Vascular-Based Biomarkers For Evaluation of Biological Product Safety and Efficacy
Felice D’Agnillo, Ph.D.
Office of Blood Research and Review
Division of Blood Components and Devices
Laboratory of Biochemistry and Vascular Biology
Biosketch
Felice D’Agnillo is a Principal Investigator in the Office of Blood Research and Review (OBRR) in the Center for Biologics Evaluation and Research (CBER). Dr. D’Agnillo heads a vascular biology research program that studies the interactions of transfusion products and microbial pathogens with the vascular system. Dr. D’Agnillo, an expert in hemoglobin- and heme-based products, also serves as a Chemistry, Manufacturing, and Controls (CMC) reviewer of regulatory applications for transfusion-related biologics.
Dr. D’Agnillo obtained his B.Sc. Honors and Ph.D. degrees in Physiology from McGill University, Montreal, Canada, where he began to investigate strategies to improve the safety of hemoglobin-based oxygen carriers (HBOCs). His doctoral studies led to a patent for a novel hemoglobin-antioxidant polymer to counter free radical reactions that mediate some of the toxicities of HBOCs. Dr. D’Agnillo then joined the laboratory of Dr. Abdu Alayash at CBER/FDA as a National of Institutes of Health Fogarty Fellow, where he continued to study the impact of hemoglobin oxidative reactions in biological settings. He was subsequently appointed as a Principal Investigator to lead a vascular biology program at CBER, where he twice received the CBER Scientific Achievement Award.
His primary areas of interest include: 1) preclinical safety evaluation of blood-derived products using biomarkers of vascular and oxidative stress in animal models of endothelial dysfunction and 2) vascular biomarkers of microbial pathogenesis. The latter research reflects the need to better understand how microbial pathogens in contaminated biologics or from pandemic and emerging infectious diseases or acts of bioterrorism (e.g. COVID-19, influenza, anthrax) influence the function and integrity of the vascular system. Together, these studies aim to improve the science-based regulation of current and anticipated therapies and promote the design of safer and more efficacious products and novel diagnostics.
General Overview
Vascular endothelial cells lining the interior surface of blood vessels can interact directly with transfused biological products or microbial pathogens introduced intravascularly as product contaminants or by infectious diseases. Endothelial cells play a major role in regulating blood clotting, inflammation, vascular tone, and new blood vessel formation. Our research program employs in vitro human endothelial cell culture systems and animal models to study how biological products or pathogens can alter the function and integrity of the vascular system. The overall goal of these studies is to improve the science-based regulation of current and anticipated therapies and to promote the design of safer and more efficacious products and novel diagnostics. Our current research activities focus on two main areas: (1) preclinical safety and efficacy evaluation of blood-derived products, including pathogen-inactivated RBCs and hemoglobin-based therapeutics using biomarkers of vascular integrity and function in animal models of endothelial dysfunction; and (2) vascular biomarkers of microbial pathogenesis.
Preclinical safety and efficacy analyses are being conducted on blood-derived products, including pathogen-inactivated RBCs and hemoglobin-based therapeutics. In the case of hemoglobin-based therapeutics, these agents are being developed as an oxygen bridge for use in resuscitation from traumatic shock and elective surgery in situations when RBCs are not available, contraindicated, or refused based on religious grounds. Given their desirable storage and compatibility characteristics, hemoglobin-based therapeutics may be particularly useful in military settings and in situations that impact the blood supply, such as major terrorist attacks or natural disasters. In the case of pathogen-inactivated RBCs, these transfusion products are derived using existing and novel pathogen reduction technologies for whole blood that have a yet-to-be-defined impact on RBC safety and quality. To address safety and efficacy issues relevant to the development of these products, we are investigating preclinical animal transfusion models and predictive biomarkers of product safety, with particular emphasis on vascular hemostasis. The insight gained from these studies will improve the regulatory decision-making process for existing blood-derived products and contribute to the rational design of next generation transfusion therapies with reduced vascular-related side effects.
Microbial pathogens in contaminated biological products or encountered through infection represent a major threat to the vascular system. Bacterial and viral diseases are often characterized by vascular endothelial dysfunction, microvascular leakage, and inflammation that can progress to loss of organ function and death. Our research focuses on understanding the molecular mechanisms by which microbial agents trigger endothelial dysfunction and injury. To do so, we use animal models and vascular endothelial cell culture systems. These efforts have included the identification of relevant vascular biomarkers triggered in response to pathogens from Gram positive and Gram negative bacteria (e.g., lipopolysaccharide, Bacillus anthracis) and viruses (e.g., influenza, SARS-CoV-2). These studies will help inform the identification of much needed surrogate markers of disease progression or recovery. Further, our research will facilitate the discovery and evaluation of improved or novel medical countermeasures and diagnostics.
Scientific Overview
Vascular endothelial cells serve as first line responders to circulating biological products or microbial pathogens by virtue of their direct contact with the bloodstream. The main focus of our research is to study the mechanisms by which biological products or pathogens alter vascular integrity and function, using both endothelial cell culture systems and animal models. Our overall goal is to advance the science-based regulation and evaluation of current and anticipated products and to promote the discovery of novel or improved therapies. The two main areas of investigation include (1) preclinical safety evaluation of blood-derived products using biomarkers of vascular integrity and function in animal models of endothelial dysfunction and (2) vascular biomarkers of microbial pathogenesis.
Blood-derived products, including pathogen-inactivated RBCs and hemoglobin-based therapeutics, have the potential to significantly enhance public health in a number of medical settings. These include trauma resuscitation, surgery, and organ preservation. In the case of hemoglobin-based therapeutics, pre-existing endothelial dysfunction associated with underlying disease (e.g., diabetes, atherosclerosis, sepsis, advanced age) has been cited as a major factor in the adverse reactions to these products. Moreover, concerns have been raised over the lack of sensitivity and specificity of standard histopathology and clinical chemistry indices for assessing the safety of this unique class of products. To address this, our research has focused on developing vascular biomarkers and animal transfusion models of vascular stress, including hyperglycemia, high cholesterol, aging, and endotoxin exposure. Using an exchange transfusion model in guinea pigs, we have reported that a polymerized hemoglobin product enhances oxidative, inflammatory, and organ injury in brain, heart, kidney, and lungs. Ongoing studies examine how hemoglobin products may exacerbate the pathophysiology of endotoxemia, which is critical for understanding the safety of these products in settings of trauma or surgery with concomitant sepsis. Recent studies also investigate the mechanisms underlying the safety profile of hemoglobin-derived products in a guinea pig model of advanced aging, as this represents a key recognized cardiovascular risk factor linked to adverse clinical responses to this product class.
These preclinical transfusion models and biomarkers are also being applied to evaluating the safety and efficacy of pathogen-inactivated RBCs preparations. The studies may in turn provide insight on how to improve existing technologies and/or design novel pathogen reduction methods. The overall aim of these studies focuses on in vivo RBC safety and tissue oxygen restoration by pathogen-reduced RBCs following acute blood loss in guinea pigs.
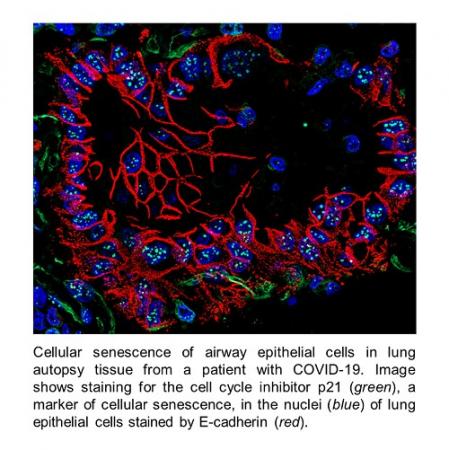
Exposure to microbial pathogens in contaminated biological products or resulting from emerging and re-emerging infectious diseases (e.g., influenza, SARS-CoV-2) represents a major threat to the vascular system. Bacterial and viral infections are often characterized by vascular endothelial dysfunction, microvascular leakage, and inflammation that can progress to loss of organ function and death. We have identified vascular endothelium as a key target for bacterial-and viral-derived pathogens that can trigger barrier dysfunction, inflammation, and thrombosis. These findings provide useful paradigms for evaluating and designing therapeutics or enhancing vascular endothelial protection against microbial pathogens. We have examined vascular biomarkers to track disease progression during viral infections such as influenza, and ongoing collaborative human autopsy studies have identified key markers of lung damage and endothelial dysfunction that provide direct mechanistic insights into the lung pathophysiology of COVID-19. These studies have also provided critical mechanistic insight on risk factors for developing severe COVID-19 disease that may also help explain the long-term health effects experienced by some COVID-19 survivors.
Important Links
- Innovation and Regulatory Science- Research Summary: FDA comparison of characteristics of hemoglobin-based oxygen carriers could guide development of safe and effective artificial blood products
- ORCID ID: 0000-0002-4134-9490
- https://www.nih.gov/news-events/news-releases/lung-autopsies-covid-19-patients-reveal-treatment-clues
Publications
- Blood Transfus 2024 Jul-Aug;22(4):316-27
Ultraviolet light and riboflavin accelerates red blood cell dysfunction in vitro and in a guinea pig transfusion model.
Baek JH, Shin HKH, Xu F, Zhang X, Williams MC, Gao Y, Vostal JG, Buehler PW, Villa C, D'Agnillo F - Toxicol In Vitro 2024 May;97:105793
In vitro test methods for evaluating high molecular weight polyethylene oxide polymer induced hemolytic and thrombotic potential.
Kim D, Natu R, Malinauskas R, Baek JH, Buehler P, Feng X, Qu H, Pinto J, Xu X, Herbertson L - Nat Commun 2023 Jun 9;14(1):3393
Intranasal or airborne transmission-mediated delivery of an attenuated SARS-CoV-2 protects Syrian hamsters against new variants.
Stauft CB, Selvaraj P, D'Agnillo F, Meseda CA, Liu S, Pedro CL, Sangare K, Lien CZ, Weir JP, Starost MF, Wang TT - Front Med 2023 Jun 13;10:1158359
Renal glomerular and tubular responses to glutaraldehyde- polymerized human hemoglobin.
Williams MC, Zhang X, Baek JH, D'Agnillo F - Int J Pharm 2023 Feb 5;632:122557
Determining critical overlap concentration of polyethylene oxide to support excipient safety assessment of opioid products.
Smith WC, Qu H, Zheng K, Hyen Baek J, Gao Y, Buehler PW, Feng X, Xu X - Int J Mol Sci 2022 Dec 29;24(1):558
HIF-1alpha-dependent metabolic reprogramming, oxidative stress, and bioenergetic dysfunction in SARS-CoV-2-infected hamsters.
Jana S, Heaven MR, Stauft CB, Wang TT, Williams MC, D'Agnillo F, Alayash AI - Nat Commun 2022 Nov 10;13(1):6792
Intranasal delivery of a rationally attenuated SARS-CoV-2 is immunogenic and protective in Syrian hamsters.
Liu S, Stauft CB, Selvaraj P, Chandrasekaran P, D'Agnillo F, Chou CK, Wu WW, Lien CZ, Meseda CA, Pedro CL, Starost MF, Weir JP, Wang TT - Front Physiol 2022 Mar 21;13:845347
ZOOMICS: comparative metabolomics of red blood cells from guinea pigs, humans, and non-human primates during refrigerated storage for up to 42 days.
Bertolone L, Shin HKH, Baek JH, Gao Y, Spitalnik SL, Buehler PW, D'Alessandro A - Sci Transl Med 2021 Nov 17;13(620):eabj7790
Lung epithelial and endothelial damage, loss of tissue repair, inhibition of fibrinolysis, and cellular senescence in fatal COVID-19.
D'Agnillo F, Walters KA, Xiao Y, Sheng ZM, Scherler K, Park J, Gygli S, Rosas LA, Sadtler K, Kalish H, Blatti CA 3rd, Zhu R, Gatzke L, Bushell C, Memoli MJ, O'Day SJ, Fischer TD, Hammond TC, Lee RC, Cash JC, Powers ME, O'Keefe GE, Butnor KJ, Rapkiewicz AV, Travis WD, Layne SP, Kash JC, Taubenberger JK - Am J Pathol 2020 Oct;190(10):2095-110
Structural integrity of the alveolar-capillary barrier in cynomolgus monkeys challenged with fully virulent and toxin-deficient strains of Bacillus anthracis.
D'Agnillo F, Zhang X, Williams MC - Front Physiol 2020 Oct 23;11:593841
ZOOMICS: comparative metabolomics of red blood cells from old world monkeys and humans.
Bertolone L, Shin HK, Stefanoni D, Baek JH, Gao Y, Morrison EJ, Nemkov T, Thomas T, Francis RO, Hod EA, Zimring JC, Yoshida T, Karafin M, Schwartz J, Hudson KE, Spitalnik SL, Buehler PW, D'Alessandro A - Bioconjug Chem 2018 May 16;29(5):1560-75
Comprehensive biochemical and biophysical characterization of hemoglobin-based oxygen carrier therapeutics: all HBOCs are not created equally.
Meng F, Kassa T, Jana S, Wood F, Zhang X, Jia Y, D'Agnillo F, Alayash AI - Toxicology 2018 Jun 1;402-3:37-49
Reversible renal glomerular dysfunction in guinea pigs exposed to glutaraldehyde-polymerized cell-free hemoglobin.
D'Agnillo F - PLoS One 2016 Aug 9;11(8):e0160875
Poly(I:C) induces human lung endothelial barrier dysfunction by disrupting tight junction expression of claudin-5.
Huang LY, Stuart C, Takeda K, D'Agnillo F, Golding B - Toxics 2016 Mar;4(1):4010006
Transcriptional suppression of renal antioxidant enzyme systems in guinea pigs exposed to polymerized cell-free hemoglobin.
Rentsendorj O, Zhang X, Williams MC, Buehler PW, D'Agnillo F - J Pathol 2016 Jan;238(1):85-97
1918 pandemic influenza virus and Streptococcus pneumoniae coinfection results in activation of coagulation and widespread pulmonary thrombosis in mice and humans.
Walters KA, D'Agnillo F, Sheng ZM, Kindrachuk J, Schwartzman LM, Keustner RE, Chertow DS, Golding BT, Taubenberger JK, Kash JC - Toxicology 2015 Jul 3;333:89-99
Sodium nitrite potentiates renal oxidative stress and injury in hemoglobin exposed guinea pigs.
Baek JH, Zhang X, Williams MC, Hicks W, Buehler PW, Felice D