GUIDANCE DOCUMENT
Immunotoxicity Testing Guidance May 1999
- Docket Number:
- FDA-2020-D-0957
- Issued by:
-
Guidance Issuing OfficeCenter for Devices and Radiological Health
Document issued on: May 6, 1999
U.S. Department of Health and Human Services
Food and Drug Administration
Center for Devices and Radiological Health
Molecular Biology Branch
Division of Life Sciences
Office of Science and Technology
Preface
Public Comment
Comments and suggestions may be submitted at any time for Agency consideration to, John J. Langone, Ph.D., HFZ-113, 12709 Twinbrook Parkway, Rockville, Maryland 20852. Comments may not be acted upon by the Agency until the document is next revised or updated. For questions regarding the use or interpretation of this guidance contact John J. Langone, Ph.D., at 301-443-2911, or by electronic mail at jzl@cdrh.fda.gov.
Additional Copies
Additional copies are available from the Internet. You may also send an e-mail request to CDRH-Guidance@fda.hhs.gov to receive a copy of the guidance. Please use the document number 635 to identify the guidance you are requesting.
Immunotoxicity Testing Guidance 1
Background
In May 1995, the Office of Device Evaluation in the Center for Devices and Radiological Health adopted General Program Memorandum G95-1. This guidance is a FDA-modified version of International Standard ISO-10993, "Biological Evaluation of Medical Devices-Part 1: Evaluation and Testing". It provides an overview of the general types of toxicity testing that should be considered for a medical device or constituent materials. At the time G95-1 was adopted, it was apparent that additional testing guidance might be needed for evaluation of individual organ or system toxicity. As a result, the framework in this document has been developed to focus specifically on immunotoxicity testing. It should be used in conjunction with the larger context of G95-1, as part of the overall evaluation of product safety.
This guidance provides assessment of the types of testing currently available for evaluating potential adverse effects of biomaterials on the immune system. It also provides a process for selecting appropriate test methods. The goal is to obtain adequate information to help make confident regulatory decisions, not to establish claims that a device or material is not immunotoxic. Evidence supporting non-immunotoxicity will not establish safety, but should provide some level of assurance that serious immunotoxic reactions are unlikely.
The framework in this guidance is intended to be a practical tool for selecting the best tests, based on our current knowledge in the field. It strongly recommends certain standardized and other commonly used reliable tests (see Table 3) because of the added quality assurance they provide for regulatory purposes. However, as a guidance, it is limited to making recommendations. It does not impose requirements on FDA or regulated industry.
Additional predictive immunotoxicity tests are needed. As these methods become available, for example in the form of consensus standards, they also will be recommended in the guidance, which will be revised and updated periodically.
What We Mean by Immunotoxicity
As used in this document, immunotoxicity refers to any adverse effect on the structure or function of the immune system, or on other systems as a result of immune system dysfunction. An effect is considered adverse or immunotoxic if it impairs humoral or cellular immunity needed by the host to defend itself against infectious or neoplastic disease (immunosuppression) or it causes unnecessary tissue damage (autoimmunity, hypersensitivity, or chronic inflammation). This definition incorporates the concept that the immune system is in a complex balance that includes interactions with other systems (e.g. nervous and endocrine) that may utilize or be affected by the same biological mediators (e.g. neuropeptide and steroid hormones).
"Change" in an immune function or level of immunological mediator may not necessarily appear as an "adverse effect", but rather as immunostimulation. Caution must be exercised in such cases, because a non-specific enhancement of the immune response that might be interpreted as a beneficial effect may result in suppression of specific immunity against a particular infection.
A decision on whether a material/device is immunotoxic must rely on the available evidence from pre-clinical test results and clinical evaluation, as well as prior history of use. Because the available data will often be less than conclusive, good judgment will play an important part in evaluating immunotoxic risk.
Purpose
The purpose of this guidance is to provide FDA reviewers and manufacturers with a systematic approach for evaluating potential adverse immunological effects of medical devices and constituent materials. It provides a coherent strategy for establishing the need for immunotoxicity testing, and guides the user through the steps involved in deciding what specific testing should be performed.
An important goal in designing this guidance has been to optimize testing requirements. Although for a given product new studies may be needed, reviewers and manufacturers are encouraged to use available data in order to minimize testing. Available data are relevant if they provide information necessary to determine the immunotoxic risk associated with the intended use of the device or material when taking into consideration the indicated patient population. Sources of such information include the scientific literature, data bases, recognized national and international standards, and guidance and related documents from within and outside FDA. Manufacturers are encouraged to discuss proposed testing with FDA to ensure that only appropriate and essential testing is performed.
The immune system is flexible and often able to utilize alternative factors and mechanisms to compensate for deficiencies in a particular immune function. For this reason, tests in appropriate animal models may provide a more accurate picture of immune system competence and a more relevant indication of immunotoxic potential than in vitro tests in which compensatory alternative mechanisms may be lacking. However, if a sound scientific rationale can be provided as to why in vitro tests will suffice, then their use is encouraged to minimize expense and numbers of experimental animals.
Format
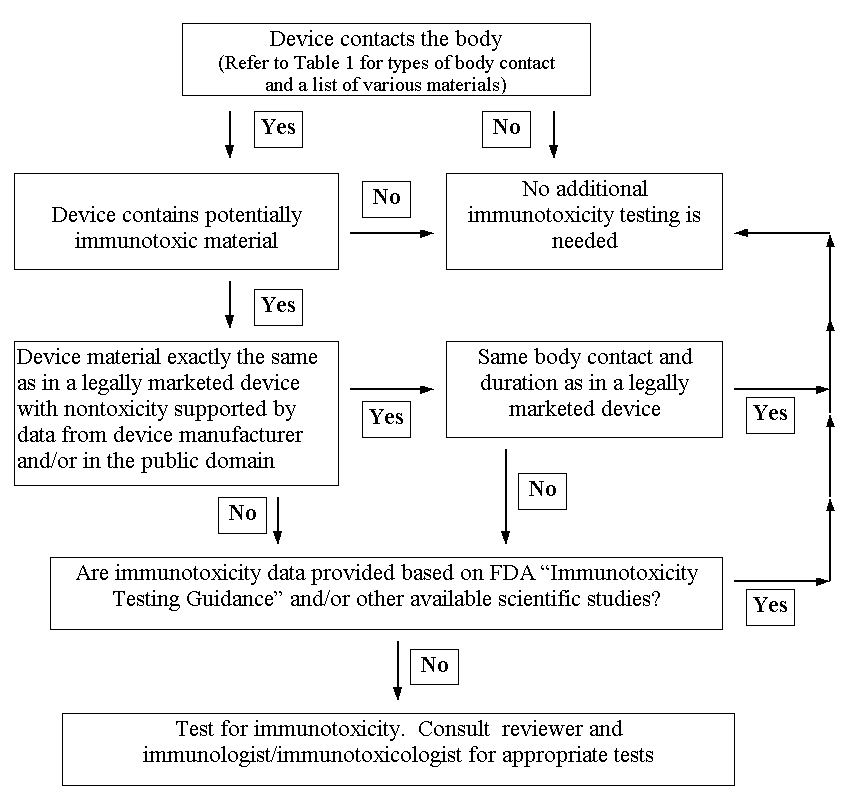
The guidance consists of a flow chart and three tables that follow the G95-1 (i.e. ISO 10993) format. The flow chart is used for determining whether immunotoxicity testing is likely to be needed, and includes the option of providing a rationale for not performing the testing based upon available published data or other sources of information on the same or like materials. This approach explicitly incorporates results of existing, scientifically sound studies into the decision making process.
How to Use This Guidance
Flow Chart for Immunotoxicity Testing
The flow chart should be used first to determine whether immunotoxicity testing may be needed to support the safety of the device. Testing generally will be appropriate for new materials or when there is concern that materials already in use for which adequate testing has not been carried out may be immunotoxic. Immunotoxicity testing may not be needed if the device material is the same as in a legally marketed device; has the same body contact, dose and duration; and there is either a long history of use without reported toxicity, or scientific data in the public domain supporting lack of toxicity. If immunotoxicity testing has been carried out as specified in G95-1/ISO 10993 as part of overall safety evaluation, then it need not be repeated. However, the Flow Chart may be used to determine if additional immunotoxicity testing is recommended beyond what is specified in G95-1/ISO 10993.
A decision on whether sufficient safety data are available for materials with an intended use different from an approved use will be made on a case-by-case basis.
Tables
When the flow chart indicates that immunotoxicity testing is recommended, Tables 1-3 are used sequentially to determine the types of testing that might be used to help evaluate product safety consistent with the intended use and indicated patient population and expected risk vs. benefit. They are intended to capture the most important adverse immunological effects and responses that might be associated with medical devices and materials, while providing flexibility in deciding what, if any, specific immunotoxicity tests will be performed.
Table 1
POTENTIAL IMMUNOTOXIC EFFECTS OF DEVICES AND CONSTITUENT MATERIALS
DEVICE NAME: _______________________________
| BODY CONTACT | CONTACT DURATION | IMMUNOTOXIC EFFECTS | |||||
|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | |||
| Surface Devices - | Skin | A | pmbx | x | |||
| B | pmbx | x | x | x | x | ||
| C | pmbx | x | x | x | x | ||
| Mucosal Membranes | A | pmbx | x | ||||
| B | pmbx | pmbx | mbx | x | x | ||
| C | pmbx | pmbx | mbx | mbx | mbx | ||
| Breached or Compromised Surface | A | pmbx | x | ||||
| B | pmbx | pmbx | mbx | mbx | mbx | ||
| C | pmbx | pmbx | mbx | mbx | mbx | ||
| External Communicating Devices - External devices that contact the circulating blood (e.g. dialyzers and immunoadsorbents); or the blood path indirectly at one point and serve as a conduit for entry into the vascular system (e.g. solution and blood administration sets); or tissue/bone/dentin (e.g. skin staples, laparoscopes, dental filling materials) | Blood Path, Direct and Indirect |
A | pmbx | x | |||
| B | pmbx | pmbx | mbx | pmbx | mbx | ||
| C | pmbx | pmbx | mbx | pmbx | mbx | ||
| Tissue/Bone/Dentin Communicating | A | pmbx | x | ||||
| B | pmbx | cpmbx | mbx | pmbx | mbx | ||
| C | pmbx | cpmbx | mbx | pmbx | mbx | ||
| Implant Devices - | Tissue/Bone, Blood, and other Body Fluids | A | pmbx | x | |||
| B | pmbx | cpmbx | mbx | pmbx | mbx | ||
| C | pmbx | cpmbx | mbx | pmbx | mbx | ||
A = Limited (
B = Prolonged (>24 hrs to 30 days)
C = Permanent (>30 days)
1 = Hypersensitivity
2 = Chronic Inflammation
3 = Immunosuppression
4 = Immunostimulation
5 = Autoimmunity
Effects Expected for Various Materials:
Plastics & Other Polymers = p
Metals = m
Ceramics, Glasses, Composites = c
Biological Materials = b
Other Materials (Specify) = x
Table 1 provides a guide to potential immunotoxic effects that might be associated with medical device materials. It follows the ISO classification scheme as specified in FDA General Program Memorandum (Blue Book Memo) G95-1 based on type and duration of body contact. These basic immunotoxic effects have been prioritized based on frequency of occurrence, duration, and severity of the reaction. Through this approach, Table 1 accounts for the major immunotoxic reactions that may be found with medical device materials, and balances the need to be inclusive while minimizing complexity.
The following glossary defines the Immunotoxic Effects shown in Table 1. Additional information may be found in the "General References" listed at the end of the guidance.
- HYPERSENSITIVITY: increased reactivity to an antigen to which a person (or animal) has been previously exposed, with an adverse rather than protective effect; sometimes used as a synonym for allergy. This Guidance includes only Type I (anaphylactic) reactions, which are mediated by IgE antibodies, and Type IV (delayed hypersensitivity) reactions mediated by T lymphocytes because they are the most common. Type I reactions also are the most serious. Type II and Type III reactions involve antibodies (IgG or IgM, but not IgE) and complement, but are relatively rare and are less likely to occur with medical devices/materials.
- CHRONIC INFLAMMATION: Inflammation is the normal tissue response to local injury. Acute inflammation is relatively short-lived (days) and characterized by neutrophils as the primary cellular infiltrate. In contrast, chronic inflammation can last up to months or longer, and is characterized by infiltration of macrophages and lymphocytes. Chronic inflammation may lead to immune granuloma formation and more serious immunological consequences, such as autoimmune disease.
- IMMUNOSUPPRESSION: Inhibition of the adaptive immune response (i.e. antibody and T cell responses); one potential consequence is more frequent and serious infections resulting from reduced host defense.
- IMMMUNOSTIMULATION: Unintended or inappropriate antigen-specific or non-specific activation of the immune system. For this guidance, immunostimulation includes a.) unintended immunogenicity of biomaterials (e.g. antibody and/or cellular immune response to a foreign protein), and b.) adjuvancy, enhancement of the immune response to an antigen by a material with which it is mixed ex vivo or in situ.
- AUTOIMMUNITY: Immune response to the body’s own constituents (autoantigens). An autoimmune response, indicated by the presence of autoantibodies or T lymphocytes reactive with host tissue or cell antigens, may (but not necessarily) result in autoimmune disease with chronic, debilitating, and sometimes life-threatening tissue and organ injury. In some cases, the specific autoantigens may not be characterized or known.
Chronic inflammation and immunostimulation have been included in Table 1 along with the more generally recognized adverse immunological effects; hypersensitivity, immunosuppression and autoimmunity. Although inflammation is part of the normal wound healing process, chronic inflammation, especially with prolonged or permanent implants, needs evaluation because it may lead to loosening of implants affixed to bone, dense- or pseudo-capsule formation, or other serious effects. Immunostimulation has been included separately from hypersensitivity to explicitly account for other enhanced immunological activity that may lead to serious clinical consequences. Examples include immunogenicity of biological or other materials, or adjuvant activity that may not result in adverse signs in relatively short term animal experiments, but are cause for concern with long-term implants or repeated exposure to injected materials that may result in sensitization or autoimmunity.
For this table, medical device materials have been placed into four categories that are broad enough to include most types of device materials. Materials were assigned to the boxes based upon body contact and duration and their potential for producing the indicated immunotoxic effects. A search of the scientific and medical literature was used as the primary basis for making these assignments.
An option for "Other Materials" that do not fall into the other four categories also is included. Examples include low molecular weight chemical stabilizers, cross-linking agents for polymers, and degradation products. These constituents, which may be present or produced in trace amounts (e.g. parts per million), should be evaluated for their potential to produce adverse immunological effects on a case-be-case basis. This approach is consistent with a method under development in CDRH for the evaluation of systemic toxicity, the hallmarks of which are chemical characterization of the materials used in the device and an assessment of the likelihood that these constituents would produce systemic effects in humans. "Other Materials" have been placed in most categories of "Body Contact" and "Duration" since these categories can not be excluded without data supporting lack of immunotoxicity.
Table 2
CLASSIFICATION OF IMMUNE RESPONSES ASSOCIATED WITH POTENTIAL IMMUNOTOXIC EFFECTS
| IMMUNOTOXIC EFFECTS | IMMUNE RESPONSES | |||||||
|---|---|---|---|---|---|---|---|---|
| HISTOPATHOLOGY | HUMORAL RESPONSE | CELLULAR RESPONSES | HOST RESISTANCE | OBSERVE FOR SIGNS OF ILLNESS | ||||
| T-CELLS | NATURAL KILLER CELLS | MACROPHAGES | GRANULOCYTES* | |||||
| 1 HYPERSENSITIVITY | NC | C (IgE in Type I Reactions Only) |
C (Type IV Reactions Only) |
NA | NA | C | NA | C |
| 2 INFLAMMATION | C | NC | C | NA | C | C | NA | C |
| 3 IMMUNOSUPPRESSION | NC | C | C | C | C | C | C | C |
| 4 IMMUNOSTIMULATION | NC | C | C | NA | NC | NA | NC | C |
| 5 AUTOIMMUNITY** | C | C | C | NA | NA | NC | NA | C |
C = Critical
NC = Non-Critical
NA = Not Applicable or Not Needed
*Basophils, Eosinophils, and/or Neutrophils
**Routine testing for autoimmunity is not recommended (see text).
Table 2 provides a set of responses that are commonly associated with the benchmark immunotoxic effects. When Table 1 indicates that one or more adverse effects may be associated with a device material, Table 2 is used to focus on the types of testing that might provide immunotoxic indications associated with those effects. As in Table 1, the classifications are sufficiently broad to encompass the predominant responses associated with the immunotoxic effects. The responses in Table 2 are designated as critical (C) or non-critical (NC). Critical indicates that there is primary importance in testing for these responses as indications of immunotoxicity. Testing for non-critical responses may be needed for adequate safety evaluation, for example when critical tests are positive. In general, any signs of immune system dysfunction should be recorded even if the observations were not included as part of a formal trial protocol. Appropriate studies should be considered to understand the basis for these responses.
TABLE 3
EXAMPLES OF TESTS, INDICATORS, AND MODELS FOR THE EVALUATION OF IMMUNE RESPONSES*
| IMMUNE RESPONSES | FUNCTIONAL ASSAYS | SOLUBLE MEDIATORS | PHENOTYPING | OTHER** |
|---|---|---|---|---|
| HISTOPATHOLOGY | NA | NA | Cell surface markers | Morphology |
| HUMORAL RESPONSE | Immunoassays (e.g. ELISA) for antibody response to antigen plus adjuvant* Plaque-forming cells Lymphocyte proliferation Antibody-dependent cell-mediated cytotoxicity Passive cutaneous anaphylaxis Direct anaphylaxis |
Complement (including C3a and C5a anaphylatoxins)*, Immune complexes | Cell surface markers | |
| CELLULAR RESPONSES | ||||
| T-CELLS | Guinea pig maximization test* Mouse local lymph node assay* Mouse ear swelling test Lymphocyte proliferation Mixed lymphocyte reaction |
Cytokine patterns indicative of T cell subsets (e.g. Th1 and Th2) | Cell surface markers (helper and cytotoxic T-cells) | |
| NATURAL KILLER CELLS | Tumor cytotoxicity | NA | Cell surface markers | |
| MACROPHAGES | Phagocytosis* Antigen presentation |
Cytokines (IL-1, TNF-alpha, IL-6, TGF-beta) | MHC markers | |
| GRANULOCYTES*** | Degranulation Phagocytosis |
Chemokines, Bioactive amines, Inflammatory cytokines, Enzymes | NA | Cytochemistry |
| HOST RESISTANCE | Resistance to bacteria, viruses and tumors | NA | NA | |
| SIGNS OF ILLNESS | NA | NA | NA | Allergy, Skin rash, Urticaria, Edema, Lymphadenopathy |
NA = Not Applicable or Not Needed
* Indicates most commonly used tests. Functional assays are generally more important than tests for soluble mediators or phenotyping. References at the end of this guidance provide detailed testing protocols.
** Animal models of some human autoimmune diseases are available (see references at the end of guidance). However, routine testing for induction of autoimmune disease by materials/devices is not recommended.
*** Basophils, Eosinophils and/or Neutrophils
Table 3 provides examples of the specific types of tests that might be used to study the responses listed in Table 2. These selected examples are only representative of the large number of tests that are currently available. Functional assays provide a more direct measure of immune system activity, and generally are more important than tests for soluble mediators, which are more important than phenotyping. The list likely will evolve as new and improved technology is developed and as additional direct or indirect markers of immune responses are validated and their predictive value documented. Examples of signs of illness as well as animal models (host resistance assays) for studying immune responses also are included.
The goal of this table is to facilitate the choice of tests, not to be inclusive or to serve as a prescription for test protocols that manufacturers must follow. The literature references on "Immunotoxicity Testing" at the end of this guidance provide detailed information on the selection and performance of a variety of test procedures that are widely used and believed to be valid. A valid test is one that has been shown to provide accurate, reproducible results that are truly indicative of the effect being studied.
Table 3 covers a large number of in vitro and in vivo procedures for measuring changes in variables indicative of immunotoxic effects. However, one common requirement is to ensure a sound statistical basis in study design that will permit differences between test and control groups to be measured at a desired level of statistical significance (usually at the p<0.05 level).="" also,="" in="" all="" studies="" care="" should="" be="" taken="" to="" mimic="" as="" closely="" as="" possible="" the="" intended="" use="" with="" regard="" to="" route="" of="" exposure/site="" of="" implantation,="" dose="" and="">
Results from preclinical immunotoxicity testing should be used to help assess biocompatibility of materials as part of the overall safety evaluation of medical devices. Significant indications of immunotoxicity may suggest that studies of immune function should be included in clinical trials and post-market studies.
Manufacturers are expected to be selective and to utilize immunotoxicity tests that are appropriate, technically valid, and predictive. That is, tests should be useful in assessing immunotoxic activity in relation to the particular responses and immunotoxic effects in question, and they must be sufficiently sensitive and specific to yield valid data. In Table 3, the tests most commonly used as indicators of a particular immune response are marked with an asterisk (*) (e.g. Immunoassays under "Humoral Response"). Other alternative valid tests are shown, and may also be considered. References at the end of the Guidance (Immunotoxicity Testing) provide details on experimental protocols, and analysis and interpretation of test results.
It should be noted that the tables will be used primarily for new materials, because the Flow Chart should exempt a large majority of materials already in use. Testing may be needed for materials currently used in medical devices if they will have a different or longer duration of body contact, or if exposure to larger doses is expected than in current applications.
Immunogenicity involving a specific immune response to a biomaterial is an important consideration because it may lead to serious adverse effects. For example, a foreign (i.e. non-human) protein may induce IgE antibodies that cause an anaphylactic (Type I) hypersensitivity reaction. An example is latex protein. In addition, low molecular weight compounds, e.g. chemical accelerators used in the manufacture of latex gloves, may induce a T cell mediated (Type IV) reaction resulting in contact dermatitis. Tests for Type I (e.g. antigen-specific IgE) and Type IV (e.g. guinea pig maximization test) hypersensitivity should be considered for materials with the potential to cause these allergic reactions.
In addition to hypersensitivity reactions, a biomaterial may elicit autoimmune responses (i.e. antibodies or T cells that react with the body’s own constituents). An autoimmune response may lead to pathological consequences (i.e. autoimmune disease). For example, a foreign protein may induce IgG or IgM antibodies that cross-react with a human protein and cause tissue damage by activating the complement system. Similarly, a biomaterial (e.g. a gel or oil) acting as an adjuvant may induce a harmful autoimmune response.
Reliable tests for autoantibodies (e.g. ELISA) and autoreactive T cells (e.g. lymphocyte proliferation) are available, and several animal models have been developed to study certain human autoimmune diseases. However, autoantibodies and autoreactive T lymphocytes may only be indicators of an autoimmune response. Even if an autoimmune response can be demonstrated in preclinical testing, convincing evidence that a biomaterial causes autoimmune disease in animals is difficult to obtain. Therefore, routine testing for induction of autoimmune disease in animal models is not recommended. Testing for autoimmunity in animals may be warranted if long-term use of a biomaterial is suspected of causing autoimmune disease in humans. However, these cases are expected to be extremely rare. Testing for autoimmunity is an additional area where discussion with immunologists/immunotoxicologists in FDA should be helpful. References at the end of this guidance provide more information on this topic.
Additional Reviewer Support
In addition to this guidance and G95-1, there are other printed and computerized sources of information that provide in depth didactic and technical details on immunotoxicity testing. Several useful references appear at the end of this guidance. Important details include background information on test sensitivity, specificity, and predictive value, basic and alternative protocols, critical variables, anticipated results, time considerations, technical trouble-shooting, and primary literature references. This information will be available to reviewers.
Consults with immunologists and immunotoxicologists in CDRH and the other centers also will provide reviewers with useful information on the performance of immunotoxicity testing and data evaluation. To enhance communication and the availability of technical expertise, an "Immunotoxicology Expertise Resource" is available on the CDRH Intranet Home Page (www.cdrh.fda.gov) that provides reviewers with access to individuals Agency-wide with a wide range of expertise in immunotoxicology. This is in addition to a moderated discussion/conferencing capability called IMMUNOTOX available within FDA that provides a confidential forum for discussion of regulatory review issues in immunology/immunotoxicology. All of these resources should be utilized by reviewers to facilitate the review process.
What the Guidance Does Not Do
This guidance provides direction on the types of immunotoxicity testing that should be considered for biomaterials. It is not a plan for establishing testing requirements for materials/devices. It does not prescribe what tests should be performed or the protocols that should be followed. Nor does it specify or restrict the sources of information that may be used to support a claim that a material is not immunotoxic, although care should be taken to insure that the basis for such a claim rests on strong scientific evidence and does not rely on proprietary data without appropriate right of reference. By incorporating flexibility into decision making, the guidance relies on sound judgment in determining the need for immunotoxicity testing and the appropriate tests that will help provide necessary and sufficient data in support of product safety.
REFERENCES
General
"Illustrated Dictionary of Immunology" (Cruse, JM and Lewis, RE, Eds) CRC Press, Boca Raton (1995).
Benjamini, E, Sunshine, G, and Leskowitz, S: "Immunology: A Short Course", (3rd Edition) Wiley-Liss, Inc., New York (1996).
Janeway, Jr., CA and Travers, P: "Immunobiology: The Immune System in Health and Disease", (3rd Edition) Current Biology-Garland, New York (1997).
Animal Models of Autoimmune Disease
Rose, NR: "Immunologic Diagnosis of Autoimmune Disease" in "Handbook of Human Immunology" (Lefell, MS, Donnenberg, AD, and Rose, NR, Eds) CRC Press, Boca Raton (1997), pp. 111-123.
"Autoimmune Disease Models: A Guidebook" (Cohen, IR and Miller, A, Eds) Academic Press, New York (1994).
"Handbook of Animal Models for the Rheumatic Diseases" (Vol. I) (Greenwald, RA and Diamond, HS, Eds) CRC Press, Boca Raton (1988).
Immunotoxicity Testing
The following references provide information on immunotoxicity testing including methodology, applications, and data evaluation. Although the list is not comprehensive, it does provide a core of didactic background and technical detail that reviewers and manufacturers should find useful. Primary references to original articles are often included.
"Clinical Diagnostic Immunology: Protocols in Quality Assurance and Standardization", (Nakamura, RM, Burek, CL, Cook, L, Folds, JM, and Sever, JL, Eds) Blackwell Science, Inc., Malden (1998).
"Manual of Clinical Laboratory Immunology", (5th Edition) (Rose, NR, de Mecario, EC, Folds, JD, Lane, HC, and Nakamura, RM, Eds) ASM Press, Washington (1997).
"Experimental Immunotoxicology" (Smialowicz, RJ and Holsapple, MP, Eds) CRC Press, Boca Raton (1996).
"Methods in Immunotoxicology" (Burleson, GR, Dean, JH, and Munson, AE, Eds) Wiley-Liss, New York (1995).
"Current Protocols in Immunology" (Coligan, JE, Kruisbeek, AM, Margulies, DH, Shevach, EM, and Strober, R, Eds) Greene Publishing Associates and Wiley Interscience, New York (1992). Supplemented periodically with new or updated methods.
1 This document is intended to provide guidance. It represents the Agency’s current thinking on the above. It does not create or confer any rights for or on any person and does not operate to bind FDA or the public. An alternative approach may be used if such approach satisfies the requirements of the applicable statute, regulations, or both.
Submit Comments
You can submit online or written comments on any guidance at any time (see 21 CFR 10.115(g)(5))
If unable to submit comments online, please mail written comments to:
Dockets Management
Food and Drug Administration
5630 Fishers Lane, Rm 1061
Rockville, MD 20852
All written comments should be identified with this document's docket number: FDA-2020-D-0957.