Precision Medicine
Most medical treatments are designed for the "average patient" as a one-size-fits-all-approach, which may be successful for some patients but not for others. Precision medicine, sometimes known as "personalized medicine" is an innovative approach to tailoring disease prevention and treatment that takes into account differences in people's genes, environments, and lifestyles. The goal of precision medicine is to target the right treatments to the right patients at the right time.
Advances in precision medicine have already led to powerful new discoveries and FDA-approved treatments that are tailored to specific characteristics of individuals, such as a person's genetic makeup, or the genetic profile of an individual's tumor. Patients with a variety of cancers routinely undergo molecular testing as part of patient care, enabling physicians to select treatments that improve chances of survival and reduce exposure to adverse effects.
Next Generation Sequencing (NGS) Tests
Precision care will only be as good as the tests that guide diagnosis and treatment. Next Generation Sequencing (NGS) tests are capable of rapidly identifying or 'sequencing' large sections of a person's genome and are important advances in the clinical applications of precision medicine.
Patients, physicians and researchers can use these tests to find genetic variants that help them diagnose, treat, and understand more about human disease.
The FDA's Role in Advancing Precision Medicine
The FDA is working to ensure the accuracy of NGS tests, so that patients and clinicians can receive accurate and clinically meaningful test results.
The vast amount of information generated through NGS poses novel regulatory issues for the FDA. While current regulatory approaches are appropriate for conventional diagnostics that detect a single disease or condition (such as blood glucose or cholesterol levels), these new sequencing techniques contain the equivalent of millions of tests in one. Because of this, the FDA has worked with stakeholders in industry, laboratories, academia, and patient and professional societies to develop a flexible regulatory approach to accommodate this rapidly evolving technology that leverages consensus standards, crowd-sourced data, and state-of-the-art open-source computing technology to support NGS test development. This approach will enable innovation in testing and research, and will speed access to accurate, reliable genetic tests.
Streamlining FDA's Regulatory Oversight of NGS Tests
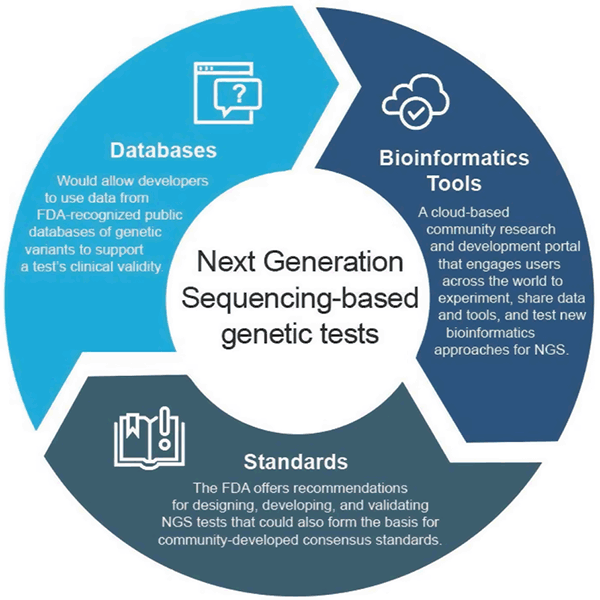
In April 2018, the FDA issued two final guidances that recommend approaches to streamline the submission and review of data supporting the clinical and analytical validity of NGS-based tests. These recommendations are intended to provide an efficient and flexible regulatory oversight approach: as technology advances, standards can rapidly evolve and be used to set appropriate metrics for fast growing fields such as NGS. Similarly, as clinical evidence improves, new assertions could be supported. This adaptive approach would ultimately foster innovation among test developers and improve patients' access to these new technologies.
Clinical Databases Guidance
The final guidance "Use of Public Human Genetic Variant Databases to Support Clinical Validity for Genetic and Genomic-Based In Vitro Diagnostics" allows developers to use data from FDA-recognized public databases of genetic variants to help support a test's clinical validity and outlines how database administrators can seek recognition for their databases if they meet certain quality recommendations. This approach incentivizes data sharing and provides a more efficient path to market.
More information about database recognition is available on the FDA’s Recognition of Public Human Genetic Variants Databases webpage.
Analytical Validation Guidance
The final guidance "Considerations for Design, Development, and Analytical Validation of Next Generation Sequencing (NGS) – Based In Vitro Diagnostics (IVDs) Intended to Aid in the Diagnosis of Suspected Germline Diseases" offers recommendations for designing, developing and validating NGS tests. The guidance also encourages community engagement in developing NGS-related standards by standards developing organizations (SDOs) since standards can more rapidly evolve with changes in technology and knowledge and can therefore be used to set appropriate metrics such as specific performance thresholds for fast growing fields such as NGS.
FDA's Bioinformatics Platform
The FDA created precisionFDA, a cloud-based community research and development portal that engages users across the world to share data and tools to test, pilot, and validate existing and new bioinformatics approaches to NGS processing. Individuals and organizations in the genomics community can find more information and sign up to participate at http://precision.fda.gov.
Questions?
Please contact us with any questions about the FDA's precision medicine efforts at OIRPMgroup@fda.hhs.gov.
What FDA Officials Are Saying
NGS Final Guidances
- Use of Public Human Genetic Variant Databases to Support Clinical Validity for Genetic and Genomic-Based In Vitro Diagnostics - Guidance for Stakeholders and Food and Drug Administration Staff (PDF - 444KB)
- Considerations for Design, Development, and Analytical Validation of Next Generation Sequencing (NGS) - Based In Vitro Diagnostics (IVDs) Intended to Aid in the Diagnosis of Suspected Germline Diseases - Guidance for Stakeholders and Food and Drug Administration Staff (PDF - 869KB)
Reports and Discussion Papers
- Optimizing FDA’s Regulatory Oversight of Next Generation Sequencing Diagnostic Tests (PDF - 424KB)
- Report: Paving the Way for Personalized Medicine (Archived)
Past Meetings
- 07/27/16: Webinar - Next Generation Sequencing (NGS) Draft Guidances: Implications for Patients and Providers
- 07/27/16: Webinar - Next Generation Sequencing (NGS) Draft Guidances: Technical and Regulatory Aspects
- 11/13/2015: Public Workshop - Use of Databases for Establishing the Clinical Relevance of Human Genetic Variants
- 11/12/2015: Standards Based Approach to Analytical Performance Evaluation of Next Generation Sequencing In Vitro Diagnostic Tests
- 02/20/2015: Optimizing FDA’s Regulatory Oversight of Next Generation Sequencing Diagnostic Tests Public Workshop

