Produce Regulatory Program Standards (PRPS)
- What are the Produce Regulatory Program Standards (PRPS)?
- Which groups provided input on the PRPS?
- What does conformance with the PRPS entail?
- Which states have a regulatory agency(s) currently enrolled in the PRPS?
- Where can I find a copy of the PRPS?
- Is funding available to assist states in implementing the PRPS?
- What other resources are available to help state programs implement PRPS?
- Who do I contact for more information about PRPS?
- Produce Regulatory Program Standards CAP Program Fact Sheet (Version November 2025)
- Produce Regulatory Program Standards Informational Brochure
What are the Produce Regulatory Program Standards (PRPS)?
The Produce Regulatory Program Standards (PRPS) establish a uniform foundation for government agencies who have actual or potential regulatory oversight and responsibility over their respective jurisdiction’s farms, produce commodities, and activities covered under the FDA's Standards for the Growing, Harvesting, Packing, and Holding of Produce for Human Consumption regulation (aka the Produce Safety Rule) (21 CFR Part 112). When fully implemented, the PRPS define a set of best practices of a produce safety regulatory system and are designed to promote increased consistency of inspections across the nation. The PRPS provides a framework for self-assessments against a national standard that every state and territory produce safety regulatory program can use to identify the strengths, gaps, and needs of their programs.
Comprising ten standards, the PRPS establish requirements for the critical elements of a regulatory program designed to protect the public from produce related illness, injury, and outbreaks; and strengthen capability to manage emergencies.
The PRPS areas of focus are:
- Standard 1 - Regulatory Foundation
- Standard 2 - Training Program
- Standard 3 - Inspection Program
- Standard 4 - Inspection Audit Program
- Standard 5 - Foodborne Illness, Outbreak, and Response
- Standard 6 - Compliance and Enforcement Program
- Standard 7 - Outreach Activities
- Standard 8 - Program Resources
- Standard 9 - Program Assessment
- Standard 10 - Laboratory Support
Which groups provided input on the PRPS?
These standards were developed in close collaboration with state produce inspection programs enrolled in the FDA-State Produce Safety Implementation Cooperative Agreement Program (aka State Produce Safety CAP), the National Association of State Departments of Agriculture (NASDA), and the Association of Food and Drug Officials (AFDO). The result of this collaborative work is the development of the first version of the PRPS. These program standards reflect an effort in which the FDA has been engaged to partner, leverage, and empower state and territory agencies to implement the vision of an Integrated Food Safety System (IFSS). When fully implemented, the program standards define a set of best practices of a regulatory system.
What does conformance with the PRPS Entail?
Conformance starts with the regulatory programs assessing their authorities, practices, and documentation against the PRPS elements to identify gaps. Programs then develop and implement a strategic improvement plan to address these gaps. This process is repeated on a regular basis to drive continuous improvement.
Full conformance with the PRPS facilitates a state of mutual reliance between the FDA and regulatory agencies, and will lessen redundancies and inconsistencies of regulatory oversight of produce throughout the nation.
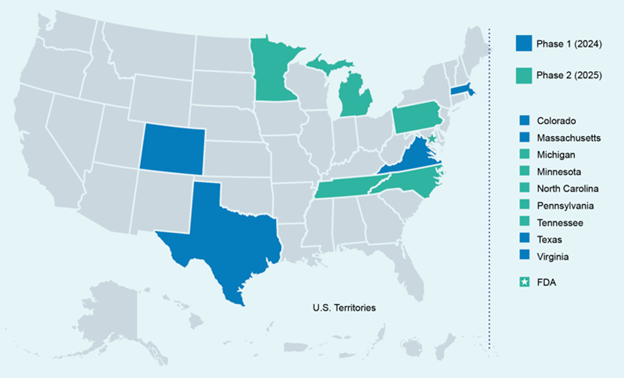
Which states have a regulatory agency(s) currently enrolled in the PRPS?
Formal enrollment in PRPS-related activities is not currently available; however, FDA has initiated a pilot program where nine programs in the State Produce CAP, as well as the FDA, have elected to complete some foundational PRPS work. The goal of the PRPS Pilot Program is to provide insight on the PRPS self-assessments process and the alignment of the PRPS with current produce safety program activities to effectively prepare for future formal enrollment in the PRPS. FDA participating in the pilot provides self-examination of processes to identify strengths and areas for improvement.
These ten PRPS pilot programs are completing PRPS self-assessments and using the results to develop PRPS strategic improvement plans. They are also providing input, data, and feedback about these processes to the FDA, NASDA, AFDO, and other Pilot Programs.
Where can I find a copy of the PRPS?
Download the latest copy of the PRPS by clicking here. You will need to have a PDF viewer installed on your computer or mobile device.
Is funding available to assist states in implementing the PRPS?
Currently, there is no dedicated funding for produce programs to implement the PRPS. Nine programs enrolled in the State Produce CAP have voluntarily begun informal implementation of the PRPS via PRPS pilot programs using existing FDA grant funding from the State Produce Safety Cooperative Agreement; more information about current grant funding for state and territorial produce safety programs can be obtained by viewing the latest Funding Opportunity Announcement (FOA).
What other resources are available to help state programs implement PRPS?
The FDA Office of Domestic Partnerships, Division of Domestic Partnership Coordination and Integration (ODP DDPCI) is responsible for conducting education and outreach to state programs on the PRPS.
Who do I contact for more information about PRPS?
Specific questions on this program can be directed to the appropriate Office of Domestic Partnerships’ contact within the Division of Domestic Partnership Coordination and Integration.
For information about program standards, please visit the Regulatory Program Standards webpage.
General questions can be directed to: ODP.Feedback@fda.hhs.gov