Expanded Access (Compassionate Use) Submission Data Archive: CDER and CBER
Expanded Access (Compassionate Use) Submission Data Archive: CDRH
CDER and CBER Archived Data (2010-2018)
CDER and CBER Tables
FY 2010 – 2018 Graphs of Expanded Access CDER and CBER Submissions
To view the graph select the appropriate link below.
- CDER Expanded Access IND and Protocol Submissions (2010-2018)
- CBER Expanded Access IND and Protocol Submissions (2010-2018)
- Combined CDER and CBER Expanded Access IND and Protocol Submissions (2010-2018)
CDER and CBER Tables
Expanded Access INDs for CDER and CBER (2010-2018)
|
Expanded Access INDs |
Individual (Single) |
Individual (Single) Patient |
Intermediate |
Treatment |
|||||
|---|---|---|---|---|---|---|---|---|---|
| received | allowed to proceed |
received | allowed to proceed |
received | allowed to proceed |
received | allowed to proceed |
||
|
FY 2018 |
CDER | 925 | 922 | 366 | 366 | 12 | 11 | 1 | 1 |
|
|
CBER | 123 | 52** | 162 | 162 | 8 | 7 | 1 | 1 |
|
FY 2017 |
CDER | 1111 | 1107 | 461 | 461 | 36 | 35 | 0 | 0 |
|
|
CBER | 40 | 36 | 80 | 70 | 11 | 10 | 2 | 2 |
|
FY 2016 |
CDER | 997 | 992 | 473 | 473 | 37 | 33 | 0 | 0 |
|
|
CBER | 28 | 27 | 90 | 90 | 5 | 5 | 4 | 2 |
|
FY 2015 |
CDER |
747 |
745 |
431 |
428 |
46 |
45 |
0 |
0 |
|
CBER |
32 |
29 |
68 |
66 |
2 |
1 |
2 |
2 |
|
|
FY 2014 |
CDER |
696 |
692 |
1069 |
1066 |
52 |
50 |
0 |
0 |
|
CBER |
22 |
19 |
45 |
44 |
1 |
1 |
1 |
1 |
|
|
FY 2013 |
CDER |
550 |
550 |
315 |
313 |
28 |
27 |
0 |
0 |
|
|
CBER |
37 |
31 |
112 |
112 |
10 |
8 |
0 |
0 |
|
FY 2012 |
CDER |
289 |
287 |
14 |
14 |
0 |
0 |
1 |
1 |
|
|
CBER |
47 |
39 |
36 |
35 |
1 |
1 |
2 |
2 |
|
10/13/2010 to 10/12/2011* |
CDER |
652 |
652 |
443 |
442 |
0 |
0 |
1 |
1 |
|
|
CBER |
22 |
18 |
24 |
24 |
4 |
4 |
1 |
1 |
|
10/13/2009 to 10/12/2010* |
CDER |
484 |
484 |
516 |
500 |
2 |
2 |
0 |
0 |
|
|
CBER |
17 |
14 |
24 |
24 |
1 |
1 |
0 |
0 |
* These reporting periods cover a one-year cohort starting the day the Final Rule for Expanded Access to Investigational Drugs for Treatment Use and Charging for Investigational Drugs went into effect. Starting with Fiscal Year 2012, the reporting period was changed to a fiscal year to match the reporting period for other IND activity reports.
** In FY18, CBER received a large number of single patient expanded access submissions for non-emergency use of the same product, for which FDA determined the risks associated with its use would be unreasonable for the patients involved or there was insufficient information to make a determination. Excluding these submissions, 91% of CBER’s non-emergency single patient expanded access submissions received were allowed to proceed.
Expanded Access Protocols for CDER and CBER (2010-2018)
|
|
Individual (Single) |
Individual (Single) Patient |
Intermediate |
Treatment |
|||||
|---|---|---|---|---|---|---|---|---|---|
| received | allowed to proceed |
received | allowed to proceed |
received | allowed to proceed |
received | allowed to proceed |
||
|
FY 2018 |
CDER | 27 | 27 | 20 | 20 | 14 | 14 | 5 | 5 |
|
|
CBER | 27 | 27 | 28 | 28 | 5 | 5 | 3 | 3 |
|
FY 2017 |
CDER | 14 | 14 | 4 | 4 | 4 | 4 | 7 | 7 |
|
|
CBER | 25 | 25 | 42 | 42 | 4 | 4 | 1 | 1 |
|
FY 2016 |
CDER | 16 | 16 | 6 | 6 | 6 | 6 | 10 | 10 |
|
|
CBER | 43 | 40 | 27 | 27 | 4 | 3 | 2 | 2 |
|
FY 2015 |
CDER |
14 |
14 |
7 |
7 |
9 |
9 |
8 |
8 |
|
CBER |
48 |
47 |
13 |
13 |
1 |
0 |
2 |
2 |
|
|
FY 2014 |
CDER |
35 |
35 |
9 |
9 |
9 |
9 |
12 |
12 |
|
CBER |
33 |
32 |
22 |
22 |
5 |
5 |
2 |
2 |
|
|
FY 2013 |
CDER |
62 |
62 |
2 |
2 |
8 |
8 |
12 |
12 |
|
|
CBER |
56 |
56 |
14 |
14 |
7 |
5 |
0 |
0 |
|
FY 2012 |
CDER |
121 |
121 |
0 |
0 |
8 |
8 |
10 |
10 |
|
|
CBER |
49 |
47 |
19 |
19 |
3 |
2 |
0 |
0 |
|
10/13/2010 to 10/12/2011* |
CDER |
89 |
89 |
3 |
3 |
1 |
1 |
11 |
11 |
|
|
CBER |
36 |
36 |
6 |
6 |
4 |
4 |
1 |
1 |
|
10/13/2009 to 10/12/2010* |
CDER |
16 |
16 |
0 |
0 |
5 |
5 |
7 |
7 |
|
|
CBER |
31 |
30 |
4 |
4 |
1 |
1 |
0 |
0 |
* These reporting periods cover a one-year cohort starting the day the Final Rule for Expanded Access to Investigational Drugs for Treatment Use and Charging for Investigational Drugs went into effect. Starting with Fiscal Year 2012, the reporting period was changed to a fiscal year to match the reporting period for other IND activity reports.
FY 2010 – 2018 Graphs of Expanded Access Submissions
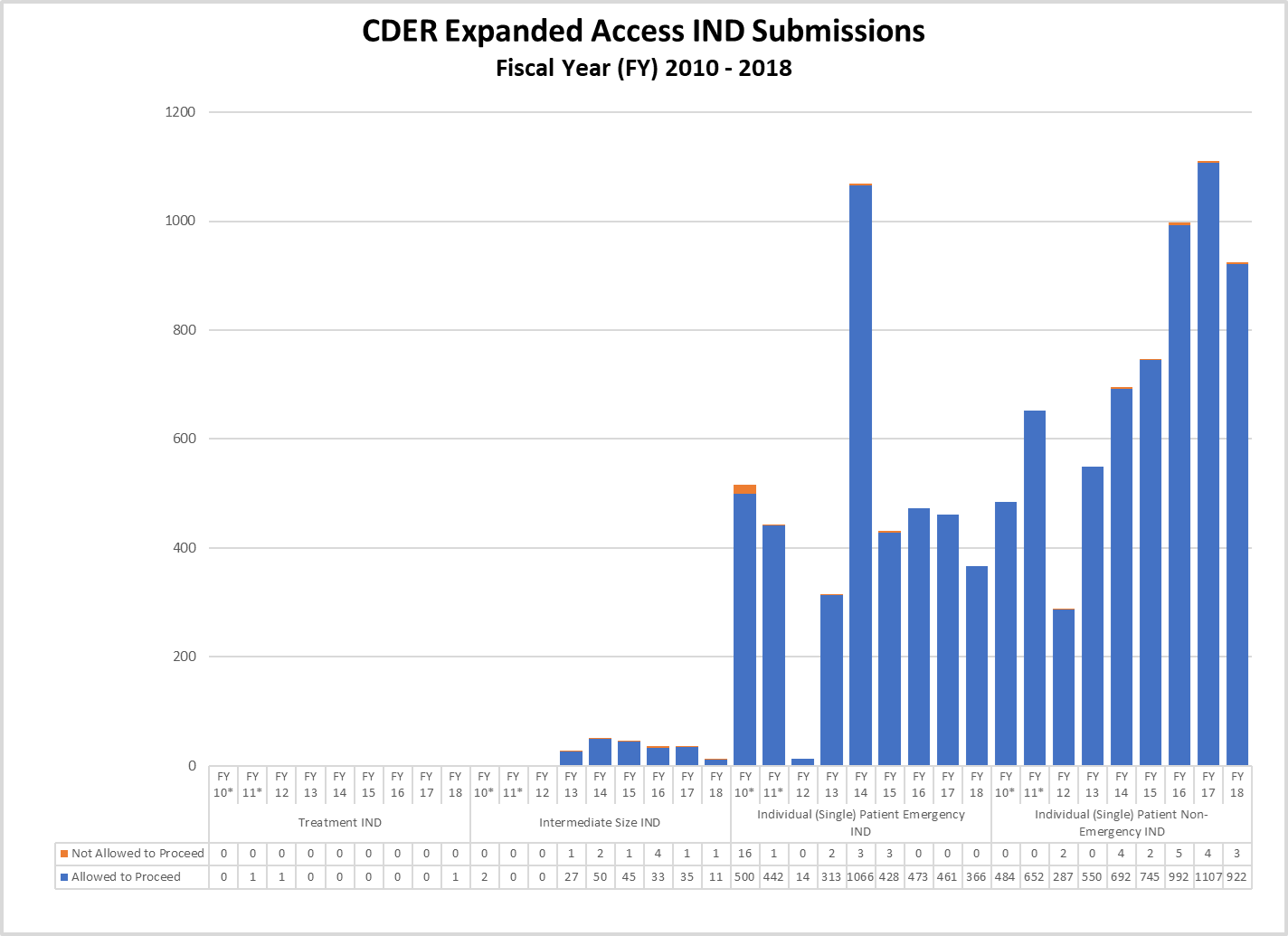
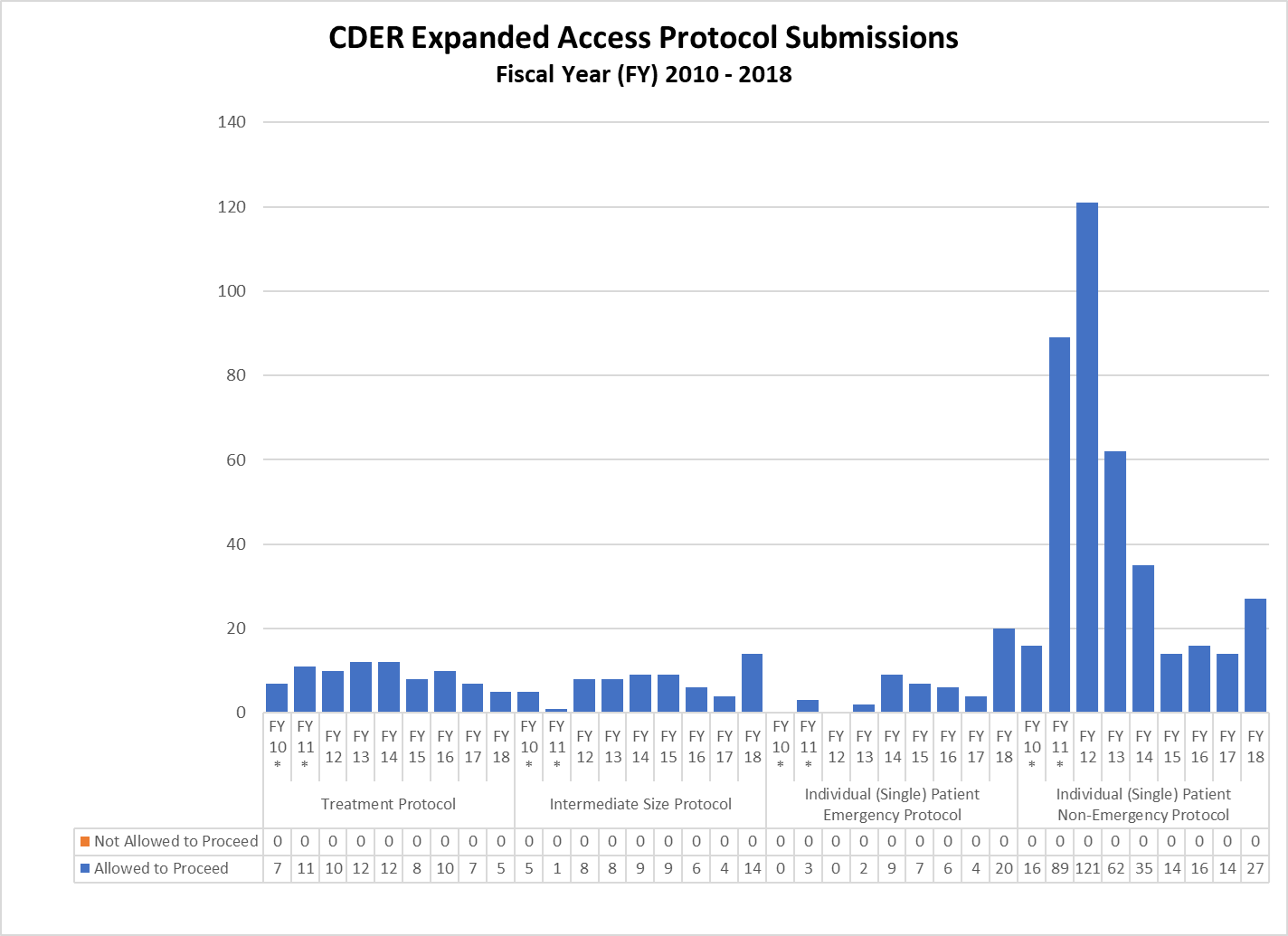
CDER Expanded Access IND and Protocol Submissions (2010-2018)
* These reporting periods cover a one-year cohort starting the day the Final Rule for Expanded Access to Investigational Drugs for Treatment Use and Charging for Investigational Drugs went into effect. Starting with Fiscal Year 2012, the reporting period was changed to a fiscal year to match the reporting period for other IND activity reports.
* These reporting periods cover a one-year cohort starting the day the Final Rule for Expanded Access to Investigational Drugs for Treatment Use and Charging for Investigational Drugs went into effect. Starting with Fiscal Year 2012, the reporting period was changed to a fiscal year to match the reporting period for other IND activity reports.
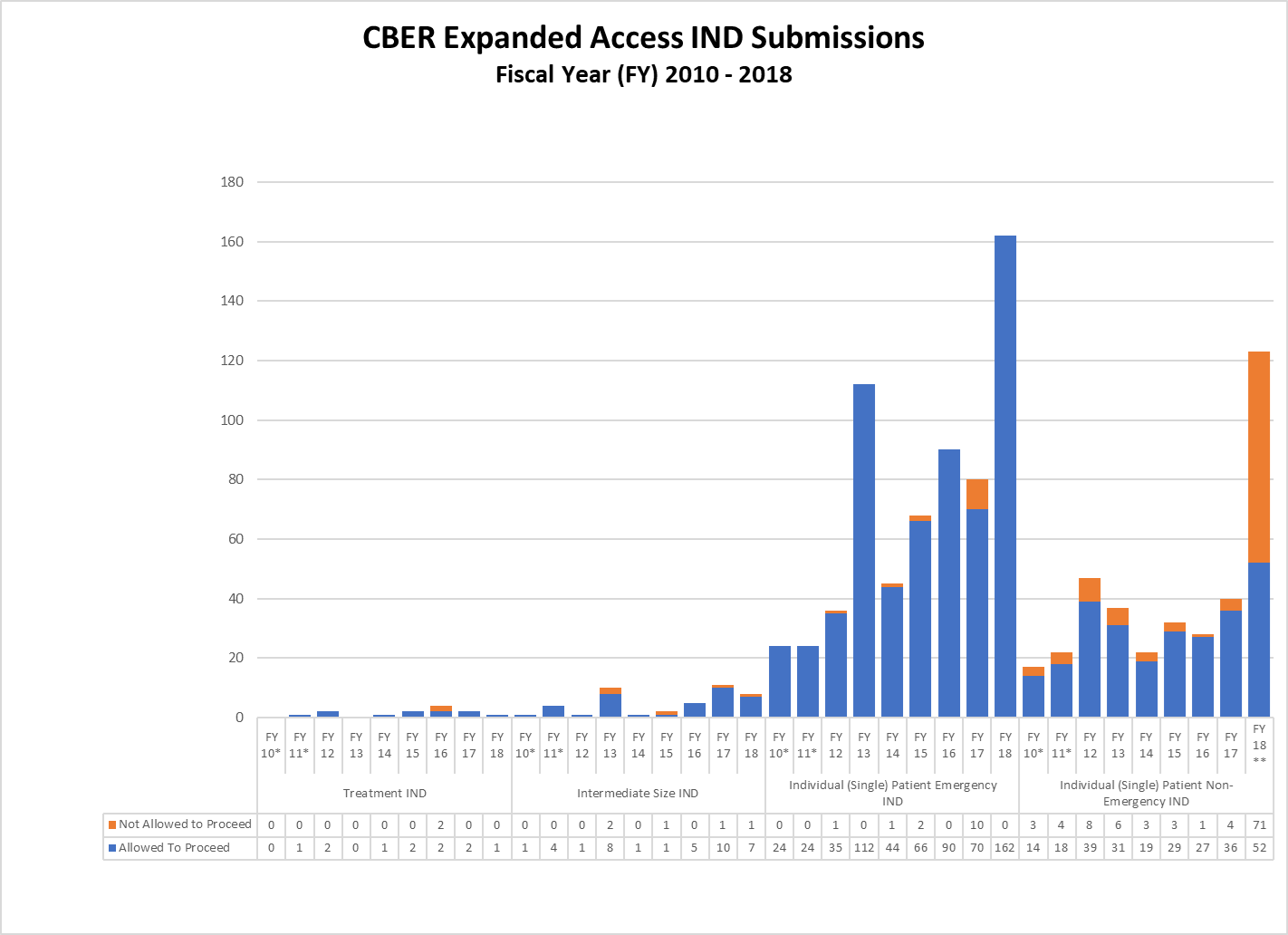
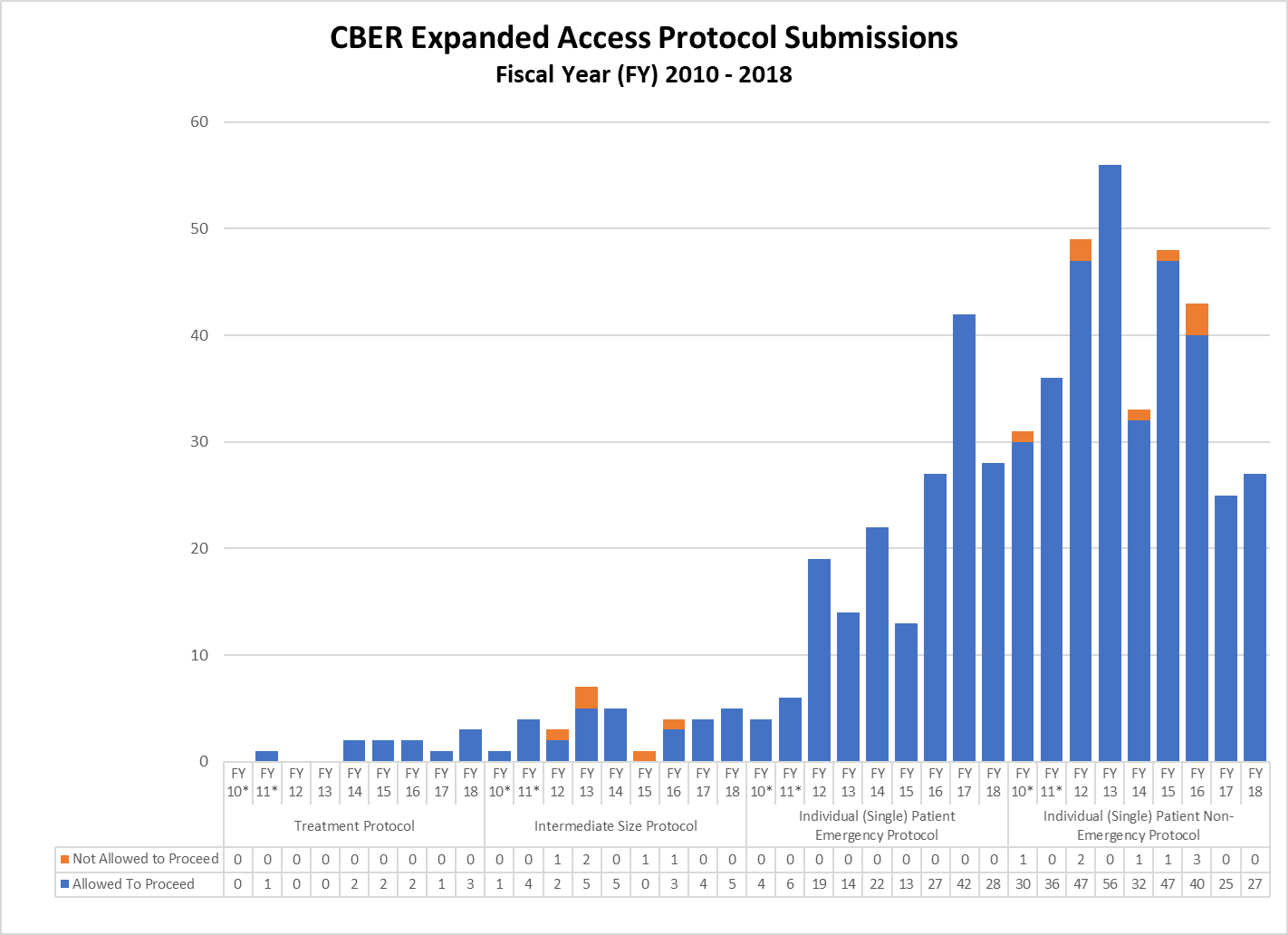
CBER Expanded Access IND and Protocol Submissions (2010-2018)
* These reporting periods cover a one-year cohort starting the day the Final Rule for Expanded Access to Investigational Drugs for Treatment Use and Charging for Investigational Drugs went into effect. Starting with Fiscal Year 2012, the reporting period was changed to a fiscal year to match the reporting period for other IND activity reports.
** In FY18, CBER received a large number of single patient expanded access submissions for non-emergency use of the same product, for which FDA determined the risks associated with its use would be unreasonable for the patients involved or there was insufficient information to make a determination. Excluding these submissions, 91% of CBER’s non-emergency single patient expanded access submissions received were allowed to proceed.
* These reporting periods cover a one-year cohort starting the day the Final Rule for Expanded Access to Investigational Drugs for Treatment Use and Charging for Investigational Drugs went into effect. Starting with Fiscal Year 2012, the reporting period was changed to a fiscal year to match the reporting period for other IND activity reports.
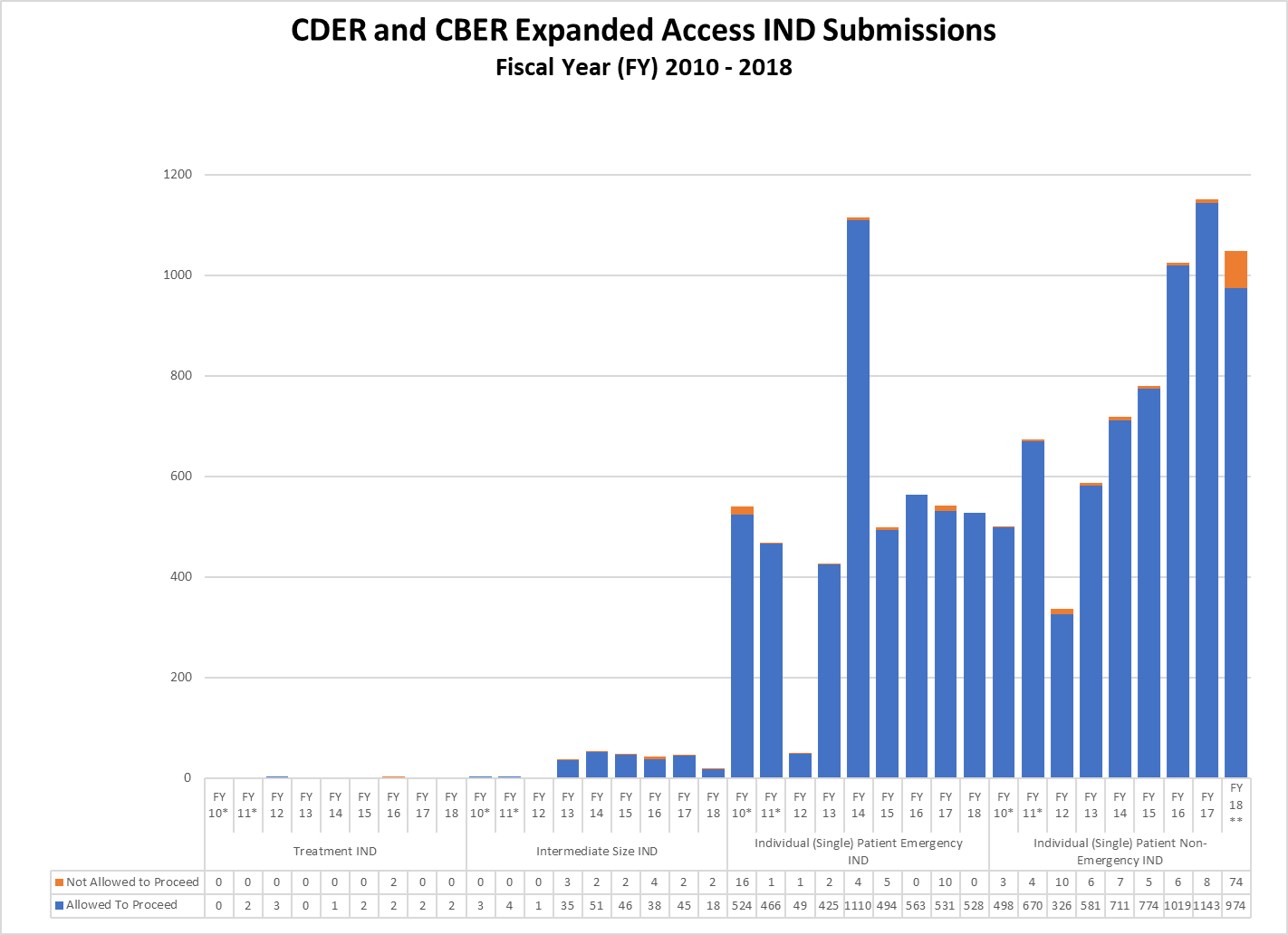
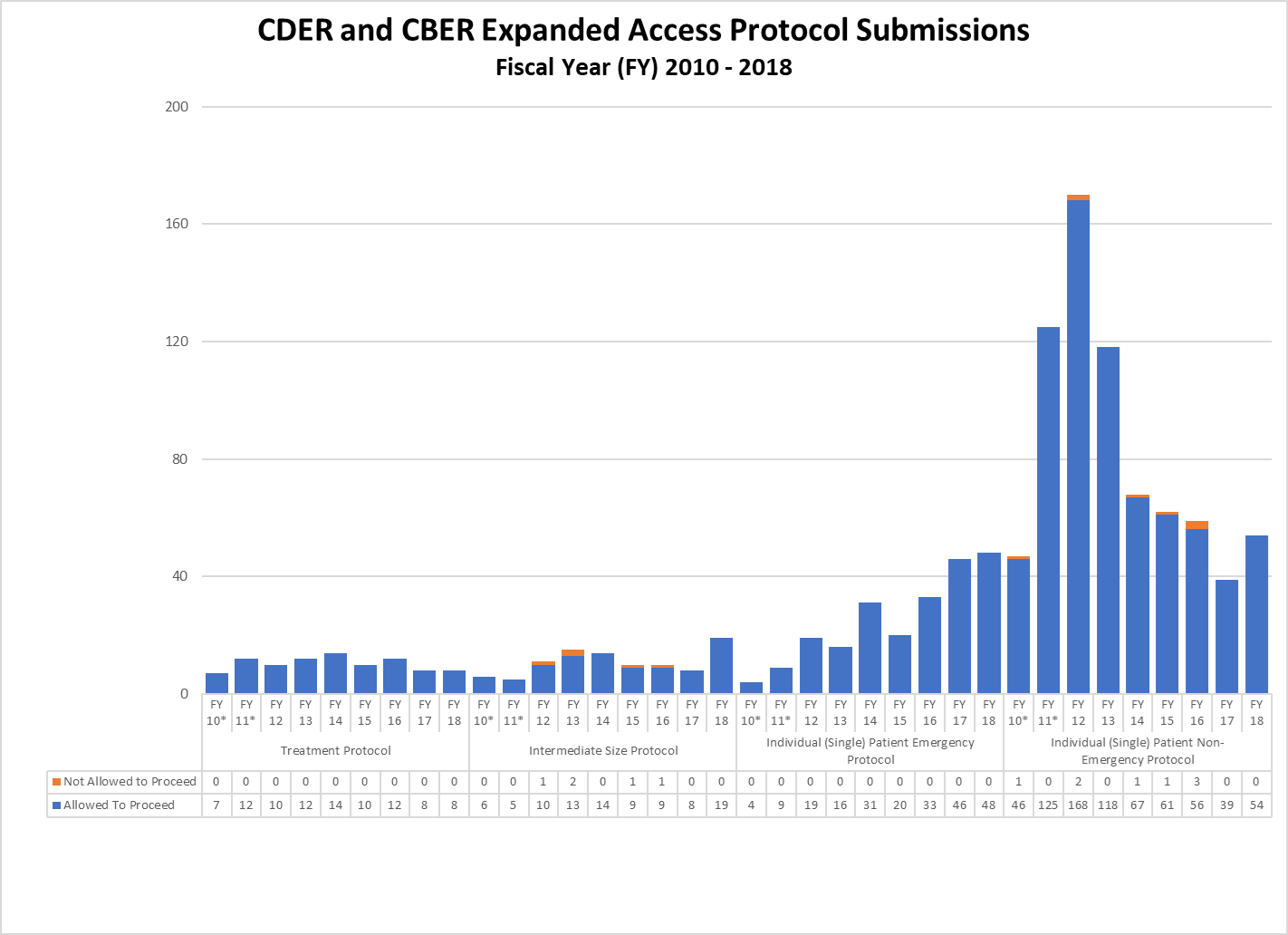
Combined CDER and CBER Expanded IND and Protocol Submissions (2010-2018)
* These reporting periods cover a one-year cohort starting the day the Final Rule for Expanded Access to Investigational Drugs for Treatment Use and Charging for Investigational Drugs went into effect. Starting with Fiscal Year 2012, the reporting period was changed to a fiscal year to match the reporting period for other IND activity reports.
** In FY18, CBER received a large number of single patient expanded access submissions for non-emergency use of the same product, for which FDA determined the risks associated with its use would be unreasonable for the patients involved or there was insufficient information to make a determination. Excluding these submissions, 91% of CBER’s non-emergency single patient expanded access submissions received were allowed to proceed.
* These reporting periods cover a one-year cohort starting the day the Final Rule for Expanded Access to Investigational Drugs for Treatment Use and Charging for Investigational Drugs went into effect. Starting with Fiscal Year 2012, the reporting period was changed to a fiscal year to match the reporting period for other IND activity reports.
** In FY18, CBER received a large number of single patient expanded access submissions for non-emergency use of the same product, for which FDA determined the risks associated with its use would be unreasonable for the patients involved or there was insufficient information to make a determination. Excluding these submissions, 91% of CBER’s non-emergency single patient expanded access submissions received were allowed to proceed.