Drug Trials Snapshots: Forzinity
HOW TO USE THIS SNAPSHOT
The information provided in Snapshots highlights who participated in the key clinical trials that supported the original FDA approval of this drug, and whether there were differences among sex, race, age, and ethnic groups. The “MORE INFO” bar shows more detailed, technical content for each section. The Snapshot is intended as one tool for consumers to use when discussing the risks and benefits of the drugs.
LIMITATIONS OF THIS SNAPSHOT
Do not rely on Snapshots to make decisions regarding medical care. Always speak to your healthcare provider about the benefits and risks of a drug.
Some of the information in this Snapshot is for presentation purposes and does not represent the approved conditions of use of this drug. Refer to the FORZINITY Prescribing Information for all the approved conditions of use of this drug (e.g., indication(s), population(s), dosing regimen(s), safety information).
Snapshots are limited to the information available at the time of the original approval of the drug and do not provide information on who participated in clinical trials that supported later approvals for additional uses of the drug (if applicable).
FORZINITY (elamipretide)
(for zin’ I tee)
Stealth Biotherapeutics Inc.
Approval date: September 19, 2025
DRUG TRIALS SNAPSHOT SUMMARY:
What is the drug for?
FORZINITY is a mitochondrial cardiolipin binder, indicated to improve muscle strength in adults and children with Barth syndrome weighing at least 30 kg.
How is this drug used?
FORZINITY is injected under the skin (subcutaneous injection) once a day.
Who participated in the clinical trials?
The clinical trial enrolled 12 patients with Barth syndrome at a single site in the United States. All patients were male aged ≥12 years and not Hispanic or Latino. All were White except for one patient who was American Indian or Alaska Native. Supportive safety information also came from other clinical trials of FORZINITY in different disease populations.
How were the trials designed?
FORZINITY was evaluated in a randomized, double-blind, placebo-controlled, crossover trial and its 192-week open-label, single arm extension period (SPIBA-201). All 12 patients completed the randomized controlled trial. Ten of these patients entered the extension period, eight of whom participated through 168 weeks of the extension period.
How were the trials designed?
SPIBA-201 was a two-part study that enrolled 12 patients with genetically confirmed Barth syndrome.
Part 1 was a 28-week, randomized, double-blind, placebo-controlled crossover study that evaluated the efficacy and safety of FORZINITY 40 mg injected subcutaneously (SC) once a day compared to placebo in patients with Barth syndrome.
Part 2 was a 192-week, single-arm, open-label extension that evaluated long-term safety, tolerability and efficacy of FORZINITY 40 mg SC in patients with Barth syndrome.
The primary endpoints for Part 1 were distance walked during 6-minute walk test and Total Fatigue Score on the Barth syndrome Symptom Assessment. Ten patients completed the randomized trial and entered Part 2. Eight of these 10 patients participated through Week 168 of Part 2.
DEMOGRAPHICS SNAPSHOT
Figure 1 summarizes how many male and female patients were enrolled in the clinical trial used to evaluate the efficacy of FORZINITY.
Figure 1. Baseline Demographics by Sex, Efficacy Population
Source: Adapted from FDA Review
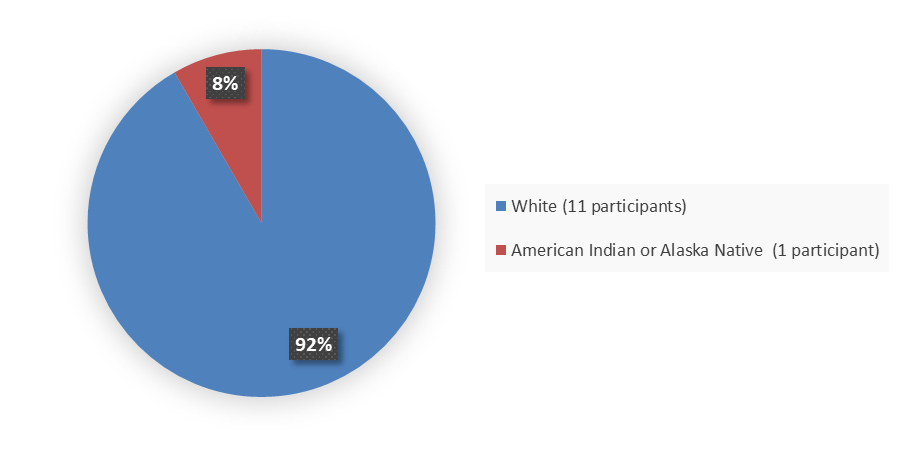
Figure 2 summarizes how many patients by race were enrolled in the clinical trial used to evaluate the efficacy of FORZINITY.
Figure 2. Baseline Demographics by Race, Efficacy Population
Source: Adapted from FDA Review
Figure 3 summarizes how many patients by age were enrolled in the clinical trial used to evaluate the efficacy of FORZINITY.
Figure 3. Baseline Demographics by Age, Efficacy Population
Source: Adapted from FDA Review
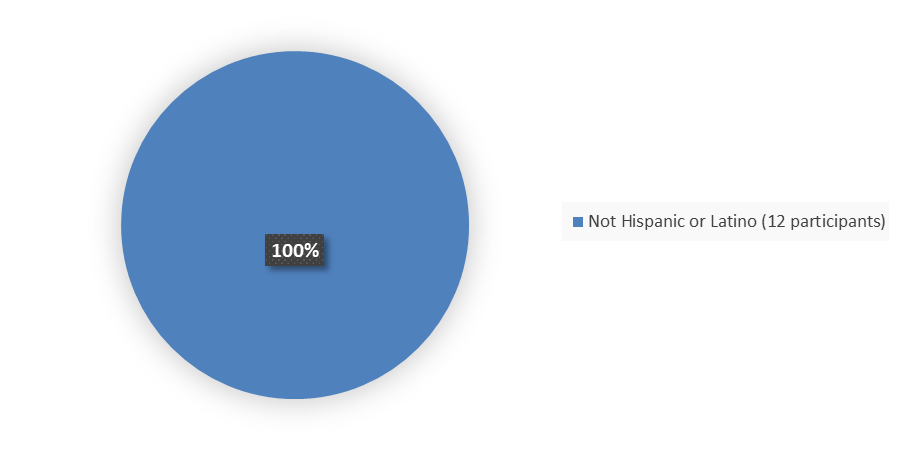
Figure 4 summarizes how many patients by ethnicity were enrolled in the clinical trial used to evaluate the efficacy of FORZINITY.
Figure 4. Baseline Demographics by Ethnicity, Efficacy Population
Source: Adapted from FDA Review
Who participated in the trials?
Table 1. Baseline Demographics, Efficacy Population, SPIBA-201 Part 1
| Demographic | N=12 |
|---|---|
| Sex, n (%) | |
| Male | 12 (100) |
| Age, years | |
| Mean (SD) | 19.5 (7.6) |
| Median (min, max) | 16.5 (12, 35) |
| Age group, years, n (%) | |
| <Median (16.5) | 6 (50.0) |
| ≥Median (16.5) | 6 (50.0) |
| Race, n (%) | |
| American Indian or Alaska Native | 1 (8.3) |
| White | 11 (91.7) |
| Ethnicity, n (%) | |
| Not Hispanic or Latino | 12 (100) |
Source: Adapted from FDA Review
What are the benefits of this drug?
FORZINITY was approved based on an increase in knee extensor muscle strength observed in a clinical trial. However, a significant difference was not shown during the initial 12-week randomized portion of the study; the observed increases occurred during a longer, open-label period.
FORZINITY was approved under FDA’s accelerated approval program, which provides earlier patient access to a promising new drug while the company continues to conduct clinical trials to confirm that the drug works well.
What are the benefits of this drug (results of trials used to assess efficacy)?
The randomized portion of SPIBA-201 (Part 1) evaluated the efficacy and safety of FORZINITY 40 mg SQ once daily for 12 weeks in 12 patients aged ≥12 years old and weighing >30 kg with genetically confirmed Barth syndrome. The primary endpoints for Part 1 were distance walked during 6-minute walk test and Total Fatigue Score on the Barth syndrome Symptom Assessment. FORZINITY was not superior to placebo on these primary endpoints.
Knee extensor muscle strength measured by handheld dynamometry was evaluated as one of the secondary endpoints in Part 1 and in the extension period (Part 2). Increases in knee extensor muscle strength were not observed during the randomized trial but were observed during the extension period. At the baseline visit at the start of the randomized trial, median muscle strength was 124 newtons. Table 2 shows descriptive changes from baseline for knee extensor muscle strength during the randomized controlled trial and extension period.
Table 2. Descriptive Statistics, Change From Baseline in Muscle Strength (newtons), Efficacy Population, Study SPIBA-201
| Trial Period | Visit | N | Median Change |
|---|---|---|---|
| Randomized controlled trial (Part 1) | Week 12 Placebo | 12 | -5 |
| Week 12 FORZINITY | 12 | 4 | |
| Open-label extension period (Part 2) | Week 12 | 10 | 34 |
| Week 24 | 9 | 68 | |
| Week 36 | 8 | 57 | |
| Week 48 | 8 | 41 | |
| Week 72 | 8 | 35 | |
| Week 168 | 8 | 63 |
Source: Adapted from FORZINITY Prescribing Insert
Were there any differences in how well the drug worked in clinical trials among sex, race, and age?
All participants were males and Not Hispanic or Latino. All but one participant was White. Also, the number that completed the open-label extension period was small. Therefore, subgroup analyses for efficacy were not conducted.
What are the possible side effects?
Some people taking FORZINITY have had allergic reactions. In some cases, these reactions were serious and required emergency medical care. These allergic reactions can affect your skin (rash, bumps, or itchy, inflamed skin) or cause breathing problems including coughing.
FORZINITY may cause skin irritation or other reactions at the injection site.
What are the possible side effects (results of trials used to assess safety)?
Table 3 summarizes the injection site reactions that occurred in FORZINITY-treated patients during the randomized, controlled trial.
Table 3. Patients With injection Site Reactions, Safety Population, Study SPIBA-201, Part 1
| Adverse Event | FORZINITY N=12 n (%) | Placebo N=12 n (%) |
|---|---|---|
| Local administration reaction | 12 (100) | 8 (66.7) |
| Injection site erythema | 12 (100) | 3 (25.0) |
| Injection site induration | 8 (66.7) | 2 (16.7) |
| Injection site pruritus | 8 (66.7) | 2 (16.7) |
| Injection site pain | 9 (75.0) | 5 (41.7) |
| Injection site bruising | 3 (25.0) | 0 |
| Injection site urticaria | 3 (25.0) | 0 |
| Injection site hemorrhage | 0 | 1 (8.3) |
Source: Adapted from FDA Review
Were there any differences in side effects among sex, race and age?
All FORZINITY-treated patients had skin irritations or other reactions at the injection site. Subgroup analyses for other side effects were not conducted due to similar reasons as provided for not conducting subgroup analyses for efficacy.
GLOSSARY
CLINICAL TRIAL: Voluntary research studies conducted in people and designed to answer specific questions about the safety or effectiveness of drugs, vaccines, other therapies, or new ways of using existing treatments.
COMPARATOR: A previously available treatment or placebo used in clinical trials that is compared to the actual drug being tested.
EFFICACY: How well the drug achieves the desired response when it is taken as described in a controlled clinical setting, such as during a clinical trial.
PLACEBO: An inactive substance or “sugar pill” that looks the same as, and is given the same way as, an active drug or treatment being tested. The effects of the active drug or treatment are compared to the effects of the placebo.
SUBGROUP: A subset of the population studied in a clinical trial. Demographic subsets include sex, race, and age groups.