Drug Trials Snapshots: BAVENCIO
HOW TO USE THIS SNAPSHOT
The information provided in Snapshots highlights who participated in the clinical trials that supported the FDA approval of this drug, and whether there were differences among sex, race, and age groups. The “MORE INFO” bar shows more detailed, technical content for each section. The Snapshot is intended as one tool for consumers to use when discussing the risks and benefits of the drugs.
LIMITATIONS OF THIS SNAPSHOT:
Do not rely on Snapshots to make decisions regarding medical care. Always speak to your health provider about the risks and benefits of a drug. Refer to BAVENCIO Prescribing Information for complete information.
BAVENCIO (avelumab)
buh-VEN-see-oh
EMD Serono, Inc.
Approval date: March 23, 2017
DRUG TRIALS SNAPSHOT SUMMARY:
What is the drug for?
BAVENCIO is used to treat a rare type of skin cancer called Merkel cell carcinoma (MCC) in adults and in pediatric patients 12 years and older. It is used in patients for whom the cancer has spread (metastatic).
BAVENCIO was approved under FDA’s accelerated approval program, which provides earlier patient access to a promising new drug while the company continues to conduct clinical trials to confirm that the drug works well.
How is this drug used?
BAVENCIO is given by a healthcare provider using a needle placed in a vein (known as intravenous infusion) over 60 minutes. BAVENCIO is given every 2 weeks.
What are the benefits of this drug?
Thirty-three percent of 88 patients who were treated with BAVENCIO experienced complete or partial shrinkage of their tumors. The majority of responding patients maintained their response for more than 6 months and nearly half (45 percent) maintained their response for more than 12 months.
More trials are ongoing to confirm the clinical benefit of BAVENCIO.
What are the benefits of this drug (results of trials used to assess efficacy)?
The table below summarizes efficacy results based on overall response rate (ORR) and duration of response according to Response Evaluation Criteria in Solid Tumors (RECIST) v1.1 as measured by blinded independent central review.
Table 2. Efficacy Results of Trial 1
| Efficacy Endpoints | Results (N=88) |
|---|---|
| Overall Response Rate (ORR) Overall response rate, (95% CI) Complete response (CR) rate, (95% CI) Partial response (PR) rate, (95% CI) | 33.0% (23.3%, 43.8%) 11.4% (6.6%, 19.9%) 21.6% (13.5%, 31.7%) |
| Duration of Response (DOR) Range in months Patients with DOR ≥ 6 months, n (%) Patients with DOR ≥ 12 months, n (%) | N=29 2.8 to 23.3+ 25 (86%) 13 (45%) |
CI: Confidence interval
BAVENCIO Prescribing Information
Were there any differences in how well the drug worked in clinical trials among sex, race and age?
The trial that looked at the benefit of BAVENCIO was too small to determine if there were any differences in sex, race and age subgroups.
Were there any differences in how well the drug worked in clinical trials among sex, race, and age groups?
The table below summarizes efficacy results of Overall Response Rate (ORR) by sex and age. Racial subgroup differences were not investigated since the majority of patients were white.
Due to the small sample size, these exploratory analyses should be interpreted with caution.
Table 3. Subgroup Analysis of Overall Response Rate (ORR) As Assessed by the Investigator
| ORR n (%) | 95% CI* | |
|---|---|---|
| Sex | ||
| Men ( N=65) | 22 (33.8) | 21.2, 45.1 |
| Women (N=23) | 7 (30.4) | 13.2, 52.9 |
| Age Group | ||
| 65 years="" (n=""> | 8(36.3) | 13.9, 54.9 |
| ≥65 years (N=66) | 21 (31.8) | 20.9, 44.4 |
*95% Exact Confidence Interval using Clopper-Pearson method
Adapted from Clinical Trial Report
What are the possible side effects?
BAVENCIO can cause the immune system to attack normal organs and tissues which may lead to serious side effects in any part of the body. The most common parts of the body that are affected are the lungs, liver, colon, kidneys, and endocrine glands. BAVENCIO may also cause serious infusion reactions (low blood pressure, difficulty breathing) when it is being given into the vein. All patients should receive medicines before the first four doses of BAVENCIO to help reduce infusion reactions.
Common side effects of BAVENCIO are tiredness, muscle and bone pain, diarrhea, nausea, infusion related reaction, skin rash, decreased appetite, and swelling of the limbs.
What are the possible side effects (results of trials used to assess safety)?
The tables below summarize adverse reactions and electrolyte abnormalities in the clinical trial.
Table 4. Adverse Reactions in ≥ 10% of Patients with Metastatic MCC Receiving BAVENCIO
| Adverse Reactions | BAVENCIO (N=88) | |
|---|---|---|
| All Grades % | Grade 3-4 % | |
| General Disorders | ||
| Fatiguea | 50 | 2 |
| Infusion‑related reactionb | 22 | 0 |
| Peripheral edemac | 20 | 0 |
| Musculoskeletal and Connective Tissue Disorders | ||
| Musculoskeletal paind | 32 | 2 |
| Arthralgia | 16 | 1 |
| Gastrointestinal Disorders | ||
| Diarrhea | 23 | 0 |
| Nausea | 22 | 0 |
| Constipation | 17 | 1 |
| Abdominal paine | 16 | 2 |
| Vomiting | 13 | 0 |
| Skin and Subcutaneous Tissue Disorders | ||
| Rashf | 22 | 0 |
| Pruritusg | 10 | 0 |
| Metabolism and Nutrition Disorders | ||
| Decreased appetite | 20 | 2 |
| Decreased weight | 15 | 0 |
| Respiratory, Thoracic and Mediastinal Disorders | ||
| Cough | 18 | 0 |
| Dyspneah | 11 | 0 |
| Nervous System Disorders | ||
| Dizziness | 14 | 0 |
| Headache | 10 | 0 |
| Vascular Disorders | ||
| Hypertension | 13 | 6 |
aFatigue is a composite term that includes fatigue and asthenia
bInfusion-related reaction is a composite term that includes drug hypersensitivity, hypersensitivity, chills, pyrexia, back pain, and hypotension
cPeripheral edema is a composite term that includes peripheral edema and peripheral swelling
dMusculoskeletal pain is a composite term that includes back pain, myalgia, neck pain, pain in extremity
eAbdominal pain is a composite term that includes abdominal pain and abdominal pain upper
fRash is a composite term that includes rash maculo-papular, erythema, and dermatitis bullous
gPruritus is a composite term that includes pruritus and pruritus generalized
hDyspnea is a composite term that includes dyspnea and dyspnea exertional
BAVENCIO Prescribing Information
Table 5. Selected Treatment Emergent *Laboratory Abnormalities in Patients Receiving BAVENCIO
| Laboratory Tests | Any Grade (N=88) % | Grade 3-4 (N=88) % | |
|---|---|---|---|
| Chemistry | |||
| Increased aspartate aminotransferase (AST) | 34 | 1 | |
| Increased alanine aminotransferase (ALT) | 20 | 5 | |
| Increased lipase | 14 | 4 | |
| Increased amylase | 8 | 1 | |
| Increased bilirubin | 6 | 1 | |
| Hyperglycemia** | - | 7 | |
| Hematology | |||
| Anemia | 35 | 9 | |
| Lymphopenia | 49 | 19 | |
| Thrombocytopenia | 27 | 1 | |
| Neutropenia | 6 | 1 | |
* Treatment emergent consists of new onset of laboratory abnormality or worsening of baseline laboratory abnormality
** Hyperglycemia limited to Grade ≥ 3 events since fasting measurements were not obtained routinely
BAVENCIO Prescribing Information
Were there any differences in side effects among sex, race and age?
The trial that looked at the side effects of BAVENCIO was too small to determine if there were any differences in sex, race and age subgroups.
Were there any differences in side effects of the clinical trials among sex, race, and age groups?
The table below summarizes adverse events during the clinical trial by sex, and age subgroup. Racial subgroup differences were not investigated since the majority of patients were white.
Due to the small sample size, these exploratory analyses should be interpreted with caution.
Table 6. Subgroup Analysis of Adverse Events by Sex
| Men N=65 n (%) | Women N=23 n (%) | |
|---|---|---|
| Any grade event | 63 (97) | 23 (100) |
| Grade ≥3 event | 40 (62) | 14 (63) |
Adapted from FDA Clinical review
Table 7. Subgroup Analysis of Adverse Events by Age
| Age 65=""> N = 22 n (%) | Age ≥65 to 75> N = 35 n (%) | Age ≥75 Years N = 31 n (%) | |
|---|---|---|---|
| Any grade event | 22 (100%) | 35 (100%) | 29 (94%) |
| Grade ≥3 event | 14 (64%) | 24 (67%) | 16 (52%) |
Adapted from FDA Clinical review
WHO WAS IN THE CLINICAL TRIALS?
Who participated in the clinical trials?
The FDA approved BAVENCIO based on evidence from one clinical trial of 88 patients with metastatic MCC and whose tumor was no longer responding to chemotherapy.
The trial was conducted in North America, Western Europe, Australia and Asia.
Figure 1 summarizes how many men and women were enrolled in the clinical trial.
Figure 1. Baseline Demographics by Sex
Clinical trial data
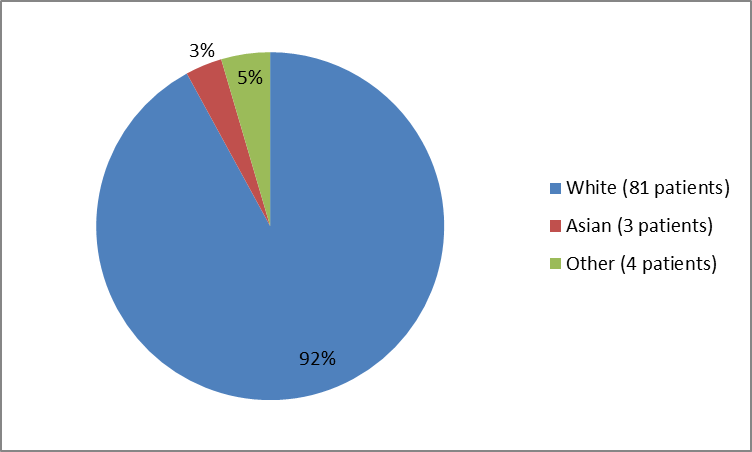
Figure 2 and Table 1 below summarize the percentage of patients by race in the clinical trial.
Figure 2. Baseline Demographics by Race
Clinical trial data
Table 1. Baseline Demographics by Race
| Race | Number of Patients | Percentage |
|---|---|---|
| White | 81 | 92 |
| Asian | 3 | 3 |
| Not collected at the site | 3 | 3 |
| Unknown | 1 | 1 |
Clinical trial data
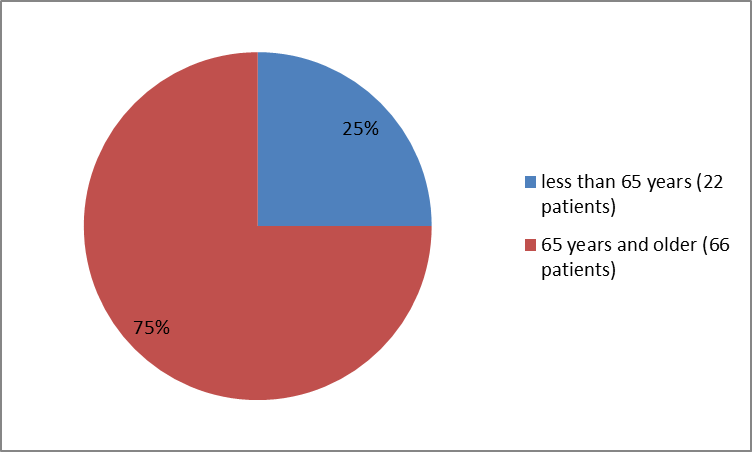
Figure 3 summarizes how many patients of certain age were enrolled in the clinical trial.
Figure 3. Baseline Demographics by Age
Clinical trial data
Who participated in the trials?
The table below summarizes demographics of patients in the clinical trial.
Table 8. Baseline Demographics of Patients in the Clinical Trial
| Demographic Parameters | BAVENCIO N=88 n (%) |
|---|---|
| Sex | |
| Men | 65(74) |
| Women | 23(26) |
| Age | |
| Median (Range) | 73 (33, 88) |
| Age Group | |
| 65> | 22(25) |
| ≥65 years | 66(75) |
| Race | |
| White | 81 (92) |
| Asian | 3 (3) |
| Not collected at the site | 3 (3) |
| Unknown | 1 (1) |
| Region | |
| North America | 51 (58) |
| Western Europe | 29 (33) |
| Australia | 5 (6) |
| Asia | 3 (3) |
FDA review
How were the trials designed?
The benefit and side effects of BAVENCIO were evaluated in one clinical trial of patients with metastatic MCC. All patients received BAVENCIO once every two weeks until either the tumors grew or the patients developed an unacceptable side effect.
The benefit of BAVENCIO was evaluated by measuring how many patients had tumor shrinkage (response) and by how long that response lasted.
How were the trials designed?
There was one single-arm, open-label, multicenter clinical trial that enrolled patients with histologically confirmed metastatic MCC whose disease had progressed (grown larger) on or after chemotherapy administered for metastatic MCC. Patients received BAVENCIO 10 mg/kg as an intravenous infusion over 60 minutes every 2 weeks until disease progression or unacceptable toxicity.
The efficacy outcome measures were confirmed overall response rate (ORR) and duration of response, according to Response Evaluation Criteria in Solid Tumors (RECIST) v1.1 as measured by blinded independent central review and duration of response.
GLOSSARY
CLINICAL TRIAL: Voluntary research studies conducted in people and designed to answer specific questions about the safety or effectiveness of drugs, vaccines, other therapies, or new ways of using existing treatments.
COMPARATOR: A previously available treatment or placebo used in clinical trials that is compared to the actual drug being tested.
EFFICACY: How well the drug achieves the desired response when it is taken as described in a controlled clinical setting, such as during a clinical trial.
PLACEBO: An inactive substance or “sugar pill” that looks the same as, and is given the same way as, an active drug or treatment being tested. The effects of the active drug or treatment are compared to the effects of the placebo.
SUBGROUP: A subset of the population studied in a clinical trial. Demographic subsets include sex, race, and age groups.