Drug Trials Snapshots: REXULTI for the treatment of schizophrenia
HOW TO USE THIS SNAPSHOT
The information provided in Snapshots highlights who participated in the clinical trials that supported the FDA approval of this drug, and whether there were differences among sex, race, and age groups. The “MORE INFO” bar shows more detailed, technical content for each section. The Snapshot is intended as one tool for consumers to use when discussing the risks and benefits of the drugs.
LIMITATIONS OF THIS SNAPSHOT:
Do not rely on Snapshots to make decisions regarding medical care. Always speak to your health provider about the risks and benefits of a drug. Refer to the REXULTI Prescribing Information for complete information.
REXULTI (brexpiprazole)
(REX-ul-TE)
Otsuka America Pharmaceuticals, Inc.
Approval date: July 10, 2015
DRUG TRIALS SNAPSHOT SUMMARY:
What is the drug for?
REXULTI is a drug used for the treatment of schizophrenia.
Schizophrenia is a brain disorder characterized by hearing voices, believing other people are reading one’s mind or controlling one’s thoughts, and being suspicious or withdrawn.
How is this drug used?
REXULTI is a tablet that is taken once a day.
What are the benefits of this drug?
REXULTI improved symptoms of schizophrenia.
What are the benefits of this drug (results of trials used to assess efficacy)?
The table below summarizes the primary efficacy endpoints for both trials. This was based on the mean change in the Positive and Negative Syndrome Scale (PANSS) total score after 6 weeks. The population represents the Efficacy population which includes all subjects in the Safety population who had at least one post-randomization efficacy evaluation for the PANSS total score.
Table 2. Summary of Efficacy Results for Trials 1 and 2
| Trial | Treatment group | N | Primary Efficacy Measure: PANSS | ||
|---|---|---|---|---|---|
| Mean Baseline Score (SD) | LS Mean Change from Baseline (SE) | Placebo –subtracted Difference a (95% CI) | |||
| 1 | REXULTI (2mg/day)* REXULTI (4mg/day)* Placebo |
180 178 178 |
95.9 (13.8) 94.7 (12.06) 95.7 (11.5) |
-20.7 (1.5) -19.7 (1.54) -12 (1.6) |
-8.7 (-13.1, -4.4) -7.6 (-12.0, -3.1) - |
| 2 | REXULTI (2mg/day) REXULTI (4mg/day)* Placebo |
179 181 180 |
96.3 (12.9) 95.0 (12.4) 94.6 (12.8) |
-16.6 (1.5) -20.0 (1.5) -13.5 (1.5) |
-3.1 (-7.2, 1.1) -6.5 (-10.6, -2.4) - |
SD: standard deviation; SE: standard error; LS Mean: least-squares mean; CI: unadjusted confidence interval;
*Dosages statistically significantly superior to placebo.
a Difference (drug minus placebo) in least-squares mean change from baseline.
Source: REXULTI Prescribing Information Section 14, Table 12
Were there any differences in how well the drug worked in clinical trials among sex, race and age?
Subgroup analyses were conducted for sex, race, and age.
Were there any differences in how well the drug worked in clinical trials among sex, race, and age groups?
The tables below summarizes the responses to REXULTI for the populations in pooled trials. The population represents the Efficacy population, which includes all subjects in the Safety population who had at least one post-randomization efficacy evaluation for PANSS total score.
Table 3. Subgroup Analysis of Primary Endpoint, Pivotal Efficacy Trials (pooled data from 2 trials)
| Subgroup | REXULTI 2 mg | LSMD 95% CI (LL,UL) |
REXULTI 4 mg | LSMD 95% CI (LL,UL) |
Placebo | |||
|---|---|---|---|---|---|---|---|---|
| LS mean change from baseline (SE) | N | LS mean change from baseline (SE) | N | LS mean change from baseline (SE) | N | |||
| Overall Response /All patients |
-19 (1) | 359 | -5.5 (-8.5, -2.5) | -20 (1) | 359 | -6.7 (-9.7, -3.7) | -13 (1) | 358 |
| Sex | ||||||||
| Male | -18 (1) | 227 | -5.1 (-8.7, -1.5) | -19 (1) | 221 | -5.6 (-9.2, -1.9) | -13 (1) | 223 |
| Female | -19 (2) | 132 | -6.0 (-11.4,-0.6) | -22 (2) | 138 | -8.9 (-14.3,-3.5) | -13 (2) | 135 |
| Age Group | ||||||||
| >=17 - <40 > years |
-18 (2) | 195 | -3.9 (-8.4, 0.5) | -17 (2) | 179 | -2.9 (-7.3,-1.7) | -14 (2) | 175 |
| >=40 - <65> | -19 (2) | 163 | -7.0 (-11.2,-2.8) | -23(1) | 180 | -10.5 (-14.6,-6.5) | -12 (1) | 183 |
| >=65 years | 0 | 1 | NA | NA | 0 | NA | NA | 0 |
| Race | ||||||||
| White | -20 (1) | 233 | -4.4 (-8.2, -0.5) | -20 (1) | 218 | -4.6 (-8.5,-0.7) | -15 (1) | 226 |
| Black or African American | -14 (2) | 80 | -3.8 (-9.3, 1.8) | -18 (2) | 92 | -8.1 (-13.6,-2.7) | -10 (2) | 85 |
| Asian | -19 (4) | 26 | -11.3 (-23.8, 1.2) | -28 (4) | 28 | -19.7 (-31.8,-7.6) | -8 (5) | 26 |
| American Indian or Alaska Native | -20 (11) | 8 | 2.5 (-29.9, 34.8) | -23 (13) | 6 | -0.9 (-38.4,36.6) | -22 (12) | 6 |
| Native Hawaiian or Other Pacific Islander | NA | 0 | NA | 0 | 1 | NA | NA | 1 |
| Other | -25 (7) | 12 | -22.4 (-41.2, -3.5) | -16 (6) | 14 | -13.7 (-31.8, 4.5) | -3 (7) | 14 |
SE: standard error; LS mean: least-squares mean; LSMD: least-squares mean difference; CI: unadjusted confidence interval, NA not analyzed
Source: Company Clinical Trial Data
What are the possible side effects?
The most common side effect was weight gain.
REXULTI can increase the risk of death in elderly who have lost touch with reality (psychosis) due to confusion and memory loss (dementia).
REXULTI may increase suicidal thoughts or actions in teenagers or young adults, especially within the first few months of treatment.
What are the possible side effects (results of trials used to assess safety)?
Table 4. Adverse Events during Treatment in the Pooled Trials*
| Placebo (N=368) |
REXULTI | ||||
|---|---|---|---|---|---|
| 1 mg/day (N=120) |
2 mg/day (N=368) |
4 mg/day (N=364) |
ALL REXULTI (N=852) |
||
| Gastrointestinal Disorders | |||||
| Dyspepsia | 2% | 6% | 2% | 3% | 3% |
| Diarrhea | 2% | 0.8% | 3% | 3% | 3% |
| Investigations | |||||
| Weight Increased | 2% | 3% | 4% | 4% | 4% |
| Blood Creatine Phosphokinase Increased | 1% | 4% | 2% | 2% | 2% |
| Nervous System Disorders | |||||
| Akathisia | 5% | 4% | 5% | 7% | 6% |
| Tremor | 0.5% | 2% | 2% | 3% | 3% |
| Sedation | 0.8% | 2% | 2% | 3% | 2% |
* Adverse reactions that occurred in ≥ 2% of REXULTI-treated patients and greater incidence than in placebo-treated patients
Source: REXULTI Prescribing Information, Section 6 Table 9
Were there any differences in side effects among sex, race and age?
Subgroup analyses were conducted for sex, race, and age.
Were there any differences in side effects of the clinical trials among sex, race, and age groups?
The table below summarizes adverse events in the pooled trials by subgroup. The population represents the Safety population (1220 patients), which includes any patient who received at least one dose of trial drug.
Table 5. Subgroup Analysis of Adverse Events during Treatment in the Pooled Trials
| Subgroup | Total REXULTI | Placebo | Relative Risk | 95% CI | |||
|---|---|---|---|---|---|---|---|
| x (%) | Total, n | x (%) | Total, n | LL | UL | ||
| Any TEAEs* | 498 (58) | 852 | 216 (59) | 368 | 1.00 | 0.9 | 1.1 |
| Sex | |||||||
| Male | 321 (60) | 534 | 135 (59) | 229 | 1.02 | 0.90 | 1.16 |
| Female | 177 (56) | 318 | 81 (58) | 139 | 0.96 | 0.80 | 1.13 |
| Age Group | |||||||
| >=17 - <40> | 256 (58) | 445 | 109 (60) | 183 | 0.97 | 0.84 | 1.11 |
| >=40-<65> | 241 (60) | 404 | 107 (58) | 185 | 1.03 | 0.89 | 1.19 |
| >=65 | 1 (33) | 3 | NA | 0 | NA | NA | NA |
| Race | |||||||
| White | 279 (52) | 536 | 118 (51) | 231 | 1.02 | 0.88 | 1.18 |
| Black or African American | 144 (71) | 202 | 65 (72) | 90 | 0.99 | 0.85 | 1.15 |
| Asian | 43 (73) | 59 | 17 (65) | 26 | 1.11 | 0.81 | 1.54 |
| American Indian or Alaska Native | 3 (16) | 19 | 2 (33) | 6 | 0.47 | 0.10 | 2.20 |
| Native Hawaiian or Other Pacific Islander | 1 (100) | 1 | 1 (100) | 1 | 0.00 | 0.00 | 0.00 |
| Other | 28 (80) | 26 | 13 (93) | 14 | 0.86 | 0.69 | 1.07 |
*TEAEs=treatment emergent adverse events, NA not analyzed
Source: Company Clinical Trial Data
WHO WAS IN THE STUDIES?
Who participated in the clinical trials?
The FDA approved REXULTI based on evidence from two clinical trials that enrolled patients with schizophrenia. The trials were conducted in the United States, Canada, Europe, South America, and Africa.
The figure below summarizes how many men and women participated in the clinical trials. The population represents the Safety population (1220 patients), which includes any patient who received at least one dose of trial drug.
Figure 1. Baseline Demographics by Sex
Source: Company Clinical Trial Data
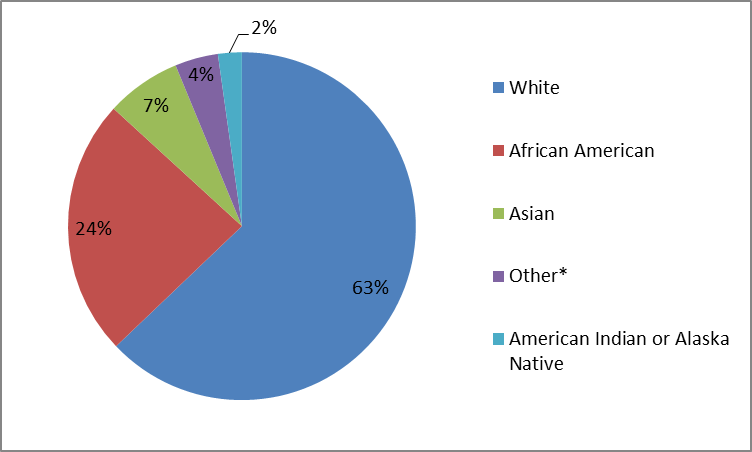
The figure and table below summarize how many patients by race participated in the clinical trials.
Figure 2. Baseline Demographics by Race
*= defined as any race not included in other categories
Source: Company Clinical Trial Data
Table 1. Baseline Demographics by Race
| Race | Number of Patients | Percentage of Patients |
|---|---|---|
| White | 767 | 63% |
| African American | 272 | 24% |
| Asian | 85 | 7% |
| Other* | 49 | 4% |
| American Indian or Alaska Native | 27 | 2% |
*=defined as any race not included in other categories
Source: Company Clinical Trial Data
The figure below summarizes how many patients by age participated in the clinical trials.
Figure 3. Baseline Demographics by Age
Source: Company Clinical Trial Data
Who participated in the trials?
The table below summarizes baseline demographics for the population that participated in the two trials. The population represents the Safety population (1220 patients), which includes any patient who received at least one dose of trial drug.
Table 6. Baseline Demographics for the Trials
| Demographic Parameters | REXULTI 1 mg (N=120) n (%) |
REXULTI 2 mg (N=368) n (%) |
REXULTI 4 mg (N=364) n (%) |
Placebo (N=368) n(%) |
Total (N=1220) n (%) |
|---|---|---|---|---|---|
| Sex | |||||
| Male | 77 (64) | 233 (63) | 224 (62) | 229 (62) | 763 (62) |
| Female | 43 (36) | 135 (37) | 140 (38) | 139 (38) | 457 (38) |
| Age | |||||
| Mean years (SD) | 39.1 (11.9) | 38.2 (10.6) | 39.7 (11.1) | 39.5 (10.8) | 39.1 (10.9) |
| Median (years) | 38.5 | 38 | 40 | 40 | 39 |
| Min, Max (years) | 18, 65 | 18, 65 | 18, 64 | 18, 64 | 18. 65 |
| Age Group | |||||
| >=17 - <40> | 63 (53) | 201 (55) | 181 (50) | 183 (50) | 628 (52) |
| >=40 - <65> | 56 (46) | 165 (44) | 183 (50) | 185 (50) | 589 (48) |
| >=65 years | 1 (1) | 2 (1) | 0 | 0 | 3 (<> |
| Race | |||||
| White | 75 (63) | 238 (65) | 223 (61) | 231 (63) | 767 (64) |
| Black or African American | 26 (22) | 84 (23) | 92 (25) | 90 (25) | 292 (24) |
| Asian | 5 (4) | 26 (7) | 28 (8) | 26 (7) | 85 (7) |
| American Indian or Alaska Native | 5 (4) | 8 (2) | 6 (1) | 6 (1) | 25 (2) |
| Native Hawaiian or Other Pacific Islander | 0 | 0 (0) | 1 (<> | 1 (<> | 2 (<> |
| Other | 9 (7) | 12 (3) | 14 (4) | 14 (4) | 49 (4) |
| Ethnicity | |||||
| Hispanic or Latino | 27 (23) | 34 (9) | 42 (11) | 40 (11) | 143 (12) |
| Not Hispanic or Latino | 93 (77) | 334 (91) | 320 (88) | 326 (88) | 1073 (88) |
|
Unknown |
0 (0) | 0 (0) | 2 (1) | 2 (1) | 4 (<> |
| Region | |||||
| United States | 44 (37) | 129 (35) | 131 (36) | 134 (36) | 438 (36) |
| Canada | 0 (0) | 1 (<> | 3 (1) | 2 (1) | 6 (<> |
| South America | 18 (15) | 26 (7) | 26 (7) | 24 (6) | 94 (8) |
| Europe | 55 (46) | 187 (50) | 181 (50) | 183 (50) | 606 (50) |
| Asia | 3 (2) | 25 (8) | 23 (6) | 25 (7) | 76 (6) |
How were the trials designed?
There were two trials that evaluated the benefit and side effects of REXULTI. In each trial, patients were randomly assigned to receive either REXULTI or placebo once daily for 6 weeks. Neither the patients nor the health care providers knew which treatment was being given until after the trials were completed.
The trials measured overall improvement in the symptoms of schizophrenia.
How were the trials designed?
Two randomized, placebo-controlled trials were conducted in adult patients who met Diagnostic and Statistical Manual of Mental Disorders, 4th Edition, Text Revision, (DSM-IV-TR) criteria for schizophrenia.
The primary endpoint was the change from baseline to Week 6 in the Positive and Negative Syndrome Scale (PANSS) total score. The PANSS is a 30-item scale that measures positive symptoms of schizophrenia (7 items), negative symptoms of schizophrenia (7 items), and general psychopathology (16 items), each rated on a scale of 1 (absent) to 7 (extreme); the total PANSS scores range from 30 to 210.
GLOSSARY
CLINICAL TRIAL: Voluntary research studies conducted in people and designed to answer specific questions about the safety or effectiveness of drugs, vaccines, other therapies, or new ways of using existing treatments.
COMPARATOR: A previously available treatment or placebo used in clinical trials that is compared to the actual drug being tested.
EFFICACY: How well the drug achieves the desired response when it is taken as described in a controlled clinical setting, such as during a clinical trial.
PLACEBO: An inactive substance or “sugar pill” that looks the same as, and is given the same way as, an active drug or treatment being tested. The effects of the active drug or treatment are compared to the effects of the placebo.
SUBGROUP: A subset of the population studied in a clinical trial. Demographic subsets include sex, race, and age groups.
PRESCRIBING INFORMATION
-
- Sex: REXULTI worked similarly in men and women.
- Race: REXULTI worked similarly in all races studied.
- Age: REXULTI worked similarly in all age groups studied.
- Sex: The risk of overall side effects appeared to be similar in men and women.
- Race: The risk of overall side effects appeared to be similar in all races studied.
- Age: The risk of overall side effects appeared to be similar in all age groups studied.