Laboratory Information Bulletin (LIB) 4421: Melamine and Cyanuric Acid Residues in Infant Formula
Determination of Melamine and Cyanuric Acid Residues in Infant Formula using LC-MS/MS
Volume 24, October 2008
Sherri Turnipseed1, Christine Casey1, Cristina Nochetto2, David N. Heller2
1 Denver Laboratory, Office of Regulatory Affairs, Food and Drug Administration
2 Office of Research, Center for Veterinary Medicine, Food and Drug Administration
The Laboratory Information Bulletin is a communication from the Division of Field Science, Office of Regulatory Affairs, U.S. Food and Drug Administration for the rapid dissemination of laboratory methods (or scientific regulatory information) which appear to solve a problem or improve an existing problem. In many cases, however, the report may not represent completed analytical work. The reader must assure, by appropriate validation procedures, that the reported methods or techniques are reliable and accurate for use as a regulatory method. Reference to any commercial materials, equipment, or process does not, in any way, constitute approval, endorsement, or recommendation by the U.S. Food and Drug Administration.
Abstract
A liquid chromatography triple quadrupole tandem mass spectrometry (LC-MS/MS) method for residues of cyanuric acid (CYA) and melamine (MEL) developed by the Center for Veterinary Medicine (CVM) has been adapted for use with infant formula. This method consists of an initial extraction with 2.5% aqueous formic acid, followed by a series of filtration, centrifugation, and dilution steps. Both compounds are analyzed in the same chromatographic program using a zwitterionic HILIC LC column. Electrospray ionization is used in both the negative ion (CYA) and positive ion (MEL) modes. Two selected reaction monitoring (SRM) transitions are monitored for both compounds. The amount of compounds present is determined with a calibration curve consisting of sample extracts from infant formula fortified from 0.25 to 5 µg/g that have been taken through the extraction procedure. The range of recovery from fortified infant samples (n =38) was 70-114 % (RSDs 4.5- 22.7 %), and 72-110% (RSDs 5.7-24.9%) for cyanuric acid and melamine, respectively. The limits of quantification and confirmation are 0.25 µg/g for both analytes in dry infant formula.
Introduction
In 2007, pet food, animal feed, wheat gluten, and other protein-based food commodities were found to contain residues of melamine and a related compound, cyanuric acid. Widespread pet illness and death was subsequently attributed to the formation of melamine-cyanurate crystals in the kidneys of these animals. At that time, several laboratories developed methods for these compounds in pet food, raw protein sources, and other animal feeds. Because animals may eat food contaminated with melamine residues, analytical methods to determine melamine residues in fish and animal tissues were also developed. Recently, there has been a problem with contamination of infant formula with melamine and cyanuric acid in China. This is suspected of causing illness to many children in that country.
A procedure has been evaluated for the simultaneous determination and confirmation of melamine and cyanuric acid in powdered milk-based infant formula. Results are reported to support the continued evaluation and application of this method. Concentrations are measured in the range of 0.25 to 5 µg/g (µg/g). Extracts at higher concentrations may be diluted into this range. The limits of quantification and confirmation are 0.25 µg/g for both analytes.
Summary of Procedure
Powdered samples (2 g) are extracted with 14 mL 2.5 % aqueous formic acid in a 50 mL polypropylene centrifuge tube. Extracts are centrifuged at 3750 g and then 1.4 mL of supernatant is transferred to a polypropylene Eppendorf tube and centrifuged at 16100 g (benchtop micro centrifuge). The extracts are filtered through PVDF syringe filters (0.2 micron pore) followed by dilution with acetonitrile and a second centrifugation at 16100 g. The extracts are analyzed by LC-MS/MS. A zwitterionic HILIC column is used to separate melamine from cyanuric acid. Melamine is detected in positive ion mode and cyanuric acid is detected in negative ion mode. Analyte concentrations are calculated using external standard calibration with a standard curve prepared in a pre-fortified control matrix which has been carried through the extraction procedure.
Standards and Reagents
Melamine (MEL). CAS #: 108-78-1. Sigma Chemical Company.
Cyanuric acid (CYA). CAS #: 504-19-8. Fisher Chemical Company or TCI America.
Acetonitrile (ACN.) LC grade. Burdick & Jackson high purity or equivalent.
Formic acid. Reagent grade ≥ 95%. Sigma-Aldrich or Fisher Chemical Company.
Water. LC grade, or purified by Millipore Milli-Q system to > 18 M-ohm resistivity, or equivalent.
Ammonium Formate. Sigma-Aldrich or Fisher. Purity > 97%.
Solutions
0.1% Formic acid in water. Transfer 1 mL formic acid to 1 L graduated flask. Dilute to volume with LC water.
Mobile Phase A. 0.1% Formic acid in Acetonitrile (5:95 v/v). Combine 50 mL of 0.1% formic acid in water with 950 mL ACN in a 1 L solvent bottle.
Mobile Phase B. 20 mM Ammonium Formate in Acetonitrile (50:50 v/v). Combine 500 mL of 20 mM ammonium formate and 500 mL of acetonitrile in a 1 L solvent bottle.
2.5% Formic acid in water. Transfer 25 mL formic acid to 1 L volumetric flask. Dilute to volume with LC grade water.
20 mM Ammonium formate. Weigh 0.63 g of ammonium formate and dissolve in 0.5 L LC grade water.
Equipment
Liquid chromatograph. Binary LC pump is recommended for accurate mixing at low flow rate and rapid response to mobile phase gradient. (Waters, Agilent, Thermo Surveyor, or equivalent)
Liquid chromatography column. ZIC-HILIC, 2.1 × 150mm, 5µm, 200 Å, (Available in US from Nest Group #Q2712-152) (Merck Sequant)
Mass Spectrometer. Triple quadrupole capable of meeting system suitability. System used in the validation of infant formula was a Thermo (San Jose, CA) TSQ Quantum triple quadrupole mass spectrometer coupled to a Thermo Surveyor LC-MS pump and autosampler.
Centrifuge. Capable of 4000 RPM with 50 mL tubes.
Microcentrifuge. Capable of 13,000 RPM with 1.5 or 2 mL tubes. (Eppendorf or equivalent)
Mixers and shakers. Single and multi tube vortex mixers (VWR), platform shaker.
Utrasonic bath. Including timer and heater
Centrifuge tubes. 50mL disposable polypropylene with caps, with graduations from 5 to 50 mL and 1.5 mL microcentrifuge tubes.
Syringe Filters. Polyvinylidene fluoride (PVDF), 13mm, 0.22um (Whatman Puradisc™)
Syringes. Three mL polypropylene.
Procedure
Standard Preparation
Individual stock solutions, MEL or CYA, ca 100 µg/mL. Weigh approximately 10 mg of each standard using a weigh boat. Record actual weight to nearest 0.1 mg. Transfer to a 100 mL glass volumetric flask. Add 70 mL 0.1% formic acid in water and sonicate for ten min. Dilute to mark with 0.1 % formic acid in water and mix thoroughly. Calculate exact concentration, correcting for purity.
Standard mixture dilution, 50 µg/mL is used for fortification and matrix calibration standards. Using volumetric pipets, transfer 5.00 mL of each stock standard into a 20 mL glass scintillation vial (stable six months).
Standard mixture dilutions, 3.6 and 1.4 µg/mL are used to prepare post-fortified control extracts for calculating matrix effects. Pipet 720 (for 3.6 µg/mL) or 280 µL (for 1.4 µg/mL) of 50 µg/mL standard mix into separate 10 mL glass volumetric flasks and bring to mark with 0.1 % formic acid in water. Transfer to scintillation vials and store at room temperature (stable six months). Dilute mixed standards at 360 and 140 ng/mL are prepared by diluting the 3.6 and 1.4 µg/mL standards 1:10 with 0.1% formic acid.
Controls, Suitability Standard, Pre-fortified and Post-fortified Control Samples
Negative controls are samples that have been tested and show signals ≤ 10% of the 0.25 µg/g equivalent standard.
Extracted matrix calibration standards are prepared at 0.25, 0.5, 1.0, 2.5, and 5.0 µg/g, by adding 10, 20, 40, 100, and 200, µL of 50 µg/mL Standard Mix, respectively, to 2 g of sample.
Pre-fortified control standard is prepared, in addition to the matrix calibration curve standards, at the level of interest (0.5 or 1.0 µg/g) by adding an appropriate amount of 50 µg/mL Standard Mix to 2 g of dry sample.
Post-fortified control samples are negative control extracts to which 50 µL of Standard Mix 360 ng/mL or 140 ng/mL have been added at the final step, to give 2.5 and 1.0 µg/g equivalent extracts, respectively. These samples can be used to calculate matrix effects and percent recoveries.
Solvent standards to calculate matrix effects equivalent to 2.5 and 1.0 µg/g, prepare solvent standards at 18 and 7 ng/mL respectively. Add 50 µL of Standard Mix 360 and 140 ng/mL to 950 µL of ACN. The 7 ng/mL solvent standard can also be used to determine initial instrument system suitability.
Sample Preparation
Include at least one negative control and one pre-fortified control for quality assurance. If percent recoveries and matrix effects need to be evaluated, include control samples for post-fortification.
- Weigh sample powder (2.0 ± 0.1g) in a 50 ml polypropylene centrifuge tube.
- Pre-fortify control and matrix calibration standards.
- Add 14 mL of 2.5% Formic acid in water to samples. Seal the tube tightly, dissolve by shaking for 15-30 sec (vortex as needed), then sonicate in ultrasonic bath and mix on multi vortex mixer for 30 min each.
- Centrifuge at 4000 rpm (3750 g) for 10 min at room temperature.
- Transfer approximately 1.4 mL of the supernatant into a 1.5 mL micro centrifuge tube.
- Centrifuge at 13,200 rpm (16100 g) for 30 min.
- Load aqueous extract into a plastic 3 mL syringe and force through a 13mm, 0.22um PVDF filter into a micro centrifuge tube. (Note: some formulations may require some force, or two filtration steps to obtain a clear solution before the next step.)Possible stopping point: aqueous extracts can be stored at 5-10 °C for future dilutions.
- Dilute sample extracts with ACN as follows: (Note: precipitation of proteins is observed at this step.).
- For controls, fortified samples, and matrix calibration standards, transfer 50 µL of the filtered extracts into a 1.5 mL microcentrifuge tube and dilute with 950 µL of ACN.
- For post-fortified control samples at 1.0 or 2.5 µg/g, transfer 50 µL of control extract and 50 µL of 140 or 360 ng/mL standard, respectively, into 900 µL of ACN.
- Vortex mix for 30 sec and centrifuge at 13200 rpm (16100 g) for 30 min.
- Transfer the supernatant to a 2 mL autosampler vial, avoiding the precipitate.
Instrument Operating Parameters
Inlet LC method
Mobile Phase A: ACN /0.1% Formic Acid in water (95/5).
Mobile Phase B: Ammonium Formate 20mM/ACN (50/50).
Column temperature: 30°C.
Injection volume: 10 µL.
Run time: 14 min.
Retention time Cyanuric acid: 3.1 min.
Retention time Melamine: 5.5 min.
Backpressure: 400 psi new column, 2000 psi maximum acceptable.
Divert valve. Divert the LC flow to waste for the first 2 min after injection.
Divert the LC flow to waste 10 min after injection.
| Time (min) | % A | % B | Flow rate (mL/min) | Gradient | Comment |
|---|---|---|---|---|---|
| 0 | 100 | 0 | 0.4 | initial | Cyanuric acid elution |
| 4.2 | 100 | 0 | 0.4 | Linear | |
| 8.0 | 65 | 35 | 0.4 | Linear | Melamine elution |
| 8.5 | 65 | 35 | 0.4 | Linear | |
| 9.0 | 25 | 75 | 0.4 | Linear | Post-elution column wash |
| 11 | 25 | 75 | 0.4 | Linear | |
| 11.2 | 100 | 0 | 0.6 | Linear | Faster equilibration |
| 13 | 100 | 0 | 0.6 | Linear | for next run |
| 14 | 100 | 0 | 0.4 | Linear |
LC-MS/MS Operating Parameters. Tune the mass spectrometer by infusing separate solutions of MEL and CYA (10 µg/mL in water at 10 µL/min) into Mobile Phase A at 0.4 mL/min. (Absolute response for MEL melamine product ions is roughly 25-fold higher than for CYA).
| Tune Parameter | Cyanuric acid | Melamine |
|---|---|---|
| Ionization Mode | Negative | Positive |
| Source Voltage (kV) | 3.3 | 3.3 |
| Ion Transfer Tube Temperature (°C) | 270 | 270 |
| Auxillary Gas Flow (N2) | 5 | 0 |
| Sheath Gas Flow (N2) | 30 | 10 |
| Skimmer offset voltage (V) | 5 | 15 |
| Peak widths (Q1 and Q3) amu | 0.7 | 0.7 |
| Scan times (sec) | 0.5 | 0.5 |
| Scan width (amu) | 1 | 1 |
| Q2 Gas (Argon) Pressure (mTorr) | 1.5 | 1.5 |
| Compound | Acquisition Time, min | Precursor Ion (m/z) | Product Ion (m/z) | Collision Energy (eV) |
|---|---|---|---|---|
| Cyanuric acid | 0 – 4.2 | 128 (-ESI) | 85 | 11 |
| 42* | 17 | |||
| Melamine | 4.2 – 14 | 127 (+ESI) | 85* | 16 |
| 68 | 32 |
* SRM used for quantification. The ion ratio of each compounds' product ions are evaluated for confirmation.
Instrumental Analysis
Equilibrate the column in Mobile Phase A at 0.4 mL/min for 30-60 min.
To evaluate system suitability, inject solvent blank (1x), mixed standard at 7.0 ng/mL (3-4x). Data should meet the signal-to-noise and ion ratio criteria before continuing.
The recommended sequence of injections is: solvent blank (Mobile Phase A), extracted matrix standards from 0.25 to 5 µg/g, solvent blank, control extracts, post-fortified extracts and solvent standards for calculation of recoveries and matrix effects, solvent blank, unknown samples, and continuous calibration standards (an extracted matrix standard as well as solvent standard at 7 ng/mL), to verify that instrument response was maintained during the run. For longer analysis runs, greater than ~10 hours, the calibration standards can be reanalyzed at the end.
Calculations
Integrate peaks. Inspect integrations and manually correct if baseline-to-baseline integration was not achieved. Calculate signal-to-noise ratio for each peak.
Use external standard calibration. Quantification was performed using the instrument's Quan Browser and the quantitative product ion transition specified for each compound. The calibration curve should not include the origin, but does include a matrix blank with a concentration of 0. Export the processed data into Microsoft Excel or equivalent spreadsheet program for further calculations:
Recovery (%) = calculated from extracted calibration curve
Matrix effect (%) = 100 × post-fortified sample / solvent standard (same conc)
The limit of quantification (LOQ) for each analyte is defined as the concentration of the lowest calibration standard used, or the lowest calibration standard which shows > 10-fold higher response than background signals in negative control sample.
Calculations for Confirmatory Analysis
Calculate ion ratios as percent relative abundances. The MEL ion ratio is m/z 68/85. For CYA the ion ratio that is evaluated is m/z 85/42. Multiply by 100 to obtain percent relative abundances.
Stability of Analyte in Samples and Extracts
Extracts in aqueous formic acid are stable for one month in the refrigerator. Once the aqueous extract is combined with acetonitrile in the injection solution, it must be analyzed within 24 hours, or else re-diluted with acetonitrile.
Acceptance Criteria for Confirmation
Critical ions must be present and exceed a signal-to-noise ratio > 5:1.
The retention time of the analytes in the samples must be within 5% of the retention time of the averages of the standards in that batch.
Melamine: The product ion is within 10% of the average relative abundance of the matrix standards. The difference is found by addition/subtraction. For example, if the average relative abundance of the standards is 53% (typical for these conditions), the acceptance range for unknowns is 43% → 63%.
Cyanuric Acid: The product ion is within 10 % of the average relative abundance of the matrix standards. The difference is found by addition/subtraction. If the average abundance ion ratio of the standards is 66% (typical), the acceptance range for the abundance ion ratio of the samples is 56% → 76%.
Treatment of High Level Samples
It was found that animal feed samples containing > 50 µg/g melamine and cyanuric acid together, equivalent to > 0.36 µg/mL in the injection solution, tend to form the melamine:cyanurate complex in solution and therefore give inaccurate results. In order to determine concentration accurately under these circumstances, any aqueous acid extract which gives extremely high values should be diluted by serial ten-fold dilutions with 2.5% formic acid prior to acetonitrile dilution, until the response from analysis of the acetonitrile dilution falls within the calibration curve. Found concentration should be corrected according to the dilution factor used. While this has not been tested with infant formula samples, it is expected that similar issues would occur with high level samples.
Results
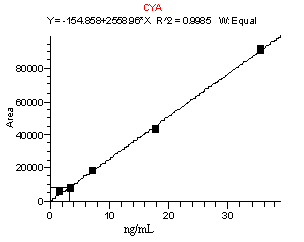
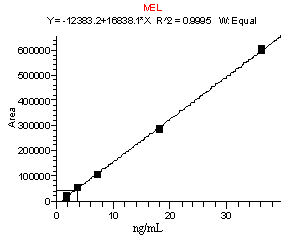
A method developed to monitor for CYA and MEL in animal feed was applied with very little modification for the determination and confirmation of these residues in infant formula. This method consists of an initial extraction of dry infant formula with 2.5% aqueous formic acid, followed by a series of filtration, centrifugation, and dilution steps. The concentration of compounds is determined with a calibration curve consisting of sample extracts from infant formula fortified to levels corresponding to 0.25 to 5 µg/g and taken through the extraction procedure. These calibration curves were linear with R2 values of greater than 0.995 (Figure 1). Recoveries of MEL and CYA for samples spiked from 0.25 to 5 µg/g are listed in Table IV below. In general, recoveries ranged from 70-114% for CYA and 71- 110% for MEL. The relative standard deviations (RSDs) were below 25% for all sample sets (n=3 to 7), and were typically 7-15%.
The majority of the validation was performed using one type of powdered infant formula (Brand A). In addition, matrix controls and 0.5 µg/g fortified samples using two other brands (one other powder and one liquid formulation) were also analyzed. Although the liquid infant formula was different in that it was "ready to feed", 2 g of that product were analyzed as well. The recoveries from the liquid formulation were lower for CYA (70%) and higher for MEL (110%). This may be an indication that it could be necessary to match the matrix curve more closely to the unknown samples. Some matrix effects were observed, with the signal for an extract post-fortified to match the final solution generally within ± 30% of the corresponding solvent standard. Usually an increased signal, or matrix enhancement, was observed. For example, when the area response for a 7 ng/mL post-fortified matrix-matched standard was compared to the same concentration of solvent standard, the ratios were 130% for CYA and 112% for MEL.
Figures 2 and 3 show SRM transitions for MEL and CYA from control infant formula and infant formula that had been fortified at 0.5 µg/g, respectively. The confirmation criteria were met for MEL in 6 of 7 fortified at 0.25 µg/g, and CYA was confirmed in 5 of these fortified samples. Both SRM transitions for MEL and CYA were detected in the samples that did not confirm, but the strict ±10% relative abundance criteria were not met. The relative abundance criteria were also not met in a few cases for CYA at the 0.5 µg/g fortification level, but a majority (14 of 16) was confirmed; MEL was confirmed in all samples fortified at 0.5 µg/g and higher. Solvent standards are used for ion ratio matching. Solvent blanks were analyzed after injections of high standards and no instrument carry-over was observed. Twelve matrix blanks were analyzed. Neither CYA nor nor MEL was confirmed in the matrix blanks at the limit of quantification.
| Sample | CYA Average % Recovery ± % RSD (n) |
MEL Average % Recovery ± % RSD (n) |
|---|---|---|
| Matrix Control Infant Formula (3 brands) | Not Confirmed (n = 12) | Not Confirmed (n = 12) |
| Method Blank | Not Confirmed (n = 1) | Not Confirmed (n = 1) |
| 0.25 µg/g Fortified Infant Formula A | 110.2 ± 14.8 % (n = 7) 5 of 7 confirmed | 83.2 ± 16.5 % (n = 6) 6 of 7 confirmed |
| 0.5 µg/g Fortified Infant Formula A-Day 1 | 94.0 ± 11.4 % (n = 5) 4 of 5 confirmed | 84.2 ± 5.7 % (n = 4) 5 of 5 confirmed |
| 0.5 µg/g Fortified Infant Formula A-Day 2 | 82.4 ± 22.7% (n = 5) 4 of 5 confirmed | 71.5 ± 24.9% (n = 5) 5 of 5 confirmed |
| 0.5 µg/g Fortified Infant Formula B | 114.3± 12.6% (n = 3) 3 of 3 confirmed | 83.8± 13.7% (n = 3) 3 of 3 confirmed |
| 0.5 µg/g Fortified Infant Formula C | 69.9 ± 4.9 % (n = 3) 3 of 3 confirmed | 109.9 ± 14.4 % (n = 3) 3 of 3 confirmed |
| 1.0 µg/g Fortified Infant Formula A | 91.7 ± 6.7 % (n = 7) 7 of 7 confirmed | 91.7 ± 10.7 % (n = 7) 7 of 7 confirmed |
| 2.5 µg/g Fortified Infant Formula A | 83.0 ± 8.3 % (n = 5) 5 of 5 confirmed | 88.9 ± 7.8 % (n = 5) 5 of 5 confirmed |
| 5 µg/g Fortified Infant Formula A | 91.9 ± 4.5 % (n = 3) 3 of 3 confirmed | 86.0 ± 7.3 % (n = 3) 3 of 3 confirmed |
Figure 1. Calibration curves for CYA (top) and MEL (bottom). Data obtained from the analysis of infant formula fortified from 0.25 to 5 µg/g and then taken through the extraction procedure.
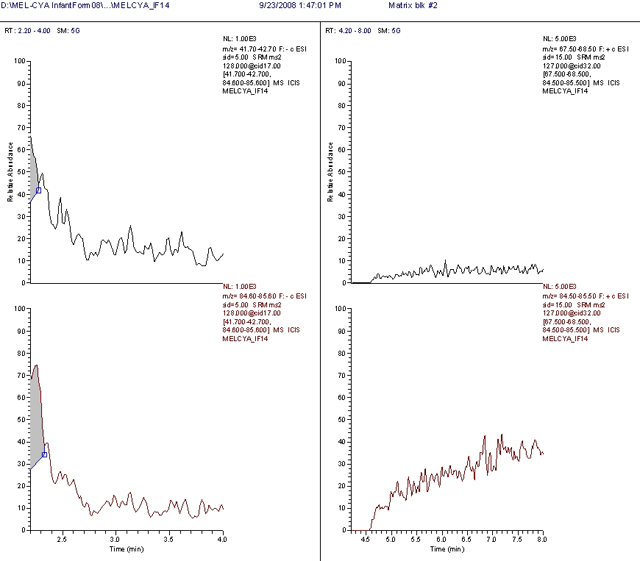
Figure 2. Control Infant Formula
Left: SRM traces for CYA (m/z 128 → 42, top and m/z 128 → 85, bottom)
Right: SRM traces for MEL (m/z 127 → 68, top and m/z 127 → 85, bottom)
Figure 3. Control Infant Formula fortified with 0.5 µg/g of CYA and MEL
Left: SRM traces for CYA (m/z 128 → 42, top and m/z 128 → 85, bottom)
Right: SRM traces for MEL (m/z 127 → 68, top and m/z 127 → 85, bottom)
References
Heller, DN, Nochetto, C. Simultaneous Determination and Confirmation of Melamine and Cyanuric Acid in Animal Feed by Zwitterionic HILIC Chromatography and Tandem Mass Spectrometry, in press, Rapid Commun. Mass Spectrom.
Litzau J, Mercer G, Mulligan K., Analytical Methods for Melamine and Triazine Analogs. Accessed May 7, 2013.
Andersen WC, Turnipseed SB, Karbiwnyk CM, Madson MR. U.S. FDA Laboratory Information Bulletin, 2007: 23: 4369. Lib. No. 4396. accessed May 7, 2013.
Karbiwnyk, CK, Andersen, WC, Turnipseed, SB, Storey, JM, Madson, MR, Miller, KE, Gieseker, CM, Miller, RA, Rummel, NG, Reimschuessel, R. Determination of cyanuric acid residues in catfish, trout, tilapia, salmon and shrimp by LC- MS/MS, in press, Anal. Chim Acta
Andersen WC, Turnipseed SB, Karbiwnyk CM, Clark, SB, Madson MR, Gieseker CM, Miller RA, Rummel NG, Reimschuessel R. Determination and Confirmation of Melamine Residues in Catfish, Trout, Tilapia, Salmon and Shrimp by LC-MS-MS. 2008, J. Ag. Food Chem 56: 4340.
Reimschuessel R., Gieseker, CM, Miller, RA, Ward, J, Boehmer, J, Rummel, N, Heller, DN, Nochetto, CN, de Alwis, H, Bataller, N, Andersen, WC, Turnipseed, SB, Karbiwnyk, CM, Satzger, D, Crowe, JB, Wilber, NR, Reinhard, MK, Roberts, JF, Witkowski, MR Evaluation of the renal effects of experimental feeding of melamine and cyanuric acid to fish and pigs. 2008, Am. J. Vet. Res., 69:1217.
Dobson RLM, Motlagh S, Quijano M, Cambron RT, Baker TR, Pullen AM, Regg BT, Bigalow-Kern AS, Vennard T, Fix A, Reimschuessel R, Overmann G, Shan Y, Dasont GP. Toxicol Sci, ePub Aug 9, 2008.
Chemical Name: melamine
IUPAC International Chemical Identifier: InChI=1/C3H6N6/c4-1-7-2(5)9-3(6)8-1/h(H6,4,5,6,7,8,9)
InChIKey: JDSHMPZPIAZGSV-UHFFFAOYAF
Chemical Name: cyanuric acid
IUPAC International Chemical Identifier: InChI=1/C3H3N3O3/c7-1-4-2(8)6-3(9)5-1/h(H3,4,5,6,7,8,9)
InChIKey: ZFSLODLOARCGLH-UHFFFAOYAR