COMPANY ANNOUNCEMENT
APS Biogroup Recalls Viralox, DPS Throat Spray, Immulox, and Immuno PRP Spray, Because of Possible Health Risk
This recall has been completed and FDA has terminated this recall.
When a company announces a recall, market withdrawal, or safety alert, the FDA posts the company's announcement as a public service. FDA does not endorse either the product or the company.
Read Announcement View Product PhotosSummary
- Company Announcement Date:
- FDA Publish Date:
- Product Type:
- Dietary Supplements
- Reason for Announcement:
-
Recall Reason DescriptionStenotrophomonas maltophilia
- Company Name:
- APS BioGroup, Inc.
- Brand Name:
-
Brand Name(s)NuMedica & Sovereign Laboratories & Pure Research Products LLC & nutrasumma
- Product Description:
-
Product DescriptionDietary supplement
Company Announcement
APS BioGroup, Inc., of Phoenix, Arizona is recalling the following products:
| Product Name | UPC # | Expiration Date |
|---|---|---|
| Sovereign Laboratories Viralox | 8 63784 00015 0 | 04/2021 |
| Pure Research Products DPS Throat Spray |
8 55602 00021 3 | 04/2021 |
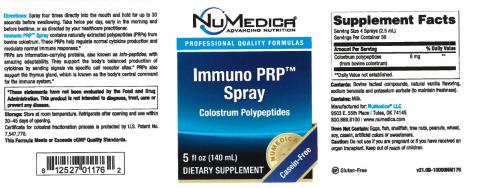
| NuMedica Immuno PRP Spray | 8 12527 01176 2 | 04/2021 |
| Nutrasumma Immulox | 1 82863 000064 6 | 04/2021 |
These products are being recalled because they have the potential to be contaminated with Stenotrophomonas maltophilia. Stenotrophomonas maltophilia is a gram-negative bacteria that can cause respiratory infection, particularly in immunocompromised individuals.
The recalled products are dietary supplements for supporting immune health and were distributed nationwide and in Switzerland. The products were sold online, including on Amazon, direct-to- consumer, and to the following retail establishments: Sprouts #205 – Claremont, CA; Sprouts #020 – Tempe, AZ; and Healthy Habit Health Foods, Phoenix, AZ.
The products come in 5 ounce plastic spray bottles marked with lot # 9068. The labels associated with each product are attached.
No illnesses have been reported to date in connection with this problem.
The potential for contamination was discovered after routine testing by the company detected elevated Aerobic Plate Counts in samples of the product that had been retained by APS BioGroup. After completing additional testing, APS BioGroup determined that the products described above may be contaminated with Stenotrophomonas maltophilia
Consumers who have purchased the affected product are encouraged to immediately stop using the product. Consumers with questions may contact the company identified on the label to return the product and for an immediate refund. You may also contact APS BioGroup at 1-720-443-5030 from 9:00AM – 5:00PM MDT, Monday through Friday, for additional information.
Company Contact Information
- Consumers:
- APS BioGroup
- 1-720-443-5030