"Covered" Tobacco Products and Roll-Your-Own/ Cigarette Tobacco Labeling and Warning Statement Requirements
On this page:
- Required Warning Statements
- Packages that are Too Small to Display the Required Warning Statement
- Zero-Nicotine: Self-certification and Alternative Required Warning Statement
- Other Labeling Requirements
Required Warning Statements
The United States District Court for the District of Columbia recently issued an order vacating the health warning requirements for cigars and pipe tobacco set forth in 21 CFR §§ 1143.3 and 1143.5 and remanding the Final Deeming Rule’s warning requirements for cigars and pipe tobacco back to the Agency. See Order, Cigar Ass’n of Am. v. U.S. Food & Drug Admin., No. 1:16-cv-01460 (D.D.C. September 11, 2020). Although the requirement has been vacated, cigar and pipe tobacco firms may choose to voluntarily comply with these health warning provisions. FDA will continue to enforce the other requirements it was already enforcing for cigars and pipe tobacco under the FD&C Act and its implementing regulations, such as not selling these products to individuals under 21 years of age or marketing them as modified risk tobacco products without an FDA order.
The court’s order does not enjoin FDA from enforcing the health warning requirements for other product categories, including Electronic Nicotine Delivery Systems (ENDS) products, hookah tobacco, and cigarette tobacco and roll-your-own tobacco products.
A “covered tobacco product” is any tobacco product deemed under the deeming final rule to be subject to chapter IX of the Federal Food, Drug, and Cosmetic Act, but excludes any component or part that is not made or derived from tobacco. For example, cigars, liquid nicotine, hookah/waterpipe tobacco, and pipe tobacco would be considered “covered tobacco products,” while vaporizers or pipes that are not pre-loaded with tobacco or a tobacco-derived substance would not.
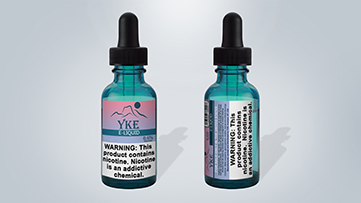
By the compliance date of August 10, 2018, roll-your-own tobacco, cigarette tobacco, or covered tobacco products (except cigars and pipe tobacco) manufactured, packaged, sold, offered for sale, distributed, or imported for sale or distribution within the United States must bear the required warning statement on the tobacco product package label, per 21 CFR § 1143.3(a)(1).1 (Cigar required warnings are described under 21 CFR § 1143.5.)
"WARNING: This product contains nicotine. Nicotine is an addictive chemical."
This required warning statement must also meet certain requirements, with respect to font, text, size, placement and formatting of the warning statement on the package labels. That is, the required warning statement on package labels must also appear directly on the package, and be clearly visible underneath any cellophane or other clear wrapping, per 21 CFR § 1143.3(a)(2), as follows:
- Be located in a conspicuous and prominent place on the two "principal display panels" of the package;
- Comprise at least 30 percent of each of the principal display panels;
- Be printed in at least 12-point font size and must occupy the greatest possible proportion of the warning label area set aside for the required text;
- Be printed in conspicuous and legible Helvetica bold or Arial bold type or other similar sans serif fonts and in black text on a white background or white text on a black background in a manner that contrasts by typography, layout, or color, with all other printed material on the package;
- Be capitalized and punctuated as indicated in 21 CFR § 1143.3(a)(1); and
- Be centered in the warning area in which the text is required to be printed and positioned such that the text of the required warning statement and the other information on the principal display panels have the same orientation.
A retailer of any cigarette tobacco, roll-your-own tobacco, or covered tobacco products (other than cigars) will not be in violation of this section for packaging that: (i) Contains a health warning; (ii) Is supplied to the retailer by the tobacco product manufacturer, importer, or distributor, who has the required state, local, or Alcohol and Tobacco Tax and Trade Bureau (TTB)-issued license or permit, if applicable; and (iii) Is not altered by the retailer in a way that is material to the requirements of this section.
These images are intended to serve as examples and alternative approaches may suffice.
Packages that are Too Small to Display the Required Warning Statement
A covered tobacco product (other than cigars), cigarette tobacco product, or roll-your-own tobacco product that is too small or otherwise unable to accommodate a label with sufficient space to bear the required warning statement must contain the required warning statement on one of the following, per 21 CFR § 1143.3(d):
- Carton
- Outer container
- Wrapper
- Tag firmly and permanently affixed to the tobacco product package.
These images are intended to serve as examples and alternative approaches may suffice.
Zero-Nicotine: Self-certification and Alternative Required Warning Statement
If the product does not contain nicotine and is made or derived from tobacco, the tobacco product manufacturer may display the alternative statement, instead of the nicotine addictiveness warning statement.
To display the alternative statement on packages and/or advertisements for a tobacco product, the tobacco product manufacturer must submit to FDA a confirmation statement certifying to be true and accurate that the product does not contain nicotine and that the tobacco product manufacturer has data to support that assertion. If you have any questions on the self-certification, you can contact FDA.
The alternative warning statement, under 21 CFR § 1143.3(c), for applicable package labels and advertisements reads:
This product is made from tobacco.
Tobacco products that do not contain tobacco or nicotine and are not made or derived from tobacco or nicotine do not meet the definition of covered tobacco product, and will not be required to carry an addiction warning or to submit a self-certification for use of the alternative statement.
Other Labeling Requirements
There are other labeling requirements. For example, tobacco products deemed under the deeming final rule to be subject to FDA’s authority, if in package form, must bear a label containing —
- the name and place of business of the tobacco product manufacturer, packer, or distributor;
- an accurate statement of the quantity of the contents in terms of weight, measure, or numerical count;
- an accurate statement of the percentage of the tobacco used in the product that is domestically grown tobacco and the percentage that is foreign grown tobacco; and
- the statement required under section 920(a)…(section 903(a)(2) of the Federal Food, Drug, and Cosmetic Act (FD&C Act)).
Section 920(a) of the FD&C Act provides that the label, packaging, and shipping containers of tobacco products shall bear the statement “Sale only allowed in the United States”.
In the guidance entitled “Interpretation of and Compliance Policy for Certain Label Requirement; Applicability of Certain Federal Food, Drug, and Cosmetic Act Requirements to Vape Shops”, FDA indicated that it does not intend to enforce section 903(a)(2)(C) of the FD&C Act for those products that are made or derived from tobacco.
1. FDA does not intend to enforce these warning statement requirements for products that were manufactured before the compliance date (August 10, 2018) of the new required warning statement for covered tobacco products, cigarette tobacco, and RYO tobacco. For important details about required warning statements, please see the small entity compliance guide.