Drug Trials Snapshot: FETROJA
HOW TO USE THIS SNAPSHOT
The information provided in Snapshots highlights who participated in the clinical trials that support the FDA approval of this drug, and whether there were differences among sex, race and age groups. The “MORE INFO” bar shows more detailed, technical content for each section. The Snapshot is intended as one tool for consumers to use when discussing the risks and benefits of the drugs.
LIMITATIONS OF THIS SNAPSHOT:
Do not rely on Snapshots to make decisions regarding medical care. Always speak to your health provider about the risks and benefits of a drug. Refer to the FETROJA Prescribing Information for complete information.
FETROJA (cefiderocol)
fet roe' jah
Shionogi Inc.
Approval date: November 14, 2018
DRUG TRIALS SNAPSHOT SUMMARY:
What is the drug for?
FETROJA is used to treat adults who have a complicated urinary tract infection (abbreviated as cUTI) including infection of the kidneys (pyelonephritis) caused by specific bacteria. It should be used only when few or no other treatment options are available.
How is this drug used?
FETROJA is a drug administered by a health care professional directly into the bloodstream through a needle in the vein. This is known as an intravenous, or IV, infusion. It is given every 8 hours.
What are the benefits of this drug?
At the end of the treatment with FETROJA, 73% of patients had resolution of cUTI symptoms and had decreased the number of bacteria in their urine. In comparison, 55% of patients who received another antibacterial drug, called imipenem/cilastatin achieved the same results.
What are the benefits of this drug (results of trials used to assess efficacy)?
The efficacy of FETROJA was established based on a composite of microbiological eradication (all Gram-negative uropathogens found at baseline at ≥ 105 CFU/mL reduced to < 104 CFU/mL) and clinical response (resolution or improvement of cUTI symptoms and no new symptoms assessed by the investigator) at the TOC visit 7 +/- 2 days after the last dose of trial drug. Efficacy was assessed in the microbiological modified intent to treat population (micro-MITT), which was a subset of all randomized patients who received any dose of the trial drug and had at least one Gram-negative uropathogen.
Table 2. Composite, Microbiological, and Clinical Response Rates at the TOC Visit (micro ITT Population)
| Trial Endpoint | FETROJA N/n (%) |
Imipenem/Cilastatin N/n (%) |
Treatment Difference (95% CI)a |
|---|---|---|---|
| Composite response at TOC | 183/252 (72.6%) | 65/119 (54.6%) | 18.6 (8.2, 28.9) |
| Micro response TOC | 184/252 (73.0%) | 67/119 (56.3%) | 17.3 (6.9, 27.6) |
| Clinical response TOC | 226/252 (89.7%) | 104/119 (87.4%) | 2.4 (-4.7, 9.4) |
CI = confidence interval; Micro-ITT = microbiological intent-to-treat; TOC = Test of Cure
aCI=95% confidence interval based on the Cochran-Mantel-Haenzel method.
FETROJA Prescribing Information
Were there any differences in how well the drug worked in clinical trials among sex, race and age?
- Sex: FETROJA worked similarly in men and women.
- Race: Majority of patients were White. The number of patients in other races was limited. Differences in how well the drug worked among races could not be determined.
- Age: FETROJA worked similarly in patients below and above 65 years of age.
Were there any differences in how well the drug worked in clinical trials among sex, race, and age groups?
Subgroup analyses for composite endpoint (microbiological eradication and clinical response) are presented in the table below.
Table 3. Subgroup Analyses for the Composite Response at TOC, micro-MITT Population
| FETROJA n/N (%) |
Imipenem/Cilastatin n/N (%) |
Difference (%) | 95% CI | |
|---|---|---|---|---|
| Sex | ||||
| Men | 84/119 (70.6) |
25/48 (52.1) |
18.5 | 2.2 to 34.8 |
| Women | 99/133 (74.4) |
40/71 (56.3) |
18.1 | 4.4 to 31.8 |
| Race | ||||
| White | 175/241 (72.6) |
64/115 (55.7) |
17.0 | 6.3 to 27.7 |
| Black or African American |
0/1 (0.0) |
0/0 | - | - |
| Asian | 8/9 (88.9) |
1/4 (25.0) |
63.9 | - |
| Native Hawaiian or Other Pacific Islander |
0/1 (0.0) | 0/0 | - | - |
| Age group | ||||
| <65 years | 87/113 (77.0) |
32/54 (59.3) |
17.7 | 2.5 to 33.0 |
| ≥65 years | 96/139 (69.1) |
33/65 (50.8) |
18.3 | 3.9 to 32.7 |
Adapted from FDA Review
What are the possible side effects?
An increased risk of dying was observed in patients treated with FETROJA as compared to other antibiotics in a separate clinical trial. Most of the deaths occurred in patients hospitalized with other kinds of severe bacterial infections (pneumonia, sepsis, or infection in the blood). The cause of death was not clear.
FETROJA may cause serious and life-threatening allergic reactions, severe diarrhea caused by C. difficile and seizures.
Common side effects that were associated with the use of FETROJA include diarrhea, infusion site reactions, constipation and rash.
What are the possible side effects (results of trials used to assess safety)?
Table 4 summarizes the most common adverse events observed in the trial.
Table 4. Selected Adverse Reactions Occurring in >2% of Patients Receiving FETROJA in the Trial
| Adverse Reaction | FETROJAa (N =300) |
Imipenem/Cilastatinb (N = 148) |
|---|---|---|
| Diarrhea | 4% | 6% |
| Infusion site reactionsc | 4% | 5% |
| Constipation | 3% | 4% |
| Rashd | 3% | <1% |
| Candidiasise | 2% | 3% |
| Cough | 2% | <1% |
| Elevations in liver testsf | 2% | <1% |
| Headache | 2% | 5% |
| Hypokalemiag | 2% | 3% |
| Nausea | 2% | 4% |
| Vomiting | 2% | 1% |
cUTI = complicated urinary tract infections
a 2 grams IV over 1 hour every 8 hours (with dosing adjustment based on renal function)
b 1 gram IV over 1 hour every 8 hours (with dosing adjustment based on renal function and body weight)
c Infusion site reactions include infusion site erythema, inflammation, pain, pruritis, injection site pain, and phlebitis.
d Rash includes rash macular, rash maculopapular, erythema, skin irritation.
e Candidiasis includes oral or vulvovaginal candidiasis, candiduria.
f Elevations in liver tests include alanine aminotransferase, aspartate aminotransferase, gamma-glutamyl transferase, blood alkaline phosphatase, hepatic enzyme increased.
g Hypokalemia includes blood potassium decreased.
FETROJA Prescribing Information
Were there any differences in side effects among sex, race and age?
- Sex: The occurrence of side effects was similar between men and women.
- Race: Majority of patients were White. The number of patients in other races was limited. Differences in side effects among races could not be determined.
- Age: The occurrence of side effects was similar between patients younger and older than 65 years of age.
Were there any differences in side effects of the clinical trials among sex, race, and age groups?
The table below summarizes the incidence of treatment-emergent adverse events (TEAEs) by subgroups.
Table 5. Summary of Incidence of TEAEs by Subgroup (Safety Population)
| FETROJA | Imipenem/cilastatin | |||
|---|---|---|---|---|
| Subgroup | n (%) | N | n (%) | N |
| Any TEAE | 122 (40.7) | 300 | 76 (51.4) | 148 |
| Sex | ||||
| Women | 76 (46.6) | 163 | 49 (59.8) | 82 |
| Men | 46 (33.6) | 137 | 27 (40.9) | 66 |
| Race | ||||
| Asian | 8 (72.7) | 11 | 4 (100.0) | 4 |
| Black or African American | 1 (100.0) | 1 | 1 (100.0) | 1 |
| Native Hawaiian or other Pacific Islander |
1 (100.0) | 1 | 1 (100.0) | 1 |
| White | 112 (39.0) | 287 | 70 (49.3) | 142 |
| Age Group | ||||
| < 65 years | 62 (43.7) | 142 | 31 (44.3) | 70 |
| ≥65 years | 60 (38.0) | 158 | 45 (57.7) | 78 |
| Ethnicity | ||||
| Hispanic or Latino | 3 (60.0) | 5 | 1 (50.0) | 2 |
| Not Hispanic or Latino | 119 (40.3) | 295 | 75 (51.4) | 146 |
| Region | ||||
| Asia | 9 (36.0) | 25 | 5 (50.0) | 10 |
| Europe | 108 (40.0) | 270 | 69 (50.7) | 136 |
| United States | 5 (100.0) | 5 | 2 (100.0) | 2 |
FDA Review
WHO WAS IN THE CLINICAL TRIALS?
Who participated in the clinical trials?
FDA approved FETROJA based on the trial (NCT02321800) of 448 patients with cUTI. The trial included patients from Europe, United States and Mexico.
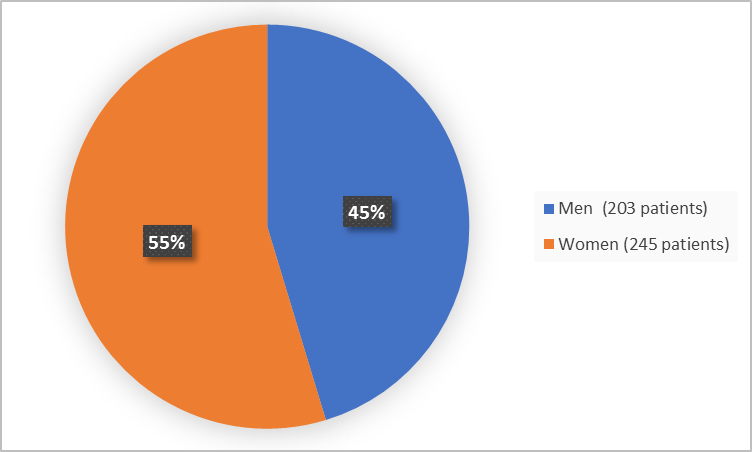
Figure 1 summarizes how many men and women were in the clinical trial.
Figure 1. Baseline Demographics by Sex
FDA Review
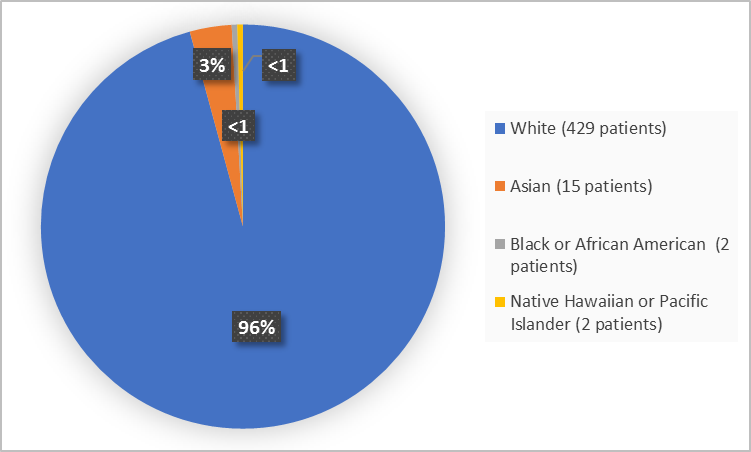
Figure 2 and Table 1 summarize the percentage of patients by race in the clinical trial.
Figure 2. Baseline Demographics by Race
FDA Review
Table 1. Baseline Demographics by Race
| Number of Patients | Percentage (%) | |
|---|---|---|
| White | 429 | 96 |
| Asian | 15 | 3 |
| Black or African American | 2 | Less than 1 |
| Native Hawaiian or Pacific Islander | 2 | Less than 1 |
FDA Review
Figure 3 summarizes by age how many patients were in the clinical trial.
Figure 3. Baseline Demographics by Age
FDA Review
Who participated in the trials?
The table below summarizes demographic information for the safety population.
Table 6. Demographic Characteristics (Safety Population)
| Demographic subgroup | FETROJA N = 300 n (%) |
Imipenem/cilastatin N = 148 n (%) |
TOTAL N = 448 n (%) |
|---|---|---|---|
| Sex | |||
| Women | 163 (54.3) | 82 (55.4) | 245 (54.7) |
| Men | 137 (45.7) | 66 (44.6) | 203 (45.3) |
| Race | |||
| White | 287 (95.7) | 142 (95.9) | 429 (95.8) |
| Asian | 11 (3.7) | 4 (2.7) | 15 (3.3) |
| Black or African American | 1 (0.3) | 1 (0.7) | 2 (0.4) |
| Native Hawaiian/Pacific Islander | 1 (0.3) | 1 (0.7) | 2 (0.4) |
| Age | |||
| Mean | 61.1 | 61.3 | 61.2 |
| Standard Deviation | 16.54 | 17.76 | 16.93 |
| Median | 65.0 | 65.0 | 65.0 |
| Age Group | |||
| < 65 years | 142 (47.3) | 70 (47.3) | 212 (47.3) |
| ≥ 65 years | 158 (52.7) | 78 (52.7) | 236 (52.7) |
| Ethnicity | |||
| Not Hispanic or Latino | 295 (98.3) | 146 (98.6) | 441 (98.4) |
| Hispanic or Latino | 5 (1.7) | 2 (1.4) | 7 (1.6) |
| Region | |||
| Europe | 270 (90.0) | 136 (91.9) | 406 (90.6) |
| Asia | 25 (8.3) | 10 (6.8) | 35 (7.8) |
| United States | 5 (1.7) | 2 (1.4) | 7 (1.6) |
FDA Review
How were the trials designed?
In the clinical trial, patients with cUTI were chosen at random to receive FETROJA, or another antibacterial drug called imipenem/cilastatin. Both treatments were given intravenously for 7-14 days and neither the patients nor the health care professionals knew which drugs were given until after the trial was complete. Patients could not be switched to an oral antibacterial drug to complete the treatment for cUTI.
The benefit of FETROJA was measured by the proportion of patients who achieved cure or improvement in their symptoms related to cUTI and a negative urine culture test in comparison to imipenem/cilastatin.
How were the trials designed?
There was one randomized, active-controlled, double blind, non-inferiority, multi center trial comparing FETROJA to imipenem/cilastatin in the treatment of adults with cUTI. Medications were given intravenously for 7 to 14 days. No switch from IV to oral antibacterial therapy was permitted.
The efficacy of FETROJA was based on non-inferiority assessment of the primary-endpoint defined as a composite of microbiological eradication (all Gram-negative uropathogens found at baseline at ≥ 105 CFU/mL reduced to < 104 CFU/mL) and clinical response (resolution or improvement of cUTI symptoms and no new symptoms assessed by the investigator) at the TOC visit 7 +/- 2 days after the last dose of study drug.
GLOSSARY
CLINICAL TRIAL: Voluntary research studies conducted in people and designed to answer specific questions about the safety or effectiveness of drugs, vaccines, other therapies, or new ways of using existing treatments.
COMPARATOR: A previously available treatment or placebo used in clinical trials that is compared to the actual drug being tested.
EFFICACY: How well the drug achieves the desired response when it is taken as described in a controlled clinical setting, such as during a clinical trial.
PLACEBO: An inactive substance or “sugar pill” that looks the same as, and is given the same way as, an active drug or treatment being tested. The effects of the active drug or treatment are compared to the effects of the placebo.
SUBGROUP: A subset of the population studied in a clinical trial. Demographic subsets include sex, race, and age groups.