Form FDA 5066 - Registration of Cosmetic Product Facility
Electronic Submissions
- Cosmetics Direct: You can submit your facility registrations using the Cosmetics Direct electronic submission portal at: https://direct.fda.gov.
- Electronic Submissions Gateway Next Generation (ESG NextGen): As an alternative, facility registrations may be submitted online through FDA’s ESG NextGen, or any Structured Product Labeling (SPL) authoring software including SPL Xforms. The FDA ESG NextGen system requires users to apply for a free account before submitting data, a process which can take one to three weeks. FDA therefore urges registrants to apply for ESG NextGen accounts well in advance of the statutory deadline for data submission.
Paper Submissions
Instructions to Complete Form FDA 5066 – Registration of Cosmetic Product Facility
Please mail the completed form to FDA’s Registration and Listing of Cosmetic Product Facilities and Products Program at: Food and Drug Administration, Office of Cosmetics and Colors, Registration and Listing of Cosmetic Product Facilities and Products Program, 5001 Campus Drive, CPK1, Room 1B-046, College Park, MD 20740-3835 or email it to RLC-PaperSubmissions@fda.hhs.gov.
The paper Form FDA 5066 contains five sections (Sections I through V). All required fields on Form FDA 5066 are designated by asterisks (*).
For additional details and instructions, please refer to the FDA Guidance for Industry: Registration and Listing of Cosmetic Product Facilities and Products.
Section I – Document type
Select one of the following document types:
- Initial Registration – Form FDA 5066 submitted to FDA for the first time to provide information about a Cosmetic Product Facility under this facility name and location.
- Amended (Changes to Registration) – Form FDA 5066 submitted to FDA to make changes to a registered Cosmetic Product Facility.
- Amended (Cancellation of Registration) – Form FDA 5066 submitted to FDA for a previously registered facility that no longer manufacturers or processes cosmetic products.
- Biennial Registration Renewal – Form FDA 5066 submitted to FDA to renew a facility registration with changes. Form FDA 5066 submitted to FDA to renew a facility registration with changes. Every person who is required to register a facility must renew such registration biennially (i.e., every two years) (section 607(a)(2) of the FD&C Act).
- Abbreviated Registration Renewal – Form FDA 5066 submitted to FDA to renew a facility registration without any changes. By selecting this document type, you are certifying that no changes have been made to your registration since the previous registration was submitted.
Section II – Registration
Provide the following required information:
- Facility name
- Facility FEI (FDA Establishment Identifier) Number1
- Street address
- City
- State or province
- Zip/postal code
- Country (if other than USA)
- Facility email
- Facility phone number (including country/area code)
- Name of the owner and/or operator of the facility
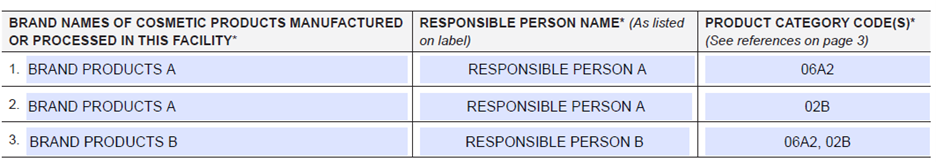
- Brand names of cosmetic products manufactured or processed in this facility (i.e., all brand names under which cosmetic products manufactured or processed in the facility are sold)
- Responsible person name2 (as listed on label) for each cosmetic product brand name
- Product category code(s)3 (see Cosmetic Product Categories and Codes)
FDA also requests that the following additional optional information be provided:
- Whether the facility registration is for a small business (optional registration)
- Parent company name (if applicable)
- Facility DUNS Number
Notes:
1 The facility registration number is the FDA Establishment Identifier (FEI). To facilitate the registration process, the owner or operator of a facility will need to obtain an FEI number before submitting the facility registration. To determine if an entity already has an FEI number, please refer to the FEI Search Portal. If your firm does not have an FEI number assigned by FDA, see How can I request an FEI? at FEI Search Portal (fda.gov).
2 Responsible Person as defined in section 604(4) of the Federal Food Drug, and Cosmetic Act (FD&C Act), means the manufacturer, packer, or distributor of a cosmetic product whose name appears on the label of such cosmetic product in accordance with section 609(a) of the FD&C Act or section 4(a) of the Fair Packaging and Labeling Act.
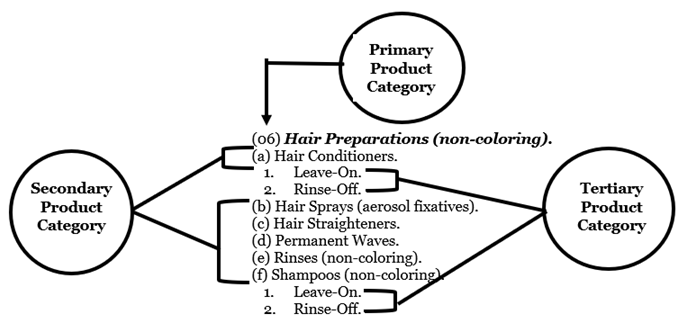
3 A product category code may have multiple product category components (e.g., primary, secondary, and tertiary product categories). Each primary product category (01 – 16, but not 17) has multiple secondary product categories (A through K for 03). A secondary product category can have several tertiary product categories (e.g., 1. Leave-on and 2. Rinse-off). For example, a hair preparation (non-coloring) product that is a hair conditioner and a rinse-off product would have a product category code of 06A2 (06-Hair Preparations (non-coloring), A-Hair Conditioners, 2-Rinse-Off). A facility registration can have one or more tertiary product category(ies) that is “leave-on” and/or “rinse-off” since a brand name can include multiple types of products (e.g., product categories 06A1 and/or 06A2).
Additional information can be found at: Cosmetic Product Categories and Codes
Product Category Codes Diagram:
Product Category Code Examples:
Example 1: Hair Conditioner; Rinse-off
Cosmetic Product Category Code: 06A2
| Primary Product Category | Secondary Product Category | Tertiary Product Category |
| (06) Hair Preparations (non-coloring) | A- Hair Conditioners | 2- Rinse Off |
Example 2: Bubble Bath
Cosmetic Product Category Code: 02B
| Primary Product Category | Secondary Product Category | Tertiary Product Category |
| (02) Bath Preparations | B- Bubble Baths | Not applicable |
Multiple product category codes may be listed on a single or separate line(s), for example, if a brand name of cosmetic products manufactured or processed at the facility includes multiple types of cosmetic products.
Additional FDA Form 5066, page 1 can be attached by the submitter to give additional information for primary, secondary, and tertiary cosmetic product categories; responsible person names; and brand names.
Section III – U.S. agent contact information (for foreign facilities)
For foreign facilities, provide the following required information:
- U.S. agent name
- U.S. agent email (if not available, enter “N/A”)
- U.S. agent phone number (including country/area code)
If applicable, a phone extension can be provided for the U.S. agent phone number.
Section IV – Confirmation statement
FDA requests that individuals submitting facility registration information to attest to the accuracy and veracity of the information submitted.
Section V – Additional contact information for authorized agent
Provide the following optional information:
- Additional contact name
- Additional contact email
- Additional contact phone number (including country/area code)
- Additional contact phone extension
Before submitting Form FDA 5066, please check your submission to ensure that all continuation pages and/or attachments have been included. The mailing and email addresses for the submission can be found at the top of this webpage.