CraniSeal Dural Sealant – P220014
This is a brief overview of information related to FDA’s approval to market this product. See the links below to the Summary of Safety and Effectiveness Data (SSED) and product labeling for more complete information on this product, its indications for use, and the basis for FDA’s approval.
Product Name: CraniSeal Dural Sealant
PMA Applicant: Pramand, LLC
Address: 201 Burlington Road, Suite 210, Bedford, MA, 01730
Approval Date: July 6, 2023
Approval Letter: Approval Order
What is it?
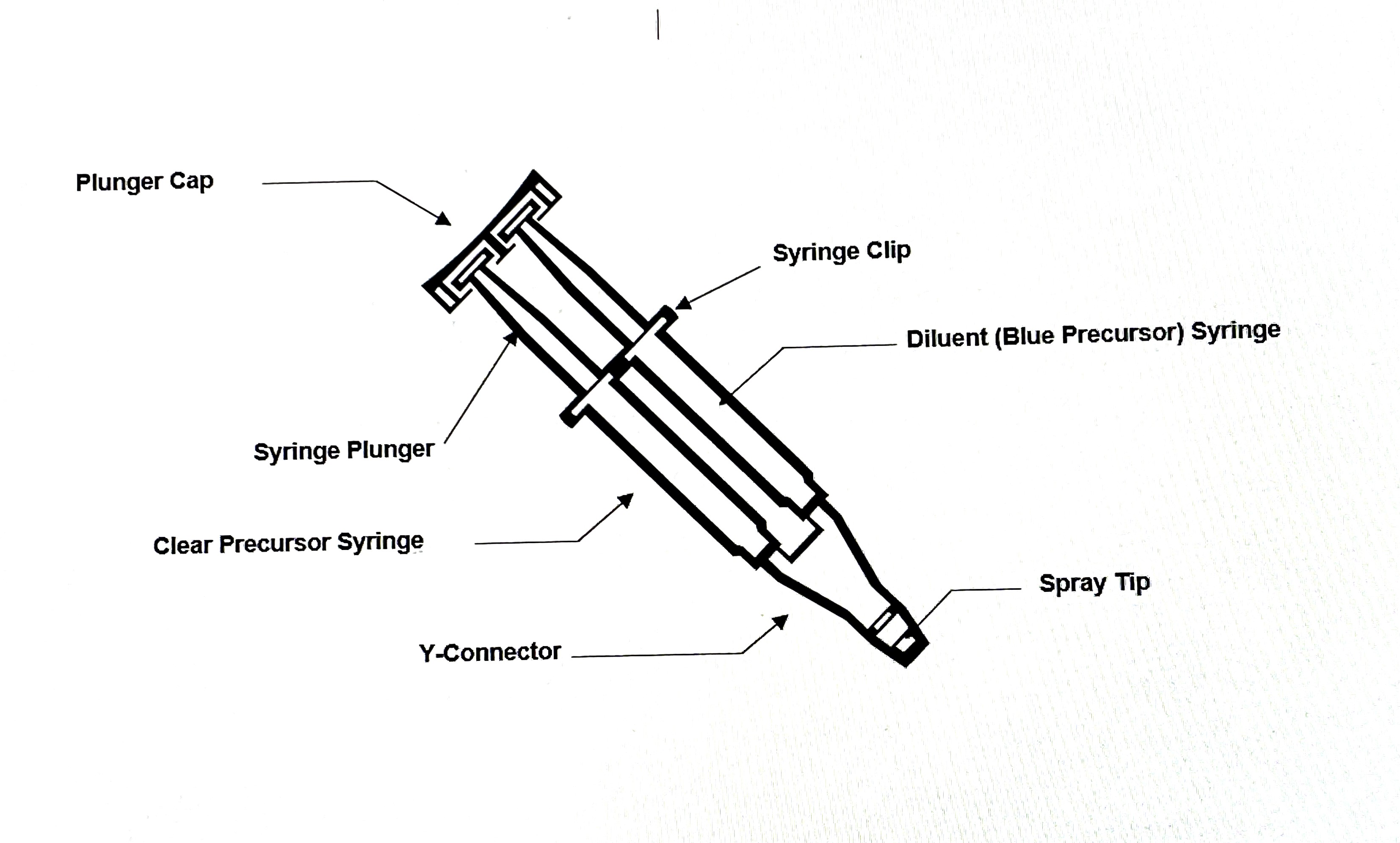
CraniSeal Dural Sealant is an absorbable polyethylene glycol (PEG) hydrogel that is applied with an applicator over stitches (sutures). This sealant prevents cerebrospinal fluid (CSF) from leaking out of a skull (cranial) surgery incision site that includes dura mater repair. The dura mater is the tough, outermost, fibrous membrane that covers the brain and spinal cord. It lines the inner surface of the skull.
How does it work?
The CraniSeal Dural Sealant is made of two substances, a PEG ester solution and a trilysine amine solution. When mixed together, these substances rapidly combine (polymerize) to form a hydrogel seal. The sealant is absorbed in about four to eight weeks and then eliminated from the body.
When is it used?
The CraniSeal Dural Sealant is indicated for use in patients 18 years of age and older as a supplemental, watertight closure for a sutured dura repair during cranial surgery.
What will it accomplish?
The effectiveness of the CraniSeal Dural Sealant was measured through a direct comparison to FDA-approved, commercially available DuraSeal Dural Sealant (P040034) in nonclinical studies because CraniSeal Dural Sealant has the same design and chemical specifications as the already marketed DuraSeal Dural Sealant.
The prior clinical experience of the DuraSeal Dural Sealant showed the device was able to seal 98% of sutured dural incisions during surgery. Those clinical outcomes can also be applied to the performance of the CraniSeal Dural Sealant.
When should it not be used?
The CraniSeal Dural Sealant should not be used:
- For surgical procedures not included in the indications for use.
- For confined bony structures where nerves are present, as nerve (neural) compression may occur from the hydrogel swelling. This hydrogel may swell to 50% greater than its original size in any direction as it seals.
Additional information (including warnings, precautions, and adverse events):