Summary of Process for EUA Issuance
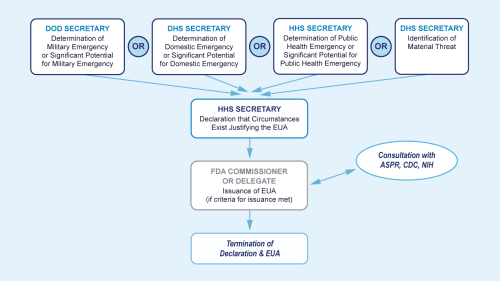
The flow chart above provides a summary of the process for Emergency Use Authorization (EUA) issuance.
Please note: A determination under section 319 of the Public Health Service Act that a public health emergency exists, such as the one issued on January 31, 2020, does not enable FDA to issue Emergency Use Authorizations. A separate determination and declaration are needed under section 564 of the Federal Food, Drug, and Cosmetic Act to enable FDA to issue Emergency Use Authorizations, provided other statutory criteria are met.
Description of chart:
Issuance of an EUA by the FDA Commissioner requires several steps under section 564 of the FD&C Act. First, one of the four following determinations must be in place:
- The Department of Defense (DoD) Secretary issues a determination of military emergency or significant potential for military emergency
- The Department of Homeland Security (DHS) Secretary issues a determination of domestic emergency or significant potential for domestic emergency.
- The Department of Health and Human Services (HHS) Secretary issues a determination of public health emergency or significant potential for public health emergency
- The DHS Secretary issues a material threat determination
After one of the above four determinations is in place, the HHS Secretary can issue a declaration that circumstances exist to justify issuing the EUA. This declaration is specific to EUAs and is not linked to other types of emergency declarations.
The FDA Commissioner or delegate, in consultation with the HHS Assistant Secretary for Preparedness and Response (ASPR), Centers for Disease Control and Prevention (CDC), and the National Institutes of Health (NIH), can then issue the EUA, if criteria for issuance under the statute are met. FDA publishes public notice of each EUA that is issued in the Federal Register.
The last step in the process is termination of declaration and EUA, if appropriate and needed.
Related Links
- Emergency Use Authorization
- FAQs: What happens to EUAs when a public health emergency ends?
- HHS Public Health Emergency EUA Authorization Declarations
- Pandemic and All-Hazards Preparedness Reauthorization Act of 2013 (PAHPRA)
- Public Readiness and Emergency Preparedness (PREP) Act
- What is an EUA? (YouTube video, 1:45)
- Emergency Use Authorization for Vaccines Explained
- The Path for a COVID-19 Vaccine from Research to Emergency Use Authorization (PDF, 723KB)

