Xenotransplantation
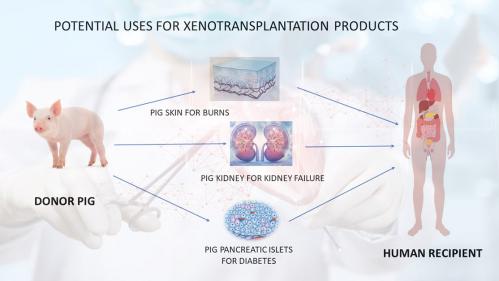
Xenotransplantation is any procedure that involves the transplantation, implantation or infusion into a human recipient of either (a) live cells, tissues, or organs from a nonhuman animal source, or (b) human body fluids, cells, tissues or organs that have had ex vivo contact with live nonhuman animal cells, tissues or organs. The development of xenotransplantation is, in part, driven by the fact that the demand for human organs for clinical transplantation far exceeds the supply.
Currently ten patients die each day in the United States while on the waiting list to receive lifesaving vital organ transplants. Moreover, recent evidence has suggested that transplantation of cells and tissues may be therapeutic for certain diseases such as neurodegenerative disorders and diabetes, where, again human materials are not usually available.
Although the potential benefits are considerable, the use of xenotransplantation raises concerns regarding the potential infection of recipients with both recognized and unrecognized infectious agents and the possible subsequent transmission to their close contacts and into the general human population. Of public health concern is the potential for cross-species infection by retroviruses, which may be latent and lead to disease years after infection. Moreover, new infectious agents may not be readily identifiable with current techniques.
Related Information
- Information and Recommendations for Physicians Involved in the Co-Culture of Human Embryos with Non-Human Animal Cells
- Human Cells or Tissues Intended for Transplant into a Human Recipient that Have Ex-vivo Contact with Live Non-human Animal Cells, Tissues or Organs Letter - ARCHIVED
- Xenotransplantation Guidances
- Human Organ and Tissue Transplantation
World Health Organization International Xenotransplantation Information
Follow CBER
Division of Communication and Consumer Affairs
Office of Communication, Outreach and Development
Center for Biologics Evaluation and Research
Food and Drug Administration
10903 New Hampshire Ave WO71-3103
Silver Spring, MD 20993-0002
(800) 835-4709
(240) 402-8010
For Updates on Twitter, follow @fdacber

