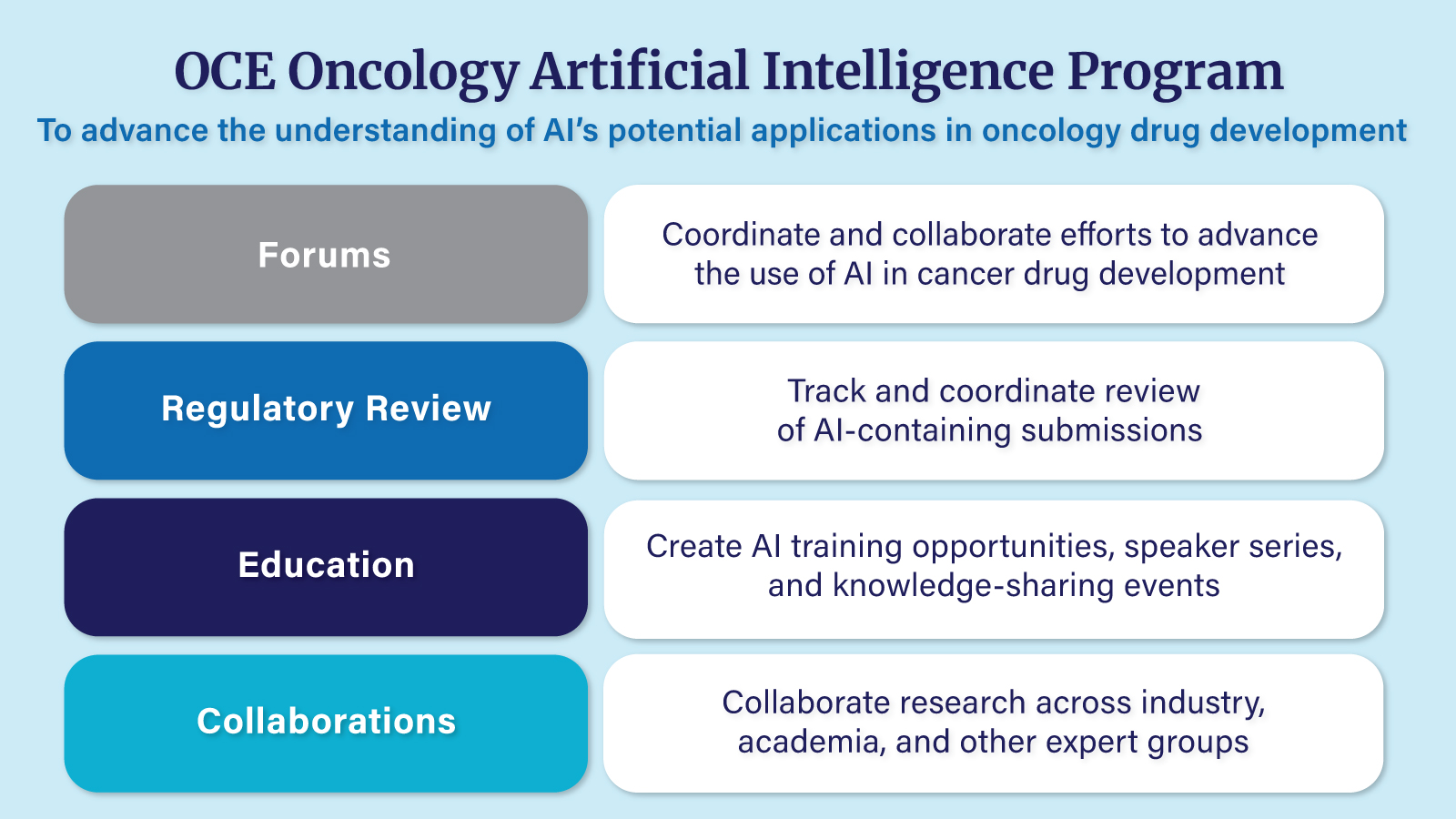
OCE Oncology Artificial Intelligence Program

The FDA Oncology Center of Excellence (OCE) began the Oncology Artificial Intelligence (AI) Program in 2023 in response to growing interest from oncology reviewers, increased engagement with drug development sponsors, and the expanding use of AI in cancer drug development. The Oncology AI Program aims to advance the understanding and application of AI in oncology drug development. It offers specialized training for reviewers on leading AI methodologies, supports regulatory science research to deepen AI-related regulatory knowledge, and streamlines the review process for applications that incorporate AI technologies.
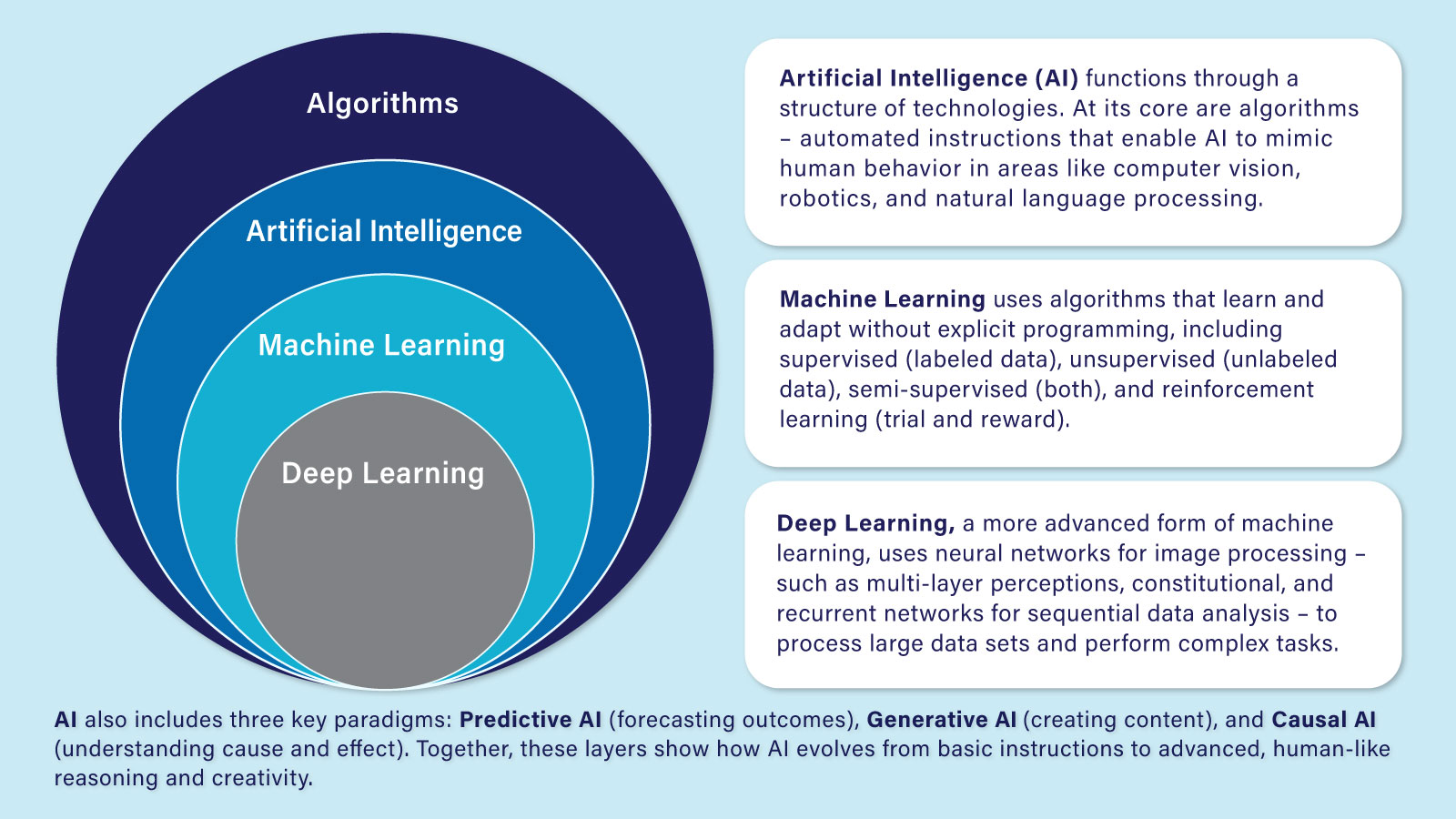
What is Artificial Intelligence?
Artificial Intelligence or AI refers to a machine-based system that can, for a given set of human-defined objectives, make predictions, recommendations, or decisions influencing real or virtual environments. AI systems use machine- and human-based inputs to perceive real and virtual environments; abstract such perceptions into models through analysis in an automated manner; and use model inference to formulate options for information or action.
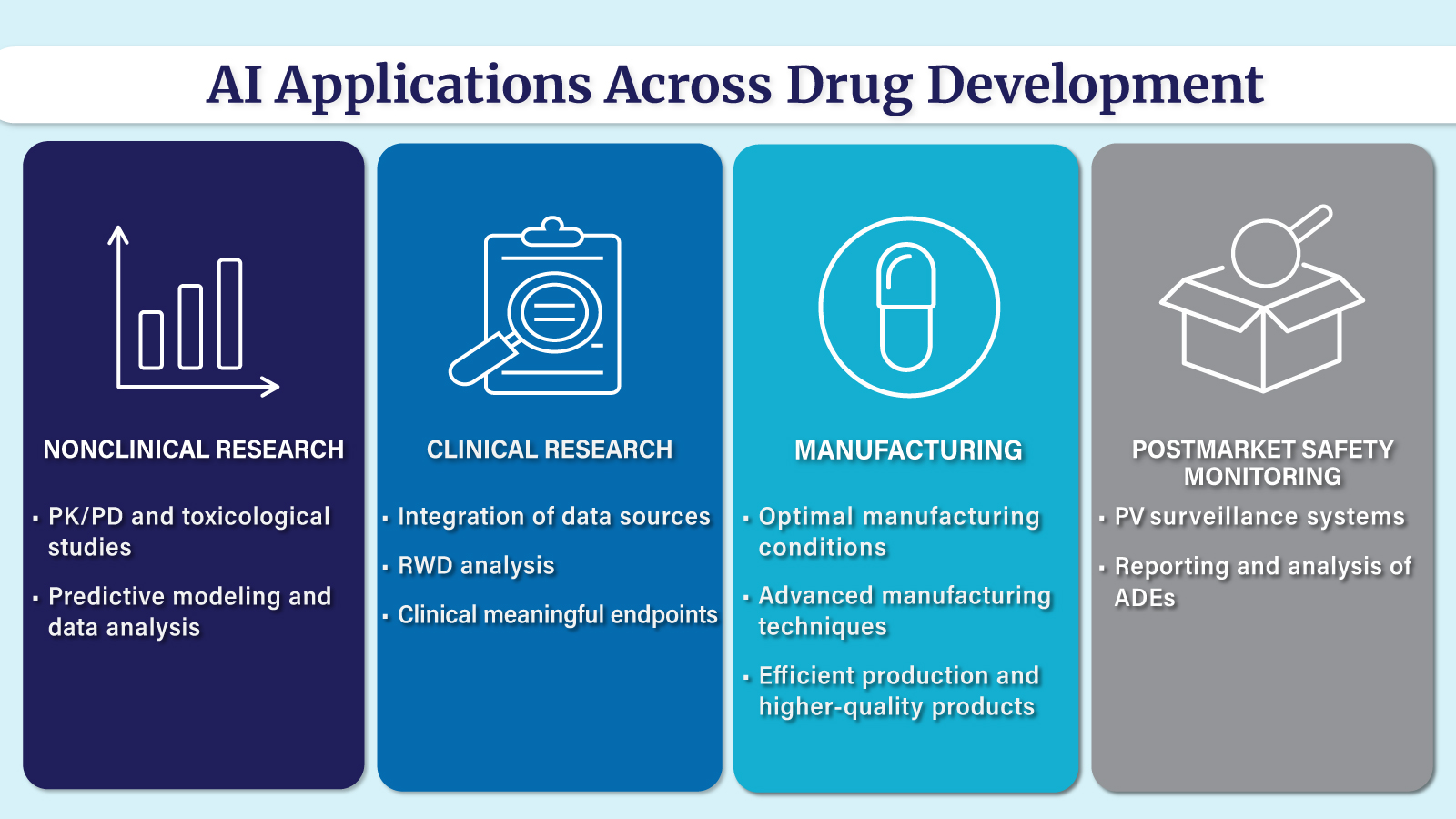
What is AI’s role in healthcare?
AI is widely recognized for its transformative potential in the medical field, offering the ability to accelerate medical product development, enhance patient care, and significantly augment the skills and decision-making capabilities of healthcare practitioners. Through its innovative applications, AI can streamline processes, improve outcomes, and ultimately shape the future of healthcare.
In 2017, the OCE led an Integrated Project Team (IPT), directed by oncology reviewers and leadership, to develop a data platform. The platform integrated data from approximately 30 internal FDA systems and external Department of Health and Human Services (HHS) websites. The success of the platform was attributed to agile sprint development and a focus on user-centered design. The data generated was reliable and supported responses to external stakeholders (including congressional inquiries), regulatory reviews, and research activities. The foundational elements of this platform were adopted by other systems within the Agency (e.g., CDEROne Analytics).
Publications
Azenkot, T., Rivera, D. R., Stewart, M. D., & Patel, S. P. (2025). Artificial intelligence and machine learning innovations to improve design and representativeness in oncology clinical trials. American Society of Clinical Oncology Educational Book, 45(3), e473590.
Presentations
ASCO Annual Meeting 2025
- Session Title: Artificial Intelligence Approaches Across the Drug Development Continuum in Oncology: A Regulatory Perspective
- Presentation Title: AI Approaches Across the Drug Development Continuum in Oncology: A Regulatory Perspective
DIA Global Annual Meeting 2025
- Session: Modernizing Evidence in Oncology: Real World Data and Artificial Intelligence in Clinical Drug Development
FDA Guidance
Artificial Intelligence-Enabled Device Software Functions: Lifecycle Management and Marketing Submission Recommendations. Draft Guidance for Industry and Food and Drug Administration Staff. January 2025.
Considerations for the Use of Artificial Intelligence to Support Regulatory Decision-Making for Drug and Biological Products. Draft Guidance for Industry and Other Interested Parties. January 2025.
Related Links
- Artificial Intelligence and Medical Products | FDA
- Artificial Intelligence for Drug Development | FDA
- Artificial Intelligence Program: Research on AI/ML-Based Medical Devices | FDA
Oncology AI Team
OCE Executive Lead: Tamy Kim, PharmD
Oncology AI Co-Lead: Jeevan Puthiamadathil, MD
Oncology AI Co-Lead: Michael Barbato, MD
Oncology AI Regulatory Program Manager: Esther Park, PharmD
Contact Information
For AI-related questions specific to existing INDs or future marketing applications, please submit a meeting request to the appropriate Office of Oncologic Diseases review division. OCE will coordinate with the division to provide responses to your specific inquiries.
For broader AI-focused questions not related to a specific application, please email: OncologyAI@fda.hhs.gov.