FDA investigates two adverse events associated with United Pharmacy’s compounded glutamine, arginine, and carnitine product for injection
Updated: March 14, 2018
FDA laboratory analysis found the glutamine in United Pharmacy’s compounded glutamine, arginine, and carnitine (GAC) 10/100/200 mg/mL product for injection is degrading before the beyond-use date (BUD) of 6 months specified on the product label.
Previously, FDA reported the product was labeled as including 10 mg/mL of glutamine, but no glutamine was found in samples from lots GAC-12 or GAC-13 tested within expiry. Since that time, FDA tested two available samples of the glutamine active pharmaceutical ingredient (API) collected from the API supplier used by United Pharmacy and determined the average purity of the samples to be within the acceptable range at 98.1% and 98.6% of its labeled claim. FDA also further tested the United Pharmacy GAC-12 and GAC-13 samples to determine whether the glutamine degraded.
Glutamine is known to break down to ammonia and pyroglutamic acid in aqueous solutions3. FDA laboratory analysis detected the presence of pyroglutamic acid within the samples in amounts roughly equivalent to the degradation of approximately 10 mg/mL of glutamine. Specifically, FDA laboratory analysis showed that the sample from lot GAC-12 that was expired at the time of testing contained 9.28 mg/mL of pyroglutamic acid, which would be the expected degradant product and concentration from 10.51 mg/mL aqueous solution of glutamine. The sample from lot GAC-13 that was within expiry contained 8.91 mg/mL of pyroglutamic acid, which would be the expected degradant product and concentration from 10.09 mg/mL aqueous solution of glutamine. These data suggest that this formulation of glutamine was chemically unstable before the expiration date was reached.
United Pharmacy recalled all of its GAC 10/100/200 mg/mL for injection products and ceased production of this formulation.
FDA encourages compounders to perform stability studies for solutions containing glutamine to confirm that the glutamine is not degrading before the product’s specified BUD.
November 8, 2017
Summary
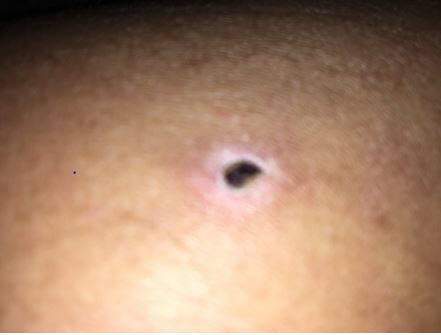
The U.S. Food and Drug Administration received an adverse event report stating that two patients developed what is described in the report as tissue erosion at the injection site following the administration of an injectable glutamine, arginine, and carnitine (GAC) product that was compounded by United Pharmacy, LLC, located in West Palm Beach, Florida. According to the report, a sample of the product was sent for testing and the pH was determined to be above 11. FDA conducted a for-cause inspection of United Pharmacy and collected samples from two batches of GAC injectable product. Analysis performed on the samples determined the pH to be 10.9, which is alkaline, and no glutamine was detected. Parenteral drugs, including compounded injectable drugs, with a high pH may lead to skin damage or other unintended health consequences.1
Adverse Events
On April 21, 2017, FDA became aware of adverse events in two patients. Both patients experienced what is described in the report as tissue erosion at the injection site following administration of GAC (see Fig.1). The report stated the GAC injectable product was compounded by United Pharmacy. According to the report, a vial of the product was sent for testing and the pH was determined to be above 11.
FDA’s Investigation and Findings
FDA conducted a for-cause inspection of United Pharmacy in response to the reported adverse events. During the inspection, FDA collected and analyzed samples of the GAC injectable product from two available lots: GAC-12 and GAC-13. The label for one of those samples states: “Inject 1 mL on an insulin syringe every other day in the morning subcutaneously.” FDA found the samples to have a pH of 10.9. In addition, United Pharmacy’s formula worksheets for GAC injectable products have an acceptance criteria for pH ranging from acidic (pH: 2) to basic
(pH: 11).
Further, FDA found the samples did not contain any amount of glutamine. The labels of the GAC injectable samples indicate that 10 mg/mL of glutamine, 100 mg/mL of arginine, and 200 mg/mL of carnitine are contained in 30 mL vials.
United Pharmacy recalled lots GAC-12 and GAC-13 on September 27, 2017.
Discussion
According to Broadhead and Gibson, injectable products formulated with a pH close to physiological pH, or approximately pH 7.4, may minimize pain, irritation, and tissue damage.1 Broadhead and Gibson note that most approved parenteral products have a pH in the range of 3 to 9, and that the recommended range for parenteral products intended for subcutaneous injection is 3 to 6.1 They also note that a pH greater than 9 may cause tissue necrosis.1 Additionally, Roethlisberger et al report that skin damage at the injection site after subcutaneous injection may be caused by physical stress (e.g., osmotic pressure) and chemical stress other than pH (e.g., solvent content).2 It is not clear whether these other factors may have contributed to what is described in the report as tissue erosion at the injection site.
Risk Summary
Compounded drugs have not been evaluated by FDA for safety, effectiveness, and quality. There is no FDA-approved injectable GAC product. When FDA reviews an application for an injectable drug, the overall safety of the product is evaluated, including, as relevant here, the suitability of the pH and the labeling of the product.
FDA’s investigation into the adverse events associated with United Pharmacy’s GAC product for injection highlights some of the risks associated with compounded drugs, which do not undergo FDA premarket review. The risks illustrated in this case include:
- the product specifications used by compounding facilities, such as pH, and the pH of the compounded drug are not evaluated by FDA for safety;
- the subcutaneous administration of an injectable product with an alkaline pH could result in skin damage or other unintended health consequences; and
- the ingredients listed on the labeling of a compounded product may not accurately reflect the ingredients contained within the product.
Compounding pharmacies should be aware of safety information related to the physical and chemical attributes of their injectable products, including pH. Because compounded products are not evaluated by FDA for safety and effectiveness, health care professionals should query the compounding pharmacy about the safety information related to the physical and chemical attributes of the compounded products they plan to inject into patients.
FDA encourages consumers, patients, and health care professionals to report adverse events or quality problems experienced with the use of compounded drug products to FDA’s MedWatch Adverse Event Reporting program:
- Complete and submit the report online at Medwatch; or
- Download and complete the form, then submit it via fax at 1-800-FDA-0178.
References
- Broadhead, J and Gibson M. 2009. Parenteral Dosage Forms. In Gibson, M. ed. Pharmaceutical Preformulation and Formulation: A Practical Guide from Candidate Drug Selection to Commercial Dosage Form. Boca Raton, FL: CRC Press LLC; pp.326-327.
- Roethlisberger D, Mahler H-C, Altenburger U, and Pappenberger A. If Euhydric and Isotonic Do Not Work, What Are Acceptable pH and Osmolality for Parenteral Drug Dosage Forms? J Pharm Sci. 2017;106: 446-456.
- Khan K, Elia M. Factors affecting the stability of L-glutamine in solution. Clin Nutr 1991;10(4):186-192.