FY2015 Regulatory Science Research Report: Nanotechnology: Postmarket: Data Analysis for Generic Drugs
Introduction
In light of the high rate of generic drug usage in the United States, effective postmarketing surveillance of generic drugs is essential for ensuring continued generic drug safety and effectiveness. Postmarketing surveillance of generics has the same goal as surveillance of new drugs, i.e., to monitor drug use, clinical effectiveness, and safety issues after a drug is marketed and reaches a larger and broader population. However, several additional and unique aspects exist with regard to surveillance of generic drugs. Evaluation of substitutability can supplement BE testing and determine if patient characteristics interact with formulation differences between products. Postmarketing surveillance of generics also allows for the identification of any product quality issues that may arise during the manufacturing process. Studies are designed to address negative perceptions and concerns among patients or healthcare providers regarding generics and promote confidence in the interchangeability of brand-name and generic drugs. Overall, monitoring drug usage trends and switching patterns over time and evaluating the effects on healthcare outcomes provides valuable information on real-world use of these products and helps to identify clinical or regulatory issues that might need to be addressed.
Research
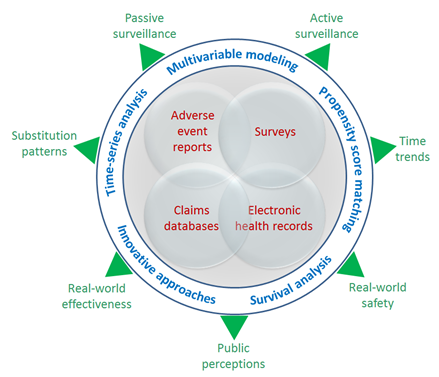
In ORS, we use several complementary approaches to generic drug surveillance that include several data sources and data analysis methods. We currently use adverse event report data from FAERS and published literature, administrative claims databases, electronic health records, and survey data. These data sources are used in a variety of approaches for postmarketing surveillance of generic drugs, including literature reviews; case report reviews (from literature, media, and social networking sites); passive surveillance evaluations; patient, pharmacist, and physician surveys; and active surveillance, which includes retrospective analyses of secondary data and prospective data collection. Postmarketing investigations involving passive and active surveillance often require complex statistical approaches for analyzing the data; these may include interrupted time-series analysis, multivariable modeling, propensity-score matching, survival analysis, and other innovative statistical methods.
Figure 22. Analyzing data for postmarketing surveillance of generics
ORS staff facilitating research in this area
- Sarah Dutcher, Xinyuan Zhang, Liang Zhao, Meng Hu, Xia Pu, Zhong Wang
Projects and Collaborators
- Assessing clinical equivalence for generic drugs approved by innovative methods
- Site PI: Aaron Kesselheim
- Grant #: U01FD004856
- Does variation in the physical characteristics of generic drugs affect patients’ experiences? Surveys of pharmacists and patients
- Site PI: Aaron Kesselheim
- Contract #: HHSF223201310232C
- Postmarketing surveillance of generic drug usage and substitution patterns
- Site PI: Ilene Harris
- Grant #: U01FD004855
- Assessing the post-marketing safety of authorized generic drug products
- Site PI: Joshua Gagne
- Grant #: U01FD005279
- Post-market authorized generic evaluation (PAGE)
- Site PI: Richard Hansen
- Grant #: U01FD005272
- Effect of therapeutic class on generic drug substitutions
- Site PI: Jodi Segal
- Grant #: U01FD005267
- Novel approaches for confounding control in observational studies of generic drugs
- Site PI: Rishi Desai
- Grant #: U01FD005555
- Structural nested models for assessing the safety and effectiveness of generic drugs
- Site PI: Ravi Varadhan
- Grant #: U01FD005556
- Comparative surveillance of generic drugs by machine learning
- Site PI: Peggy Peissig
- Contract #: HHSF223201510112C
Publications and Presentations
- Kesselheim AS, Polinski HM, Fulchino LA, Isaman DL, Gagne JJ. Modified regulatory pathways to approve generic drugs in the U.S. and a systematic review of their outcomes. Drugs 75(6):633-50 (2015)
- Kesselheim AS, Gagne JJ. Product-Specific Regulatory Pathways to Approve Generic Drugs: The Need for Follow-up Studies to Ensure Safety and Effectiveness. Drug Safety 38(10):849-53 (2015)
- Khokhar B, Park Y, Pradel F, Palumbo F, Kiptanui Z, Dutcher S, Jiang W, Zuckerman I. Patients’ Awareness of Bioequivalence Study Methods Supporting Generic Venlafaxine Extended Release (ER) Tablet Approval. Abstract #274. 31st International Conference on Pharmacoepidemiology. August 22-26, 2015. Boston, Massachusetts.
- Segal JB, Daubresse M, Lee CY, Moechtar M, Dutcher S, Jiang W, Romanelli R. Therapeutic Class Differences in Generic Usage. Abstract # 245. International Conference on Pharmacoepidemiology. August 22-26, 2015. Boston, Massachusetts.
- Park JY, Kiptanui Z, Palumbo F, Zuckerman I, Pradel F, Khokar B, Jiang W, Cai W, Dutcher S. Comparing clinical outcomes of brand and generic drugs: systematic literature review of acarbose, calcitonin nasal spray, and venlafaxine ER tablets. American Society of Health-System Pharmacists Midyear Clinical Meeting. December 7-11, 2014. Anaheim, CA.
- Kesselheim AS, Gagne JJ, Eddings W, Franklin JM, Ross KM, Fulchino LA, Campbell EG. Prevalence and Predictors of Generic Drug Skepticism Among Physicians: Results of a National Survey. JAMA Intern Med. 2016 Jun 1;176(6):845-7.
- Gagne JJ, Polinski JM, Jiang W, Dutcher SK, Xie J, Lii J, Fulchino LA, Kesselheim AS. Switch-backs associated with generic drugs approved using product-specific determinations of therapeutic equivalence. Pharmacoepidemiol Drug Saf. 2016 Apr 22.
- Kesselheim AS, Gagne JJ, Franklin JM, Eddings W, Fulchino LA, Avorn J, Campbell EG. Variations in Patients' Perceptions and Use of Generic Drugs: Results of a National Survey. J Gen Intern Med. 2016 Jun;31(6):609-14.
- Romanelli RJ, Nimbal V, Segal JB. Provider-level variation and determinants of outpatient generic prescribing in a mixed-payer healthcare system. 22nd Annual Health Care Systems Research Network Conference. April 13-16, 2016. Atlanta, GA.
- Nimbal V, Segal JB, Romanelli RJ. Estimating generic drug utilization with electronic health records from a mixed-payer ambulatory healthcare delivery system. 22nd Annual Health Care Systems Research Network Conference. April 13-16, 2016. Atlanta, GA.
- Iyer G, Marimuthi S, Segal JB, Singh S. Identification of generic drugs in the FDA adverse event reporting system. International Society for Pharmacoepidemiology Mid-Year Meeting. April 10-12, 2016. Baltimore, MD.
- Marimuthu S, Iyer G, Segal JB, Singh S. Patient Relevant Outcomes Associated with Generic Drugs in FDA's Adverse Event Reporting System. International Society for Pharmacoepidemiology Mid-Year Meeting. April 10-12, 2016. Baltimore, MD.
- Daubresse M, Lee CY, Moechtar M, Dutcher S, Romanelli R, Segal JB. Predictors of generic thyroid hormone utilization among the commercially insured. International Society for Pharmacoepidemiology Mid-Year Meeting. April 10-12, 2016. Baltimore, MD.
Outcomes
- Research projects in progress