FDA’s Infant Formula Product Testing Results
Introduction | About the Market Basket Survey | 2023-2025 Results
The FDA is committed to protecting babies and young children from exposure to contaminants. As part of Operation Stork Speed and our ongoing surveillance work, the FDA is testing infant formula for certain contaminants, such as lead, mercury, cadmium, arsenic, per- polyfluoroalkyl substances (PFAS), and pesticides, among others, to help ensure that infant formula remains safe, available, and nutritious for U.S. families.
Key Findings
Through the FDA’s rigorous oversight, monitoring, and testing protocols, parents and caregivers can continue to feel confident about the safety of infant formula. The FDA's results to date (listed below) represent the largest and most comprehensive examination of chemical contaminants in infant formula available on the U.S. market and affirm that infant formula is safe.
This work builds upon the FDA’s broader Closer to Zero initiative, which strives to reduce dietary exposure to contaminants to as low as possible, while maintaining access to nutritious foods. The initiative prioritizes foods for babies and young children because their smaller body sizes and developing organ systems make them more vulnerable to the potential effects of these contaminants.
In our initial phases of testing as a part of Operation Stork Speed, the FDA is conducting surveys of samples that are representative of the infant formula products most commonly sold in the U.S. Market basket samples are taken from products purchased at retail stores. The data serves as a signal-detection tool to inform the agency’s understanding of contaminant levels in food products and to guide our risk management approaches to promote and protect public health.
Market basket surveys provide a snapshot of products available at retail at a specific point in time and help identify trends that inform further testing. However, they do not capture the full range of variability that may occur across different production lots of the same product.
Contaminants can be present in foods, even in small amounts, due to their natural occurrence in soil, water, and air. Modern testing methods can now detect extremely small amounts of these contaminants in food, but it is important to remember that the detection of a contaminant does not automatically mean a product is unsafe.
Given the purpose and limitations of market basket testing, the FDA is conducting further testing and implementing risk management approaches, such as working to establish action levels for contaminants in infant formula, to ensure the ongoing safety of the infant formula supply. The FDA continues to engage with infant formula manufacturers to discuss potential ways through which these contaminants may enter the supply chain, including through ingredient sourcing and manufacturing processes, as well as measures to reduce the levels of these contaminants in infant formula to as low as possible.
FDA scientists are at the forefront of developing new and more sensitive testing methods, applicable in a variety of food matrices, to measure very low levels of certain contaminants.
To ensure accurate results when testing infant formulas for contaminants, it is necessary to use methods specifically developed and validated for foods, including infant formulas. Testing for contaminants requires implementing special conditions and procedures, such as clean room environments, dedicated glassware and instruments, and specialized cleaning procedures, to control for potential laboratory contamination.
When testing infant formula, the FDA’s Human Foods Program (HFP) utilizes specialized and specific methods that are developed and validated according to FDA Validation Guidelines in HFP labs. Most of these methods are available to the public, for review and use, through the FDA Foods Program Compendium of Analytical Laboratory Methods: Chemical Analytical Manual (CAM). In addition, see Elemental Analysis Manual (EAM) for Food and Related Products.
What Did We Test?
We tested 312 infant formula samples covering 16 brands, with multiple lots tested per brand.
- Formula type: 278 powdered, 11 concentrated liquid, 23 ready-to-feed
- Protein source: 258 cow's milk, 44 soy, 10 amino acid-based
What Did We Test For?
Arsenic, Cadmium, Lead, Mercury, 30 PFAS, 318 Pesticides, 21 Phthalates, and 1 Non-phthalate Plasticizer
Overarching Results
Across the products tested, the majority of infant formula samples had undetectable or very low levels of contaminants, affirming the safety of the U.S. infant formula supply. While contaminant levels were low, some samples had values that have initiated additional follow-up actions as a part of the Agency’s robust monitoring and oversight efforts. This will include conducting further testing and continuing to engage with manufacturers on measures to reduce contaminant levels to as low as possible.
Understanding the Results
For each contaminant we reported the following values to help illustrate typical levels measured:
- Not detected: The amount, if present, was too small for our testing methods to detect
- Range: The lowest to highest levels (concentrations) detected
- 95th percentile (reported as 95%): The level below which 95% of samples fell (meaning only 5% of samples were higher)
- Median: The midpoint in a set of data when the numbers are put in order from lowest to highest. It helps show a common number in the group.
- Parts per billion (ppb): One part per billion is equivalent to one drop of water in an Olympic-size swimming pool
The following is a summary of the results, presented as concentrations in formula as prepared for feeding to allow for comparison between samples.
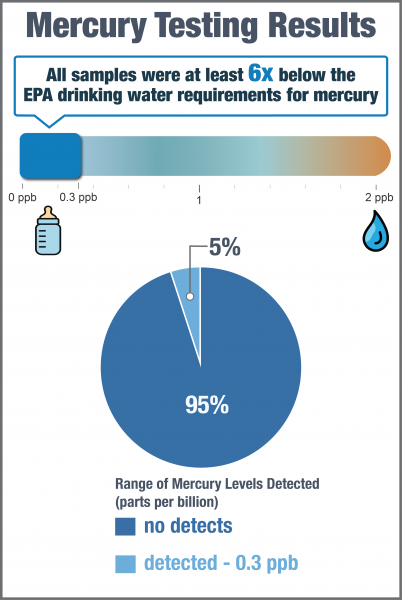
Mercury was not detected in 296 out of 312 samples (95%). Among all samples, concentrations ranged from not detected to 0.3 ppb. Given the high number of non-detects, a 95th percentile and median were not calculated.
Analytical Results for Toxic Elements in Infant Formula (FY2023-2025) in XLSX
Data separated by each of these elements: lead, mercury, cadmium, and arsenic.
Analytical Results for Toxic Elements in Infant Formula (FY2023-2025) in PDF
Data separated by each of these elements: lead, mercury, cadmium, and arsenic.
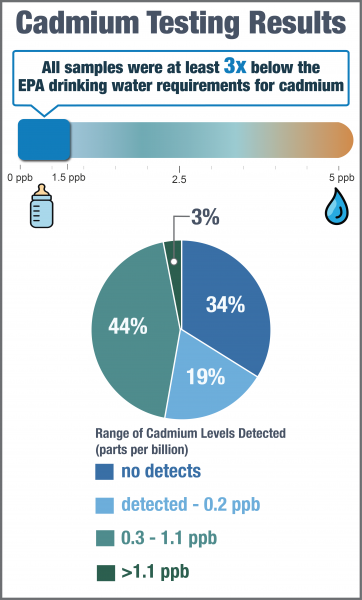
Cadmium was not detected in 106 out of 312 samples (34%). Among all samples, concentrations ranged from not detected to 1.5 ppb. 95% of all samples had less than 1.1 ppb of cadmium. The median cadmium concentration was 0.2 ppb.
Analytical Results for Toxic Elements in Infant Formula (FY2023-2025) in XLSX
Data separated by each of these elements: lead, mercury, cadmium, and arsenic.
Analytical Results for Toxic Elements in Infant Formula (FY2023-2025) in PDF
Data separated by each of these elements: lead, mercury, cadmium, and arsenic.
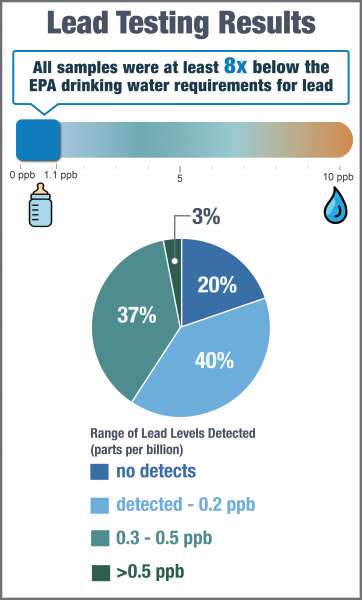
Lead was not detected in 61 out of 312 samples (20%). Among all samples, concentrations ranged from not detected to 1.1 ppb, with 95% of samples containing less than 0.5 ppb of lead. The median lead concentration was 0.2 ppb.
Analytical Results for Toxic Elements in Infant Formula (FY2023-2025) in XLSX
Data separated by each of these elements: lead, mercury, cadmium, and arsenic.
Analytical Results for Toxic Elements in Infant Formula (FY2023-2025) in PDF
Data separated by each of these elements: lead, mercury, cadmium, and arsenic.
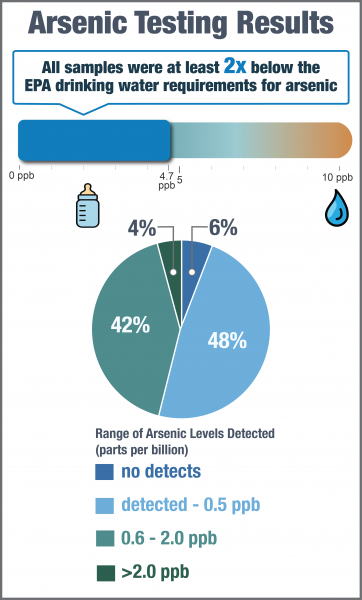
Arsenic was not detected in 18 out of 312 samples (6%). Among all samples, concentrations ranged from not detected to 4.7 ppb. 95% of all samples had less than 2.0 ppb of arsenic. The median arsenic concentration was 0.5 ppb.
*Testing of a subset of samples showed that arsenic, when present, was in its inorganic form.
Analytical Results for Toxic Elements in Infant Formula (FY2023-2025) in XLSX
Data separated by each of these elements: lead, mercury, cadmium, and arsenic.
Analytical Results for Toxic Elements in Infant Formula (FY2023-2025) in PDF
Data separated by each of these elements: lead, mercury, cadmium, and arsenic.
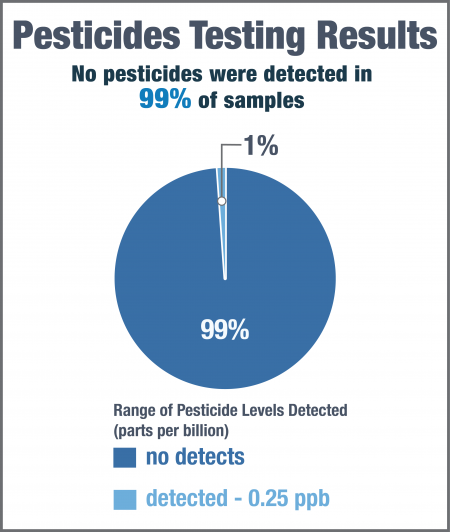
Each of the samples were tested for 318 different pesticides, including glyphosate and glufosinate. 309 samples (99%) had no detectable pesticides, and glyphosate and glufosinate were not detected in any samples. Given the high number of non-detects, a 95th percentile and median were not calculated.
Only three samples had detectable pesticide levels, all at very low concentrations: 0.25 ppb chlorpyrifos in one sample, 0.20 ppb malathion in another sample, and 0.20 ppb piperonyl butoxide in a third sample.
Analytical Results for Pesticides in Infant Formula (FY2023-2025) in XLSX
Analytical Results for Pesticides in Infant Formula (FY2023-2025) in PDF
Each of the samples were tested for 30 different PFAS compounds. Most PFAS compounds (25 of 30) were not found in any samples.
Five PFAS were detected in a subset of samples at concentrations ranging from 0.51 ppt to 150 ppt, depending on the compound. The most commonly detected was Perfluorooctanesulfonic acid (PFOS), found in half of samples at detectable concentrations ranging from 0.51 to 6.0 ppt, with 95% of all samples containing less than 2.9 ppt of PFOS. Given the high number of non-detects, the median was not calculated.
PFAS levels are often reported in parts per trillion (ppt) because testing methods can detect these contaminants at extremely low concentrations. One part per trillion is equivalent to one second in 32,000 years, or one drop in 1,000 Olympic-size swimming pools.
Analytical Results for PFAS in Infant Formula (FY2023-2025) in XLSX
Analytical Results for PFAS in Infant Formula (FY2023-2025) in PDF
Each of the samples were tested for 21 phthalates and 1 non-phthalate plasticizer. Phthalates were not detected in 167 out of 312 samples (54%). Among the compounds tested:
- Seven phthalates were not detected in any samples
- Five phthalates were rarely detected (in fewer than 2% of samples)
- The remaining compounds that were detected had concentrations ranging from not detected to 145 ppb.
The most commonly detected phthalate compounds were DEHP and DINP:
DEHP was not detected in 253 out of 312 samples (81%). Among the samples, DEHP concentrations ranged from not detected to 57 ppb. Given the high number of non-detects, a 95th percentile and median were not calculated.
DINP was not detected in 260 out 312 samples (83%). Among the samples, DINP concentrations ranged from not detected to 145 ppb. Given the high number of non-detects, a 95th percentile and median were not calculated.
Analytical Results for Phthalates in Infant Formula (FY2023-2025) in XLSX
Analytical Results for Phthalates in Infant Formula (FY2023-2025) in PDF
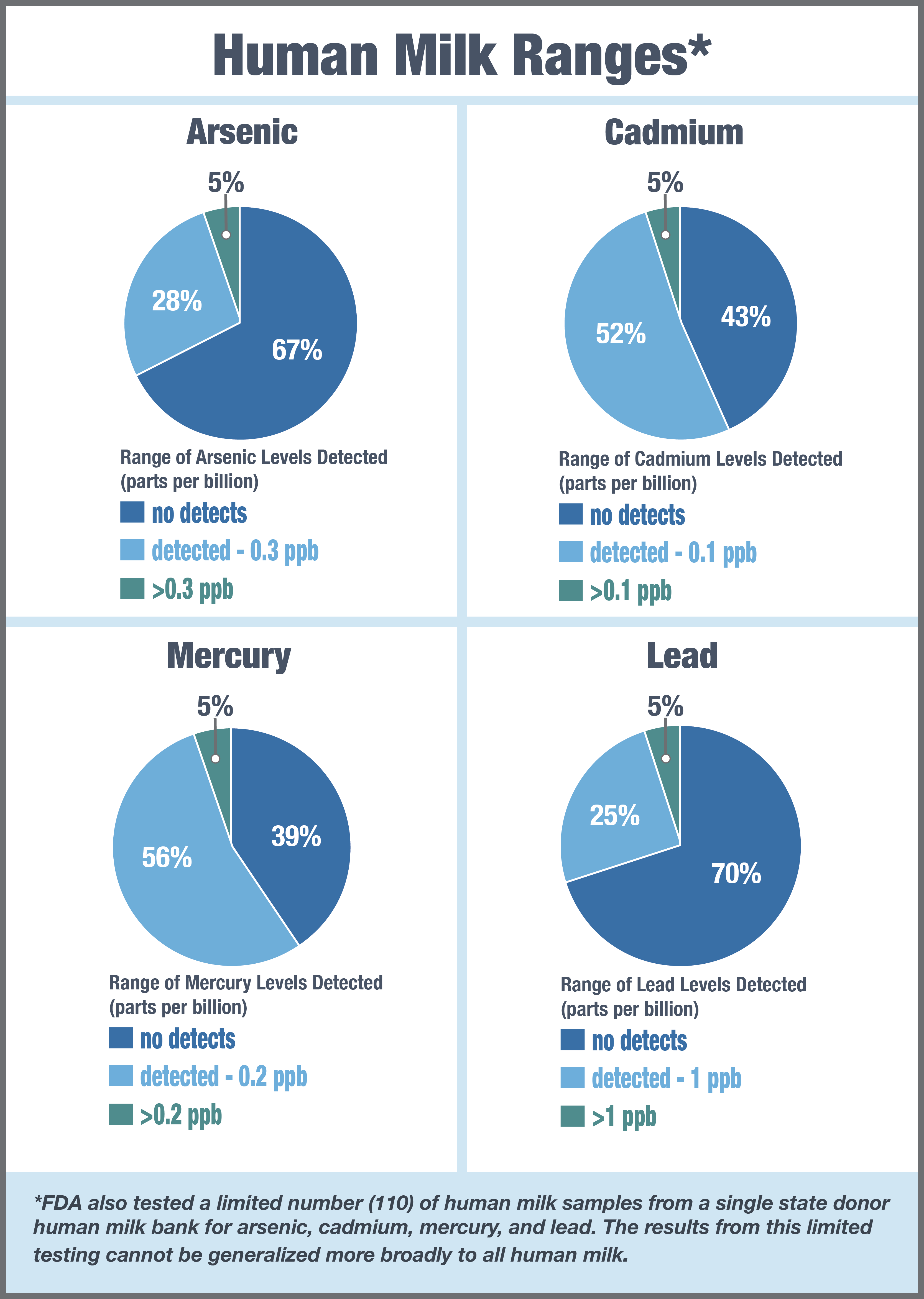
While this phase of testing focused specifically on infant formula products, the FDA also tested a limited number (110) of human milk samples from a single state donor human milk bank for arsenic, cadmium, mercury, and lead.
Overall, the majority of samples (85%) had at least one detectable contaminant, while 17 samples (15%) had no detects of any of the contaminants. Mercury demonstrated the highest detection rate at 61%, followed by cadmium at 57%, arsenic at 33%, and lead at 30%.
However, the results from this limited testing cannot be generalized more broadly to all human milk. While we recognize that naturally occurring and human-generated environmental contaminants can be present in human milk, breast milk remains the optimal source of nutrition for infants whenever possible.
Analytical Results for Human Milk (FY2023-2025) in XLSX
Analytical Results for Human Milk (FY2023-2025) in PDF