) and the MRCA Menu Census survey. In conjunction with the FDA, the NAS developed a model for estimating intake that combined the MRCA data with the USDA/NFCS information. The MRCA data provided information on the frequency of consumption of foods over a 14-day period; the USDA/NFCS data provided portion size information. Intakes derived from multiple-day surveys of individuals were considered more representative of long-term or chronic intake than single-day survey-derived estimates. The relationship of food-consumption frequency, portion size, and substance concentration data to the estimated daily intake (EDI) of a substance x for a single individual is captured in the following equation.
where:
F = Total number of foods in which substance "x" can be found
Freqf = Number of eating occasions of food "f" over "N" survey days
Portf = Average portion size for food "f"
Concxf = Concentration of the substance "x" in food "f"
N = Number of survey days
B. Food Intake of Sub-populations
OFAS typically estimates intake of substances that may be consumed by the general population ("all ages," aged 2+ years), children aged 2-5 years, and, if needed, infants and children less than 2 years of age. Estimates of intakes of substances by sub-populations identified as target populations are performed as required. For example, if OFAS were to assess the safety of a food containing a substance that showed toxic effects in pregnant animals, the food intake for women of child-bearing age would be used in the analysis.
An intake estimate of a food ingredient or contaminant at a given percentile, based on consumption of a single food, reflects the consumption of that food by a sub-population, the "eaters-only" sub-population, of the total-sample population. Therefore, these percentile intakes based on individual foods cannot be summed to obtain an eaters-only intake of the ingredient or contaminant from all of the foods considered. This topic is further illustrated in the section entitled "Statistical Approaches to Data Analysis".
C. Types of Food Intake Estimates
1. Chronic Intake
For the evaluation of the safety of an ingredient or the assessment of risk associated with long-term intake of a contaminant, OFAS evaluates data concerned with lifetime (chronic) intake of the substance. For representing the probable consumption pattern of a population during an extended period of time, OFAS uses food consumption data from a multi-day survey. In addition, for the estimate of chronic intake, the concentration values for the substance in food should reflect the levels likely to be found over time. A regulatory upper-use level can be used for an ingredient expected to be consumed at a relatively constant level over time. Also, OFAS might use an upper-use level rather than a typical or recommended level to avoid the possibility of underestimating the intake by individuals who frequently consume the product containing the ingredient or who prefer a brand of food formulated with the maximum permitted level of the ingredient.[43] On the other hand, for a persistent and ubiquitous contaminant, such as lead or other heavy metal, OFAS would use an analytically-determined mean concentration, including non-detects, to reflect chronic intake.
As the various food-consumption databases provide only snapshots of food-consumption for limited periods of time, intake estimates based on these databases are generally conservative as measures of average daily chronic intakes or average daily intakes over a lifetime for individuals within the surveyed population.
2. Acute Intake
OFAS is also asked to address very short-term or even single occasion intakes, especially for contaminants associated with acute toxic effects. To estimate contaminant intake in these cases, food consumption from single eating occasions or from one-day food intakes would be used. Using the concentration of the contaminant from the high end of the distribution of measured levels ensures that the intake estimate associated with the risk assessment would be conservative for a "high level" consumer. When food consumption data are not available, or are thought to be underreported in the surveys (e.g., for alcoholic beverages), eating scenarios may be developed (see "Special Cases-Dietary Scenarios," below).
D. Statistical Approaches to Data Analysis
The 1992 Environmental Protection Agency (EPA) guidelines for exposure assessment concisely state: "Exposure assessments are done for a variety of purposes, and for that reason, cannot be easily regimented into a set format or protocol. Each assessment, however, uses a similar set of planning questions, and by addressing these questions the assessor will be better able to decide what is needed to perform the assessment and how to obtain and use the information required."[44] After defining the nature of the risk associated with the substance under consideration, the sub-populations at risk, and the food in which the substance may be found, information on the intakes of the identified foods and the concentration of the substance in each of those foods is compiled.
The availability of food consumption data reported by each participant in a multi-day survey makes it possible to combine these data with concentration data and estimate the intake of a substance by each participant. A distribution of the intakes of all individuals in the target sub-population can then be constructed and used in the risk assessment. Using software[45] for accessing the primary food consumption data, OFAS can obtain intake estimates that reflect probable intake.
When only summary food consumption survey data are available[46], assumptions are made for combining intakes from the consumption of the individual foods (or food groupings) containing the substance. An example of food-intake summary data is presented in Table 1.
Table 1
| Percent eaters | Food Intake (Eaters-only) | ||
| mean (g/p/d) | 90th percentile (g/p/d) | ||
| Food A | 65 | 25 | 45 |
| Food B | 23 | 15 | 35 |
Table 2
If the concentration of substance "X" is 10 µg/g in food A and 25 µg/g in food B, the intake of "X" from the consumption of each food is that shown in Table 2 below.
| Concentration (µg/g) | Intake of "X" (Eaters-only) | ||
|---|---|---|---|
| mean (µg/p/d) | 90th percentile (µg/p/d) | ||
| Food A | 10 | 250 | 450 |
| Food B | 25 | 375 | 875 |
The mean and 90th percentile intakes of "X" from eaters of both foods A and B cannot be directly obtained from intakes in Table 2. The eaters-only intake from consumption of food A cannot be added to that from food B in Table 2 because the 65% of the population that eats food A is not identical to the 23% of the population that eats food B. To sum individual intakes, the intakes are expressed in terms of the total sample. Total-sample mean intakes can be summed. In the above example, the total-sample mean intake of "X" (provided it is found only in foods A and B) is 249 µg/p/d (65% of 250 plus 23% of 375).
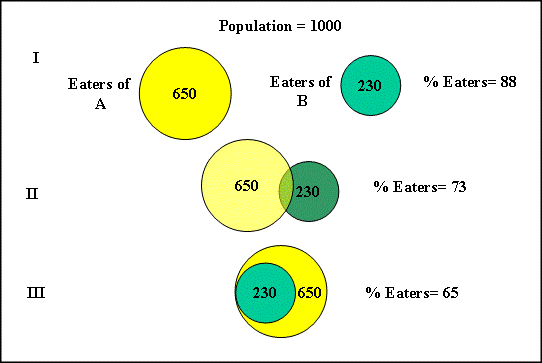
To convert the total-sample mean intake to an eaters-only intake, the percentage of the population that consumes either foods A or B (or both) is determined. The percentage of eaters of A or B (or both) lies between 65 and 88%, as shown in Figure 1.
Figure 1. Effect of Overlap of Eater Sub-Populations on Total Eater Population
I) No overlap - Largest eater population
II) Partial (statistical) overlap
III) Total overlap - Smallest eater population
In case (I), the populations of eaters of food A and of food B are different (i.e., the populations are independent). Hence, the percentage of the total population that eats either food A or B is 88% (65% + 23%). This scenario represents the eaters of "either-or" type foods (e.g., coffee or tea, soda or diet soda, etc.). In case (II), a consumer's choice of one food (e.g., lasagna) is unrelated to the choice of another food (e.g., chocolate ice cream). The overlap of the two populations is a statistical sampling equal to the product of the two percentages of eaters (65% x 23% = 15%).[47] Hence, the percentage of the total population that eats A or B (or both) is 73% (i.e., the theoretical maximum percentage of eaters (case (I)) minus the eaters of foods A and B, or 88% - 15% = 73%). In case (III), a consumer who has chosen to eat food B will also eat food A so that the percentage of eaters of A or B (or both) is equal to the percentage of eaters of A. For example, dieters might use packets of artificial sweetener (food B) and drink diet soft drinks (food A). The percentage of eaters of A or B in case (III) would be 65%.[48]
The size of the consuming population is calculated on a case-by-case basis. Case (I) is less likely to occur when many foods contain the substance of interest because almost everyone will consume one or another of the foods containing the ingredient or contaminant. The most conservative estimates of intake are made by assuming the lowest possible eating population (case (III)). In most cases, assumption of a statistical blend (case (II)) should provide a reasonable estimate of the size of the consuming population and hence a reasonable estimate of intake (see "Multiple-Use Additives-Probable Intake for an Emulsifier," below).
E. Upper Percentile Estimates
OFAS estimates upper percentile intakes of substances in the diet to account for individuals who are considered "high level" consumers of specific foods that contain these substances. The 90th, 95th and 97.5th percentile intakes are used by various regulatory bodies in the world to represent "high level" consumers. OFAS typically uses the 90th percentile intake estimates (based on 2- or 3-day survey data) to represent long-term or "lifetime averaged" daily intake estimates, in large part, because of the conservatisms built into the intake estimates. Information on the distribution of food intakes for the fraction of the population that can be considered "high level" consumers of the foods of interest provides a basis for estimating the intakes of components of the foods. Approaches for estimating upper percentile dietary intake from summary consumption data for individual foods or food groupings are discussed in this section.
Examination of food frequency and other food-consumption surveys conducted in the U.S. shows that consumption at the 90th percentile for most commonly-consumed foods is approximately 2 times the mean consumption for that food,[49] and intake at the 95th percentile is approximately 4 times the mean. Thus, a crude approximation of intake of a substance at the 90th percentile can be obtained by doubling the calculated mean intake and at the 95th percentile by quadrupling the mean. OFAS sometimes refers to the intake at the 90th percentile obtained in this manner as the "pseudo-90th percentile" intake, in order to distinguish it from an intake for consumers of the food at the 90th percentile estimated from actual food intake distribution data.
F. Probabilistic Modeling
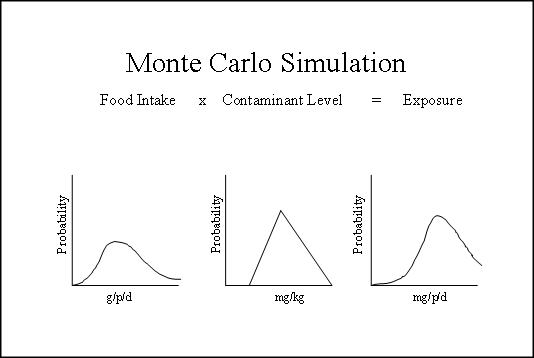
Computer-based Monte Carlo simulations for probabilistic modeling are used by OFAS to calculate percentile intakes for substances.[50] These simulations generate results for models in which one or more inputs can be defined by a distribution of values (here, food intakes and substance concentrations). Rather than using a single value for such an input (e.g., a point estimate such as mean or 90th percentile food intake), the simulation selects a value at random from the distribution of possible values for that input, uses that value to calculate an outcome for the model, stores the result, and then repeats the procedure a predetermined number of times (or iterations). For each iteration, all data inputs, defined as probabilistic expressions, are randomly sampled such that each iteration is likely to produce a different outcome. Once a specified number of iterations has been completed, the set of results is collected and statistical measures (e.g., mean, standard deviation) are calculated.
Figure 2 shows the distribution of inputs for a typical Monte Carlo Simulation.
Figure 2. Monte Carlo Simulation
The output from the Monte Carlo simulation is a range of possible outcomes from which a probability distribution function is prepared. An intake at a designated percentile can be obtained directly from the distribution function.[51] Further, from this distribution of intakes, the full range of intakes (i.e., outcomes) can be viewed, the probability of occurrence for any selected level of intake can be read, and the proportion of the population above the selected level can be determined. In effect, the extent to which intake is likely exceed the selected level can be estimated. For an example of the use of this method, see "Multiple-use Additives-Probable Intake for an Emulsifier".
The strength of the Monte Carlo modeling technique lies in its ability to yield a more realistic estimation of intake of a substance for given percentiles of a population of eaters, when more than one food source contributes to the intake, than does summing individual food intakes from summary data.[52] When modeling intake with a Monte Carlo simulation, food-consumption data, ingredient use levels, or contaminant concentrations, and eater/non-eater variables can be accessed as distributions (or "Yes/No toggles" in the case of eater/non-eater), using the complete range of available information.
Consumers' food choices can be considered, when performing Monte Carlo simulations, through the use of correlation functions. As an example, the scenario in which all peanut butter eaters are assumed to be jelly eaters (a reasonable assumption for the sub-population of young American children) can be tested. More general guidelines for the Monte Carlo computational systems for probabilistic modeling of dietary intake of food chemicals and nutrients are available.[53]
V. APPENDIX
The following examples demonstrate how OFAS reviewers determine the EDI for: (1) food ingredients intentionally added to food based on uses proposed by a petitioner or notifier; (2) for currently-regulated uses of these food ingredients; and (3) for other food constituents, based on experimentally-determined concentrations in food. The examples were selected to illustrate the diverse nature of the issues addressed by OFAS. First, two simple examples (single-use ingredients) are used to highlight inherent conservatisms in OFAS's estimates resulting from: (1) the assumptions necessary for pre-market assessment of the ingredient (e.g., broadest possible food groups and complete replacement of other technologically-equivalent additives by the new ingredient); and (2) the use of a maximum proposed concentration of an ingredient in food. When the use of such conservatisms leads to an EDI above a toxicologically established acceptable daily intake (ADI), a petitioner or notifier may submit additional data to allow a refinement that could lower the estimate. The responsibility for providing data for reevaluation remains with the sponsor of the ingredient. Second, examples are given of more complex cases (e.g., multiple-use ingredients), where the ingredient may be used in multiple food groups and additional assumptions are necessary to arrive at a reasonable estimate of probable intake. Third, examples of intake estimates pertaining to a processing aid and to contaminant problems are presented. Finally, two special cases requiring information not readily available or a novel approach are discussed.
A. Single-Use Additives
Example 1. Intake of a Volatile Antimicrobial
A volatile anti-microbial (VAM) agent is intended for general use in baked goods. Submitted data demonstrated that the VAM content of baked goods after storage with VAM was between 0.4 and 1.5%. However, a regulatory limit of 2% was requested. The use of this proposed limit to estimate intake of the VAM would be conservative, because the submitted data showed that 1.5% was the likely maximum residual level of the VAM. Therefore, the technical limit of 1.5% was considered sufficiently conservative to estimate the EDI of the VAM.
The baked goods food category (21 CFR 170.3(n)(1)) is quite large, encompassing all breads, cakes and pies, crackers, pancakes, ice cream cones, and more. The daily intake for an individual in the general population (the all-ages (aged 2+ years) group) consuming baked goods at the mean is approximately 130 grams/person day (g/p/d). The consumption of baked goods at the 90th percentile is approximately 260 g/p/d.
OFAS cannot predict the percentage of baked goods that will be treated with the VAM. Assuming that all baked goods receive treatment, an extremely conservative assumption on both technical and market grounds, and that the VAM is absorbed into baked goods at a level of 1.5%, the mean EDI for the VAM is 2.0 g/p/d (i.e., (0.015) x (130 g/p/d)).
This EDI is highly conservative because: (1) all baked goods (including home-prepared products) were assumed to have been treated with the VAM, (2) these products contain the VAM at the maximum residual level, and (3) the consumer is exposed to the VAM through consumption of the VAM-treated products daily over a lifetime. In reality, not all baked goods will be treated with the VAM, so only consumers seeking treated products will be exposed to the VAM at levels comparable to that calculated - an unlikely scenario. Moreover, permitting application of an antimicrobial that would lead to intakes of two grams per person per day would be a highly improbable expectation. Refinements to the estimate could be made if the types of baked goods treated with the VAM were restricted.
Example 2. Estimated Daily Intake for a Flavor Enhancer in Sweet Pickles
a. Estimate Based on Disappearance Data
A flavor enhancer is intended to be used in sweetened pickles, through its addition to pickling spice, at a level no greater than 1% (weight basis). Additionally, the pickling spice is used in the pickle brine at a level from 0.025% to 0.05%. Data from a trade association demonstrated that 4 pounds of brine will pickle 6 pounds of vegetables. The trade association also estimated that 50% of pickles are sweetened and that Americans consume 9 pounds of pickles per capita per year. Therefore, the annual per capita disappearance of sweetened pickles is 4.5 pounds or 5.6 g/p/d.[54]
Assuming that the flavor enhancer is present at 1% in the pickling spice and that the brine contains 0.05% of the spice, the per capita intake of the flavor enhancer is 19 µg/p/d:
(5.6 g sweetened pickles/p/d) x (4 lbs brine/6 lbs pickles) x (0.0005 spice/brine) x (0.01 flavor enhancer /spice) = 1.9 x 10-5 g/p/d or 19 µg/p/d
b. Estimate Based on Food Consumption Data
The eaters-only mean intake of pickles (and related products) is 7 g/p/d based on a food consumption survey. The EDI of the flavor enhancer for eaters of sweetened pickles is 24 µg/p/d:
(7 g pickles/p/d) x (4 lbs brine/6 lbs pickles) = 4.7 g brine/p/d
Brine contains 0.05% spice: 4.7 g/p/d x 0.0005 = 2.4 x 10-3 g spice/p/d
Flavor enhancer is 1% of spice: 2.4 x 10-3 g/p/d x 0.01 = 2.4 x 10-5 g/p/d or 24 µg/p/d
This EDI for the flavor enhancer is conservatively high because (1) all sweetened pickles are assumed to be pickled in brine containing flavor enhancer-treated spice, (2) all of the pickling spice is assumed to be transferred to the pickles, and (3) the consumer is exposed to the flavor enhancer from pickles daily over a lifetime. The magnitude of the overestimate of the EDI from these conservative assumptions cannot be evaluated from the available information. However, the EDI could be refined if the concentration of the spice in the pickles (as consumed) was available.
The intake of the flavor enhancer obtained from the food consumption data agrees well with that obtained using the disappearance data. Per capita estimates from disappearance data are generally lower than eaters-only mean intakes based on food consumption data, because the per capita estimates include non-eaters, as well as eaters.
B. Multiple-Use Additives
Example 3. Probable Intake of an Emulsifier
An additive already regulated for use as an emulsifier in baked goods, baking mixes, dairy product analogs, frozen dairy desserts and mixes, and whipped milk products; as a texturizer in biscuit mixes; and as a coating component in certain fruits at levels limited only by good manufacturing practices is intended for use as an emulsifier in "imitation" seafood.
To estimate the cumulative intake of the additive from its regulated and intended uses, the assessment conservatively assumes that the emulsifier: (1) is used at the maximum use level for all regulated food groups and for "imitation" seafood, and (2) would completely replace all other technologically-equivalent additives.
Intake of the emulsifier from the regulated uses was calculated using food consumption data for the total sample, rather than for the eaters-only population, because of the high probability that the entire population would consume some foods containing the emulsifier. Given the likelihood that the number of eaters of at least one of the specified foods would be close to 100%, the choice to use total sample is reasonable. In developing the new intake estimate, the food sub-category "crustacean shellfish" was selected because "imitation" seafood is intended to replace crab and lobster meat. The mean food intakes and estimation of the chronic intake of the emulsifier are shown in Table 3 below. Note that the intakes for each food category can be summed because they were based on total-sample data.
According to the table, the additional use in "imitation" seafood contributes 3.3 mg/p/d to the cumulative mean EDI of the emulsifier, which is less than a 1% increase in the EDI. As discussed earlier, by assuming the intake of a substance at the 90th percentile is approximately twice the mean[49], the intake at the 90th percentile would be approximately 800 mg/p/d.
Table 3. Estimated Daily Intake of Emulsifier (2+ years, mean intake, total-sample basis).
| Food Category (use level) | Food Subcategory | Food Intake (g/p/d) | Emulsifier Intake (mg/p/d) |
|---|---|---|---|
| Fish, Seafood (0.3%) | Shell fish (crustacean) | 1.1 | 3.3* |
| Milk products (0.5%) | Chocolate Milk | 5.6 | 28 |
| Flavored Milk | 0.04 | 0.2 | |
| Eggnog | 0.4 | 2.0 | |
| Sour Cream | 1.1 | 5.5 | |
| Creams | 1.4 | 7.0 | |
| Diet Milk Products | 1.9 | 9.5 | |
| Cheese (0.5%) | Processed Cheese | 6.6 | 33 |
| Cottage Cheese | 4.3 | 22 | |
| Cream Cheese | 0.8 | 4.0 | |
| Diet Cheese Products | 0.4 | 2.0 | |
| Other Cheese products | 1.7 | 8.5 | |
| Baked Goods (0.5%) | Breads, Rolls | 39 | 200 |
| Cakes | 6.4 | 32 | |
| Ice Cream, Milk (0.5%) | Ice Cream, Milk | 6.6 | 3.3 |
| Dairy Products, Analogues (0.5%) | Toppings | 0.35 | 1.8 |
| Whiteners | 0.64 | 3.2 | |
| Total (simple sum) | 395 | ||
| * Sample calculation: (1.1 g/p/d) x (0.003) = 3.3 x 10-3 g/p/d = 3.3 mg/p/d | |||
Further evaluation of the intake estimate at the 90th percentile for the emulsifier was carried out using a Monte Carlo analysis. Lognormal distributions of the intakes of each of the foods were combined with a triangular distribution[55] of use levels to obtain distributions of incremental exposures to the emulsifier from each food. These intake distributions were then combined to derive the distribution of overall intake of the emulsifier. The intake at the 90th percentile, 650 mg/p/d, calculated using this approach is more realistic than that obtained using the point estimate approach, as it is based on a distribution analysis that more closely reflects actual food consumption.
Continuing further, a per capita estimate of the intake of the emulsifier from the proposed use in "imitation" seafood was calculated from poundage disappearance data. Market data (early 1990s) for "imitation" seafood products available commercially indicated: 150 million pounds of seafood products are produced per year and 55% of all seafood products are "imitation" seafood. Assuming "imitation" seafood contains the emulsifier at a level of 0.3% and the U.S. population is 249 million (1990), the per capita intake of the emulsifier is 1.3 mg/p/d:
[(150 x 106 lbs seafood/yr) x (1 yr/365 d) x (454 g/lb) x (0.55 "imitation" seafood/all seafood) x (0.3 g emulsifier/100 g "imitation" seafood)]/ (249 x 106 people) = 1.3 mg/p/d
This per capita value appears to be in agreement with the estimated mean intake of 3.3 mg/p/d obtained using consumption of "imitation" seafood from the food consumption survey. Typically, per capita poundage data would underestimate intake relative to intake based on food consumption survey data.
Example 4. Probable Intake of a Color Additive (Canthaxanthin)
Canthaxanthin is approved for use (21 CFR 73.75) at a level of 30 mg/lb (67 mg/kg) in solid or semi-solid food and 30 mg/pint (64 mg/kg) in liquid food, except where prohibited by food standards of identity.
To assist in the development of the Codex General Standard for Food Additives (GSFA),[56] the U.S. contributed intake estimates for canthaxanthin from its use in foods in the U.S., using disappearance data and food consumption data, as described below.
a. Per Capita Estimate
Poundage (or "disappearance") data on food uses of canthaxanthin reported by the U.S. industry to the NAS in 1987[33] and the following related assumptions were used:
- i) 1,050 lb of canthaxanthin were reported to be used in foods
- ii) Response rate to the NAS survey is typically 60%
- iii) U.S. population is 244 million (1987)
The per capita intake of canthaxanthin is calculated as follows:
| [[ | ( | 1,050 lb | )x( | 0.454kg | )x( | 1000 g | )x( | 1 yr | ) | ]/0.6]/244 x 106 p = 0.0088 mg/p/d |
| yr | lb | kg | 365 d |
Multiplication by 2 gives 0.018 mg/p/d as the per capita intake at the 90th percentile.[49]
b. Estimate Based on Food Consumption Data
The dietary intake of canthaxanthin from its use in foods in the U.S. was calculated from U.S. food consumption data corresponding to those GSFA food categories (bouillons; soups; seafood soup mixes; seafood sauces and bases; and pasta-based main or side dishes) identified by the U.S. food industry and the maximum acceptable use levels for those food categories (see Table 4, below). The estimated eaters-only intake of canthaxanthin from its use in each of the five food categories is shown in Table 4, below.
Table 4. GSFA food categories and use levels for canthaxanthin reported in the U.S. and canthaxanthin intake estimates using the combined USDA 3-day 1989-1992 CSFII food consumption database.
| GSFA Food Categories | Max. Use level (mg/kg) | % eaters | Total-sample canthaxanthin Intake (mg/p/d)** | Eaters-only canthaxanthin Intake (mg/p/d) | ||
|---|---|---|---|---|---|---|
| Mean | 90th Pctle | Mean | 90th Pctle | |||
| Bouillons | 0.022 | 1.2 | 0.018 x 10-3 | NA*** | 1.5 x 10-3 | 3.8 x 10-3 |
| Soups (excluding bouillon & seafood soups) | 0.047 | 25.8 | 1.8 x 10-3 | 7.0 x 10-3 | 7.0 x 10-3 | 1.3 x 10-3 |
| Seafood soup mixes | 15 | 1.87 | 0.034 | NA*** | 1.8 | 2.8 |
| Seafood sauces & bases | 15 | 0.38 | 1.2 x 10-3 | NA*** | 0.32 | 0.48 |
| Pasta-based main or side dishes | 100 or 0.068 | 32.1 | 0.9 | 0.02 | 2.8 | 9.4 |
** Total-sample mean intake = (Eaters-only mean intake) x (% eaters)
***The intake at the 90th percentile could not be calculated due to the low percentage of eaters
The following steps describe the method for calculating the cumulative eaters-only intake of canthaxanthin based on the data presented in Table 4:
Step 1: Obtain the total-sample mean intake (TSMI) of canthaxanthin for each food category by multiplying the TSMI of the food by the use level for canthaxanthin in the corresponding food category.
Step 2: Obtain the cumulative TSMI of canthaxanthin by summing the TSMIs for canthaxanthin in each food category.
Step 3: Calculate the percent eaters of any or all the foods in which canthaxanthin is used:
% Eaters = {1 - [(1 - eatersfood 1) x (1 - eatersfood 2) x . . . x (1 - eatersfood n)]} x 100
where "eatersfood n" is the fraction of the population that consumes a given food; and "(1 - eatersfood n)" is the fraction of "non-eaters" for that food. Subtraction of the fraction of "non-eaters" of the foods from unity gives the fraction of eaters of any or all of the foods. This relationship assumes no correlation between the consumption of any or all the foods in which the additive is used.
Step 4: Obtain the cumulative eaters-only mean intake of canthaxanthin by dividing the cumulative TSMI for canthaxanthin (Step 2) by the percent eaters (Step 3).
Step 5: Approximate the cumulative eaters-only intake of canthaxanthin at the 90th percentile by multiplying the cumulative eaters-only mean intake by two.
As the percent eaters approaches 100%, the cumulative eaters-only intake will approach the cumulative total-sample intake. The cumulative intake of canthaxanthin from its multiple uses in foods and the per capita intake are summarized in Table 5.
Table 5. Summary of cumulative intake of canthaxanthin from uses in the U.S.: per capita and food consumption database estimates.
| Per Capita Estimate (mg/p/d) | Food Consumption Database Estimate | |||||
|---|---|---|---|---|---|---|
| Total-Sample Intake (mg/p/d) | Eaters-Only Intake (mg/p/d) | |||||
| Mean | Pseudo-90th Pctle | Mean | 90th Pctle | % Eaters | Mean | 90th Pctle |
| 0.0088 | 0.018 | 0.94 | 0.58 **** | 51.4 | 1.8 | 6.3 |
**** See text for explanation of the mean being higher than the 90th percentile value.
Several important issues with regard to the intake estimates based on food consumption data are noteworthy.
First, the data in Table 4 indicate that only two food categories in which the use of canthaxanthin is reported are statistically robust. These are the categories with a relatively high percent of eaters: soups (excluding bouillon and seafood soups), 25.8% eaters; and pasta-based main or side dishes, 32.1% eaters. The low percent eaters (approximately 1%) for the remaining food categories makes them statistically weak. However, a food category with a relatively low percentage of eaters, but for which canthaxanthin is used at a relatively high level can appear to provide a large contribution to the total intake (e.g., seafood soup mixes). Additionally, a low percentage of eaters would suggest that the data are not necessarily representative of the U.S. population.
Second, the mean total sample intake of canthaxanthin (Table 5) is greater than the intake at the 90th percentile. When the number of eaters in a group of foods is relatively small, the distribution of food (and ingredient) intake can be skewed toward the higher percentile consumers. As the mean intake shifts toward higher percentiles, it can exceed the intake at the 90th percentile. In this example, however, the total sample intake distribution is skewed toward higher percentiles due to the input of two use levels for the "pasta-based main or side dishes" category that differ by approximately three orders of magnitude (0.068 mg/kg and 100 mg/kg). As a result, the total sample intake distribution of canthaxanthin for pasta-based foods is bimodal and skewed toward the higher percentiles (primarily due to those foods containing 100 mg/kg canthaxanthin), which contributes to the total sample mean intake for the pasta-based foods being greater than the intake at the 90th percentile. Consequently, because the pasta-based food category also has the largest percent eaters of all the food categories, the mean cumulative total sample intake is also greater than the cumulative intake at the 90th percentile.
Third, the cumulative eaters-only intakes at the mean and 90th percentile (Table 5) are less than for any single food category (e.g., seafood soup mixes and pasta-based main or side dishes, Table 4). This is because the percent eaters for all food categories is higher than that for any one food category. As the percent eaters increases, intake will decrease because the intake (food or ingredient) is divided by the percent eaters (see Step 4 in the description of the calculation method, above).
Fourth, the total-sample intakes are, in general, lower than the respective eaters-only intakes (Tables 4 and 5) because the former include "non-eaters" of the foods, whereas the latter include only eaters.
Fifth, the per capita estimates are generally lower than total-sample intakes based on food consumption data because of the limitations associated with poundage data (e.g., underreporting of poundage), as discussed previously.
C. Processing Aids
A processing aid is often not expected to remain in the finished food after its technical effect has been accomplished, nor to have any intended effect in the food should residues remain. Therefore, the use level is not pertinent to calculating the EDI unless, as a worst-case scenario, one assumes that all of the additive is carried over to or remains in the treated food. This assumption, however, is rarely needed for processing aids.
Generally, the concentration data for a processing aid are obtained by analyzing the treated food for residues of the additive. These data are used in conjunction with food intake data to obtain the EDI. For some processing aids, however, this procedure is not sufficient to determine the EDI. An ion-exchange resin, used as the support for an immobilized preparation of glucose isomerase in the preparation of high fructose corn syrup, presents such a case, as shown in the next example.
Example 5. Intake of Enzyme Immobilizing Agents and Their Residues
The daily intake of residues from an ion-exchange resin used for the immobilization of glucose isomerase during manufacture of high fructose corn syrup (HFCS) is calculated based on assumptions concerning the useful service life of the resin:
- i) the resin is 8% of the enzyme preparation
- ii) 1 gram of enzyme preparation produces 11,000 grams HFCS
- iii) 20% of the resin is lost into the HFCS through column leaching over the service life of the column
- iv) The intake of HFCS is 63 g/p/d (per capita 2002 USDA-ERS estimate)
Based on these assumptions,[57] the resulting intake of the resin from consumption of HFCS is 91 mg/p/d (i.e., (63 g HFCS/p/d) x (1.45 x 10-6 g resin/g HFCS)). This EDI reflects the usual conservatisms: all HFCS entering the marketplace is produced using the resin; all column degradation leachate will remain in the HFCS; the production of HFCS will always be carried out using the maximum enzyme preparation-to-syrup ratio; and the consumer is exposed to the HFCS daily over a lifetime. If additional information, such as the percent leachate removed during processing, were provided, these data could be used to refine the EDI.
D. Chemical Contaminants and Other Constituents
The procedures for estimating the intake of a contaminant or adventitious constituent in a food generally parallel those used for intentionally added ingredients. It is imperative, however, to understand the nature of the concern for the substance in question in order to perform an intake estimate that is useful. As mentioned previously, impurities can be present in foods as a result of the manufacturing process (including through their presence in ingredients) or they can arise from natural or environmental sources.
In the case of a petitioned or notified ingredient, OFAS could rely on a specified maximum limit for a contaminant or other constituent for estimating the EDI (the worst-case scenario) to determine whether a safety concern exists. A petitioner or notifier might be able to supply additional data on the actual level of the impurity in an ingredient to allow an adjustment of the EDI, if needed. In the case of a contaminant in food, however, the concentration chosen for calculating its intake will depend on the available data and on the nature of the risk. A worst-case scenario may be overly alarmist. The concentration used for a contaminant posing an acute risk would probably be the maximum of the observed experimental levels. If the risk is chronic in nature, a mean concentration, to represent the likely daily, weekly, or monthly intake over a lifetime, would be more appropriate.
For the simple case (no acute risk) for which the presence of an impurity in a single product or class of products might pose a safety concern, the intake of the impurity can be calculated from its mean concentration in the food and the intake of the food. The next example illustrates the case of a naturally-occurring contaminant in an ingredient with a limited use. More complex cases (e.g., presence of a contaminant in multiple foods) require the use of the same methodology described above for multiple-use food ingredients.
Example 6. Intake of Residues in a Refined Food Oil
A refined food oil is intended for use in candies and confections (i. e., chocolate products) at levels from 5-30%. The crude oil contains 3-7% unsaponifiable matter that could be reduced to a specified maximum of 1.5% upon refining. The unsaponifiable material remaining after refining consists primarily of triterpene alcohols and sterols, including α-spinosterol. As α-spinosterol is unique to this oil among the edible oils, an estimate of intake of this constituent is needed for the safety evaluation.
The intake of the oil from its use in chocolate products is calculated first. The chronic EDI is 2.2 g/p/d (2+ years). Data indicated that the typical residual sterol level in the oil was approximately 0.05%, of which 40% is α-spinosterol. Therefore, use of the oil in chocolate results in a total potential intake of approximately 1.1 mg/p/d sterols ( i.e., (0.05% sterol in oil) x (2.2 g oil/p/d)), and the intake of α-spinosterol is 40% of that for all sterols (i.e., 0.4 mg/p/d).
Example 7. Intake of Acrylamide in Foods
Acrylamide has been detected in a wide range of food products. In general, high-temperature cooking methods, such as frying, roasting, or baking, can lead to formation of acrylamide in specific foods. However, the levels formed vary widely among different foods and among production lots of the same food products. FDA formulated a model for estimating intake of acrylamide from the consumption of various foods common in the diet using Monte Carlo simulations.[58] For this analysis, the intake distribution for each food (survey-derived) was combined with the analytically-determined levels of acrylamide in each food. The acrylamide intake from each food was calculated according to the following simple model:
[food intake] x [acrylamide level] x [eaters (yes or no)].
The incremental intakes from each food were added, after correcting for the percentage of eaters of each food, to produce an intake for each iteration of the model. These summed intakes, over 25,000 iterations, were combined to derive the distribution of overall intake of acrylamide. The data and model used to calculate the acrylamide intake are available on the FDA website.[59] Using this model, FDA estimated that mean acrylamide intake from consumption of more than 60 food types is 0.4 μg/kg bw/d.
E. Special Cases
The above examples point out some of the considerations routinely used by OFAS in preparing an intake estimate. For many cases, however, additional or different assumptions are made because of novel factors that arise. Two examples will be discussed: intake of components of tablets (e.g., vitamin supplements) and the use of dietary scenarios. Other special cases may arise that would require development of different approaches than those outlined here.
Example 8. Intake of Tableting Agents
Tableting agents are used in the preparation of vitamin and mineral supplements. Because the pattern of consumption of dietary supplements differs from the pattern of intake of typical foods, FDA needed a novel approach for the intake evaluation. FDA had previously determined, through a telephone survey in 1980, that the average daily intake of vitamin or supplement pills in the U.S. was one tablet and that one tablet weighed approximately 600 mg.[60] Since the 1980 survey, consumer awareness of vitamin and mineral supplements has increased. More recent survey data (NHANES 1999-2000) indicates that the mean number of supplements taken by adults has increased to nearly two per day [61]. Therefore, FDA assumes an intake value of two tablets per day for estimating intake of tableting agents in dietary supplements.
As an example calculation, a fatty acid ester is intended as a formulation aid in tablets at a level of 1-3%. Assuming ingestion of two 600 mg tablets per person per day, with each tablet containing no more than 3% fatty acid ester, the intake of the ester is 36 mg/p/d. The assumption that all dietary supplements are formulated with the fatty acid ester at its maximum intended use level adds a conservative element to the estimate.
Since 1994, consumer intake of dietary supplements has likely increased dramatically. Future considerations of intake estimates of components of dietary supplements should reconsider the validity of the assumption of two dietary supplement tablets per person per day.
Example 9. Dietary Scenarios
When relevant food consumption or substance concentration data are unavailable, a dietary scenario can be developed to complete an intake analysis. For example, OFAS might be asked to assess the safety of one lot of a rarely eaten food product that is being considered for regulatory action because of a contaminant that could pose a health risk. In this instance, an intake estimate for the contaminant could be based on a dietary scenario that uses the amount of the food consumed per eating occasion (or per day). This quantity can be combined with either the experimentally-determined concentration of the contaminant or a concentration chosen to be representative of the contaminated lot.
In the 1980s, fish caught downstream from paper mills were found to contain elevated levels of chlorinated dioxins and furans, a family of related chlorinated compounds associated with the chlorine bleaching of wood and paper products, which are known carcinogens at extremely low levels. Because fish consumption data were unavailable at the time for the target population of subsistence fishermen, OFAS created a dietary scenario that assumed that the normal intake of fish protein by this group equaled the total protein of the average person's diet of red meat, chicken, and fish. Using data from a national food consumption survey, exposure to dioxin was calculated assuming that the red meat, chicken, and fish consumed by the subsistence fishermen were contaminated at the levels in the fish caught near the mills.
The estimation of intake of a macronutrient substitute, such as a fat replacer, that could affect the nature of some consumers' diets, also required application of a dietary scenario. The potential availability of a fat replacer raised concern that highly motivated individuals would substitute the replacer for all added fat in their diet. The intake estimate for a fat replacer that could be used at home in food preparation that included baking and frying, was based on consideration of food energy obtained from fat intake. Assuming a man (19-50 years) requires 2,900 calories per day[62] to maintain an appropriate weight and 40% of these calories are derived from fat intake (not unusual for a substantial fraction of the U.S. population), 1,160 calories per day would be consumed as fat. At 9 calories per gram of fat, fat intake would amount to 129 grams per day.
In the 1980s, approximately 50% of dietary fat was estimated to be derived from added triglycerides (lard, tallow, butter, margarine, vegetable oils, shortening).[63] Thus, "added fat" amounted to 64 grams per day. If the fat replacer were intended to substitute for "added fat" on a one-to-one basis, the EDI for the fat replacer would be 64 g/p/d. If the fat replacer is non-caloric (i.e., it is a non-metabolizable substance), the 64 grams of replacer represents a decrease of about 576 (9 x 64) calories from the reference caloric intake of 2,900 calories. If a consumer unknowingly ate, on average, 576 additional calories[64] to compensate for the shortfall and continued to maintain a 2,900 calorie diet, additional fat replacer would be consumed as part of these extra calories, presumably in the same proportion of calories from fat in the total diet (40%). This would result in an additional intake of 13 g/p/d (i.e., (576 total calories) x (0.4 calories fat/total calories) x (0.50 g added fat/g total fat) x (1 g fat/9 calories fat)) of the replacer, bringing the total fat intake to 77 g/p/d (i.e., 64 g/p/d + 13 g/p/d) as an upper limit for an average man.
The use of dietary scenarios is based on assumptions that are reasonable reflections of a consumer's actions. The assumptions are clearly delineated and defensible. If possible, a second independent approach to estimating intake is used to corroborate the result obtained using a dietary scenario.
[1] This guidance has been prepared by the Office of Food Additive Safety, Center for Food Safety and Applied Nutrition (CFSAN) at the U.S. Food and Drug Administration.
[2] For more information, access http://www.cfsan.fda.gov/~dms/opa-guid.htm#cg
[3] Generally, animal studies are used to determine an acceptable daily intake (ADI), that has to be greater than the estimated daily intake (EDI) to ensure safety.
[4] Paracelsus, 16th century.
[5] Code of Federal Regulations, Title 21, Part 184, Subpart B, Section 184.1095 (abbreviated as 21 CFR 184.1095).
[6] Prior-sanctioned substances are those materials that were approved for use in writing by the FDA or United States Department of Agriculture (USDA) before 1958. Regulations for prior-sanctioned substances are listed in 21 CFR 181.
[[7][ In accordance with a proposed rule (proposed 21 CFR 170.36; 62 FR 18938; April 17, 1997). See http://www.cfsan.fda.gov/~rdb/opa-gras.html
[[8][ "Petitioner" refers to any group, individual, or company that submits a petition to amend the regulations concerning food and color additives under the Act. "Notifier" refers to any group, individual, or company that submits a voluntary notification to FDA of their determination that a particular use of a substance is GRAS, in accordance with the GRAS proposed rule.
[9] For relevant issues, see: 1) Kroes, R., Muller, D., Lambe, J., Lowik, M.R.H., van Klaveren J., Kleiner, J., Massey, R., Mayer, S., Urieta, I., Verger, P., and Visconti, A. (2002) Food Chem. Toxicol. 40, 327-385. 2) FAO/WHO (1997) "Food Consumption and Exposure Assessment of Chemicals" WHO/FSF/FOS/97.5, Geneva.
[10] Fifth edition, published by the National Academy Press, Washington, DC, 2003.
[11] 21 CFR 109 addresses unavoidable contaminants in food for human consumption.
[12] For relevant issues see: 1) Wait, A.D., and Bowers, T.S. (1993) Environ. Lab., 5 (Feb/Mar), 20-23. 2) Helsel, D.R. (1990) Environ. Sci. Technol., 24 (12), 1766-1774. 3) Travis C.C., and Land, M.L. (1990) Environ. Sci. Technol., 24 (7), 961-962. 4) Hornung, R.W, and Reed, L.D. (1990) Appl. Occup. Environ. Hyg., 5 (1), 46-51. 5) Kroes et al. (2002) cited in footnote 9.
[13] For more information, see Chemistry Guidelines for Direct Food Additive Petitions at Recommendations for Submission of Chemical and Technological Data for Direct Food Additive Petitions)
[14] For a discussion of data sources, and their use in intake estimation, see: 1) Life Sciences Research Office, Federation of American Societies for Experimental Biology (FASEB) (July 1988) "Estimation of Exposure to Substances in the Food Supply", Anderson, S.A., Editor, FDA Contract No. 223-84-2059, Life Sciences Research Office, Bethesda, MD. 2) Life Sciences Research Office, Federation of American Societies for Experimental Biology (December 1986) "Guidelines for Use of Dietary Intake Data". Anderson, S.A., Editor, Life Sciences Research Office, Bethesda, MD. 3) References cited in footnote 9. 4) WHO Offset Publication No. 87 (1985), "Guidelines for the Study of Dietary Intakes of Chemical Contaminants," WHO, Geneva.
[15] Cited in review article of Dybing et al. (2005) "Human exposure and internal dose assessments of acrylamide in food" Food Chem. Toxicol. 43, 365-410.
[16]For a review of food intake methods, see: 1) WHO (2000) "Human Exposure Assessment" Environmental Health Criteria 214, WHO, Geneva. 2) Kroes et al. (2002) cited in footnote 9.
[17] One measure of food portion sizes that may be used by FDA is the reference amount customarily consumed (RACC) per eating occasion. RACC values are available in 21 CFR 101.12 for the general food supply (individuals four years of age and older) and for infant and toddler foods.
[18] Diet History Questionnaire, Version 1.0. National Institutes of Health, Applied Research Program, National Cancer Institute, 2002. For more information, see http://riskfactor.cancer.gov/diet/assess.
[19] NHANES I-III were conducted by the National Center for Health Statistics (NCHS) of the Centers for Disease Control and Prevention (CDC), Department of Health and Human Services (DHHS).
[20] Useful information pertaining to food and nutrition surveys, including NCHS, CSFII, and NHANES, is available from the Food and Nutrition Information Center through the USDA/ARS website (www.nal.usda.gov/fnic/etext/000056.html).
[21] For a detailed discussion of food consumption surveys previously conducted by USDA, see: 1) Tippett, K.S., Enns, C.W., and Moshfegh, A.J. (1999) Nutr.Today 34, 33-46; and 2) www.ars.usda.gov/services/docs.htm?docid=7787.
[22] Madans, J.H., Sondik, E.J., and Johnson, C.L. (2003) J. Nutr. 133, 575S.
[23] Bodner, J.E., and Perloff, B.P. (2003) J. Food Compos. Anal. 16, 347-358.
[24] Due in part to respondent burden and survey costs, the CSFII surveys now collect fewer days of dietary recall data than in previous surveys. In the previous 1989-91 CSFII and 1985-86 CSFII Surveys, the number of observations were 3 and 6 non-consecutive 24-hour recalls, respectively. For further information, see 1) Carriquiry, A.L. (2003) J. Nutr. 133: 601S-608S; 2) Dwyer, J, Picciano, M.F., Raiten, D.J. and Members of the Steering Committee (2003) J Nutr 133: 609S-623S; 3) Institute of Medicine (2000) Dietary Reference Intakes. "Applications in Dietary Assessment" National Academy Press, Washington, DC. Relevant references cited therein include 1) NRC (1986) Nutrient adequacy. Assessment using food consumption surveys. National Academy Press, Washington, DC; 2) Nusser, S.M., Carriquiry, A.L., Dodd, K.W., and Fuller, W.A. (1996) J. Am. Stat. Assoc. 91: 1440-49.
[25] Published by National Academy Press, Washington DC, 2000-2004.
[26] Nationwide dietary intake data from NHANES 1999-2000 and NHANES 2001-2002 are available from the CDC through the NHANES website (www.cdc.gov/nchs/nhanes.htm).
[27]Examples of 7-day surveys are available at http://statistics.defra.gov.uk/esg/publications/efs/default.asp and http://europa.eu.int/comm/health/ph_projects/1999/monitoring/fp_monitoring_1999_annexe_fr_01_en.pdf Also see: 1) Turrini, A., Saba, A., Perrone, D., and Cialfa, E., D'Amicis, A, (2001) Eur. J. Clin. Nutr. 55(7), 571-588. 2) Volatier, J.L. (2000) Enquete INCA Individuelle et Nationale sur les Consonmations Alimentaires. Tec & Doc Lavoisier ed. Paris. 3) Becker, W., and Pearson, M. (1997) Riksmaten 1997-98. Dietary habits and nutrient intakes in Sweden 1997-98. (http://www.slv.se/templates/SLV_Page_6558.aspx).
[28] WHO (1985), see footnote 14.
[29] USDA publications that are available at www.ers.usda.gov/publications/sb965 include "Situation and Outlook" publications (e.g., "Sugar and Sweeteners") and "Food Consumption, Prices, and Expenditures, 1970-97" report (Putnam, J.J., and Allshouse, J.E. 1999), Economic Research Service, US Dept. of Agriculture, Statistical Bulletin No. 965.
[30] Available at www.ers.usda.gov/data/foodconsumption/.
[31] Putnam, J.J., and Allhouse, J.E. Food Consumption, Prices, and Expenditures, 1970-90. Economic Research Service, U.S. Dept. of Agriculture. Statistical Bulletin No. 840, 1992.
[32] Crane N.T., Lewis, C.L., and Yetley, E.A. (1992) Am. J. Pub. Health, 82, 862-866.
[33] "1987 Poundage and Technical Effects Update of Substances Added to Food." (1989) Committee on Food Additive Survey Data, Food and Nutrition Board, Institute of Medicine, National Academy of Sciences, Washington, D.C.
[34] Information about FDA's Total Diet Study is available at www.cfsan.fda.gov/~comm/tds-toc.html
[35] Several versions of the diets can be accessed through the TDS website at: http://www.cfsan.fda.gov/~comm/tds-food.html
[36] Reports from the workshops and links to WHO's database can be found at: http://www.who.int/foodsafety/publications/chem/en/.
[37] The use of "biomarkers" to estimate exposure to food chemicals has been discussed in a recent review article by Kroes et al. (2002), cited in footnote 9.
[38] Ponce, R.A., Bartell, S.M., Kavanagh, T.J., Woods, J.S., Griffith, W.C., Lee, R.C., Takaro, T.K., and Faustman, E.M. (1998) Regulatory Toxicol.Pharmacol. 28, 96-105.
[39] Turner, P.C., Nikiema, P., and Wild, C.P. (1999) Mutation Res. 443, 81-93.
[40]Anderson, W., Castle, L., Scotter, M., Massey, R.C., and Springall, C. (2001) Food Additives and Contaminants, 18, 1068-1074.
[41] Potischman, N., and Freudenheim, J.L. (2003) J. Nutr. 133, 873S-874S.
[42] The National Cancer Institute (NCI) and National Institute of Environmental Health Sciences (NIEHS) co-fund several research projects to develop exposure assessment methods applicable to epidemiological research on cancer (http://epi.grants.cancer.gov/ResPort/Biomarkers.html).
[43] Yetley, E.A., Beloian, A.M., Lewis, C.J. (1992) "Dietary Methodologies for Food and Nutrition Monitoring." In: Dietary Methodology Workshop for the Third National Health and Nutrition Examination Survey. DHHS Pub. No. (PHS) 92-1464, pp 58-67.
[44] EPA, Federal Register, 57, 22888-22938, May 29, 1992.
[45] OFAS uses commercially-available software packages that use USDA's 2-day 1994-96 CSFII and 1998 Supplemental Children's Survery or the NHANES studies.
[46] Summary data refer to all reports that are derived from primary data. That is, when primary intake data have been collated, weighted, and divided into age/sex, percentile, or other subgroupings, the resulting information is "summary" data.
[47] The probability of two independent events occurring simultaneously is the product of the probabilities of each event occurring.
[48] The reverse example, eaters of diet soft drinks always using packets of sweetener, could be true only if the size of the two populations were equal.
[49] WHO (1985), see footnote 14.
[50] For various probabilistic modeling of dietary exposure to micronutrients, pesticides, and acrylamide, see: 1) Food Additives and Contaminants 20 (Supp. 1), S1-S87, 2003. 2) Matthys et al. (2005) Food Chem. Toxicol. 43, 271-278.
[51] Commercial software for analysis using Monte Carlo methodology is available from a number of suppliers.
[52] Petersen, B.J., Chaisson, C.F., and Douglass, J.S. (1994) Am. J. Clin. Nut. 59 (Supp.), 204S.
[53] For more information, see http://www.tchpc.tcd.ie/montecarlo.
[54] (9 lbs pickles/p/yr) x (1 yr/365 d) x (454 g/lb) x (0.50 sweetened pickles/all pickles) = 5.6 g/p/d.
[55] A triangular distribution is so named because a minimum, a most likely, and a maximum value is used to delineate a probability function, which, graphically, is triangular in shape.
[56] See Codex Alimentarius Commission (www.codexalimentarius.net), Official Standards (Codex STAN 192-1995) and documents associated with the Codex Committee on Food Additives and Contaminants (CCFAC) on the Codex website for information related to the GSFA.
[57] 0.016 g of resin ((1 g enzyme preparation) x (0.20 leaching factor) x (8 g resin/100 g enzyme)) would be lost into the HFCS (11,000 g), resulting in a residual concentration of 1.45 x 10-6 g resin per g of HFCS (0.016 g resin/11,000 g HFCS).
[58] @Risk Software for Risk and Decision analysis: (http://www.palisade.com).
[59] Available online at http://www.cfsan.fda.gov/~dms/acrydino.html.
[60] Stewart, M.L., McDonald, J.T., Levy, A.S., Schucker, and R.E., Henderson, D.P. (1985) J. Am. Diet. Assoc. 85, 1585 - 1590.
[61] Radimer, K., Bindewald, B., Hughes, J., Ervin, B., Swanson, C., Picciano, MF. 2004. "Dietary supplement use by U.S. adults: data from the National Health and Nutrition Examination Survey, 1999-2000" Am J Epidemiol 160:339-349.
[62]Calories and weight: the USDA pocket guide. (1990). For more information, access http://www.hoptechno.com/book20.htm.
[63] Park, Y.K., and Yetley, E.A. (1990) Am. J. Clin. Nutr., 51, 738-748
[64] Caloric compensation in animals has been shown to occur when up to 40% of caloric material is substituted with inert materials. See Peterson, A.D. and Baumgardt, B.R. (1971) J. Nutr., 101(8), 1057-1068, and references therein.
The above guidance document supercedes the previous version dated September 1995.
Related Information
Submit Comments
Submit comments on this guidance document electronically via docket ID: FDA-2013-S-0610 - Specific Electronic Submissions Intended For FDA's Dockets Management Staff (i.e., Citizen Petitions, Draft Proposed Guidance Documents, Variances, and other administrative record submissions)
If unable to submit comments online, please mail written comments to:
Dockets Management
Food and Drug Administration
5630 Fishers Lane, Rm 1061
Rockville, MD 20852
All comments should be identified with the title of the guidance.