COMPANY ANNOUNCEMENT
Lupin Pharmaceuticals Inc. Announces a Nationwide Recall of Mibelas™ 24 Fe (Norethindrone Acetate and EthinylEstradiol 1mg/0.02mg Chewable Tablets and Ferrous Fumarate 75 mg) Tablets Due to Out of Sequence Tablets and Missing Expiry/Lot Information
This recall has been completed and FDA has terminated this recall.
When a company announces a recall, market withdrawal, or safety alert, the FDA posts the company's announcement as a public service. FDA does not endorse either the product or the company.
Read Announcement View Product PhotosSummary
- Company Announcement Date:
- FDA Publish Date:
- Product Type:
- Drugs
Generic Drugs - Reason for Announcement:
-
Recall Reason DescriptionMissing Expiry
- Company Name:
- Lupin Pharmaceuticals Inc.
- Brand Name:
-
Brand Name(s)Lupin
- Product Description:
-
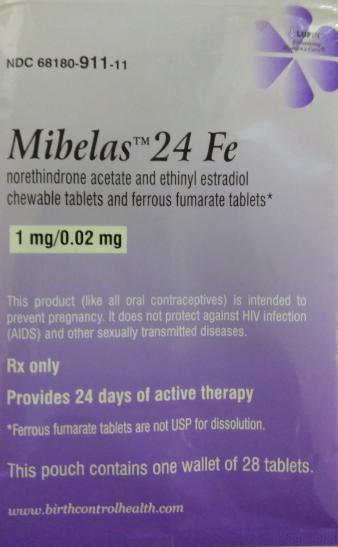
Product DescriptionMibelas 24 Fe (Norethindrone Acetate and Ethinyl Estradiol 1 mg/0.02 mg chewable and ferrous fumarate 75 mg)
Company Announcement
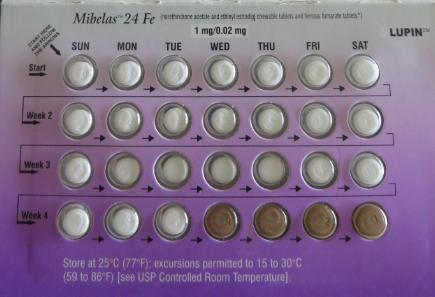
Baltimore, Maryland, Lupin Pharmaceuticals Inc. announced today that it has voluntarily recalled lot L600518, Exp 05/18 of Mibelas 24 Fe (Norethindrone Acetate and Ethinyl Estradiol 1 mg/0.02 mg chewable and ferrous fumarate 75 mg) Tablets at the consumer level. A confirmed market complaint indicated a packaging error, where the blister was rotated 180 degrees within the wallet, reversing the weekly tablet orientation and making the lot number and expiration date no longer visible. The first four days of therapy would have had four non-hormonal placebo tablets as opposed to the active tablets.
As a result of this packaging error, oral contraceptive tablets that are taken out of sequence may place the user at risk for contraceptive failure and unintended pregnancy. The reversing the order may not be apparent to either new users or previous users of the product, increasing the likelihood of taking the tablets out of order. For patients in whom a pregnancy is contraindicated or in whom concomitant medication(s) may have teratogenic effects, an unintended pregnancy may cause significant adverse maternal or fetal health consequences, including death.To date there have been no reports of such adverse events.
This product is an oral contraceptive indicated for the prevention of pregnancy in women who elect to use oral contraceptives. These products are packaged in blister packs containing 28 tablets: 24 white to off-white tablets of active ingredients debossed with “LU” on one side and “N81” on the other; and 4 tablets of inert ingredients debossed with “LU” on one side and “M22” on the other side. This product was distributed Nationwide in the U.S.A. to wholesalers, clinics and retail pharmacies.
| NDC | Lot No | Expiration |
|---|---|---|
| 68180-911-11 (Wallet of 28 tablets) 68180-911-13 (Carton of 3 wallets) |
L600518 | 05/31/2018 |
Lupin is notifying its distributors and customers by recall letter and is arranging for return of all recalled products. Consumers who have the affected product should notify their physician and return the product to the pharmacy or place of purchase.
Consumers with questions regarding this recall can contact Lupin by phone 1-800-399-2561, 8:00 am to 5:00 pm EST, Monday through Friday. Consumers should contact their physician or healthcare provider if they have experienced any problems that may be related to taking this drug product.
Adverse events or quality problems experienced with the use of this product may be reported to the FDA's MedWatch Adverse Event Reporting Program either online, by regular mail or by fax.
- Complete and submit the report Online: www.fda.gov/medwatch/report.htm
- Regular Mail or Fax: Download form www.fda.gov/MedWatch/getforms.htm or call 1-800-332-1088 to request a reporting form, then complete and return to the address on the pre-addressed form, or submit by fax to 1-800-FDA-0178.
This recall is being conducted with the knowledge of the U.S. Food and Drug Administration.
Company Contact Information
- Consumers:
- 1-800-399-2561