2021 FDA Science Forum
Using Real World Evidence on Repurposed Drugs to Inform Future Clinical Trials to Treat Covid-19
- Authors:
- Center:
-
Contributing OfficeCenter for Drug Evaluation and Research
Abstract
Background
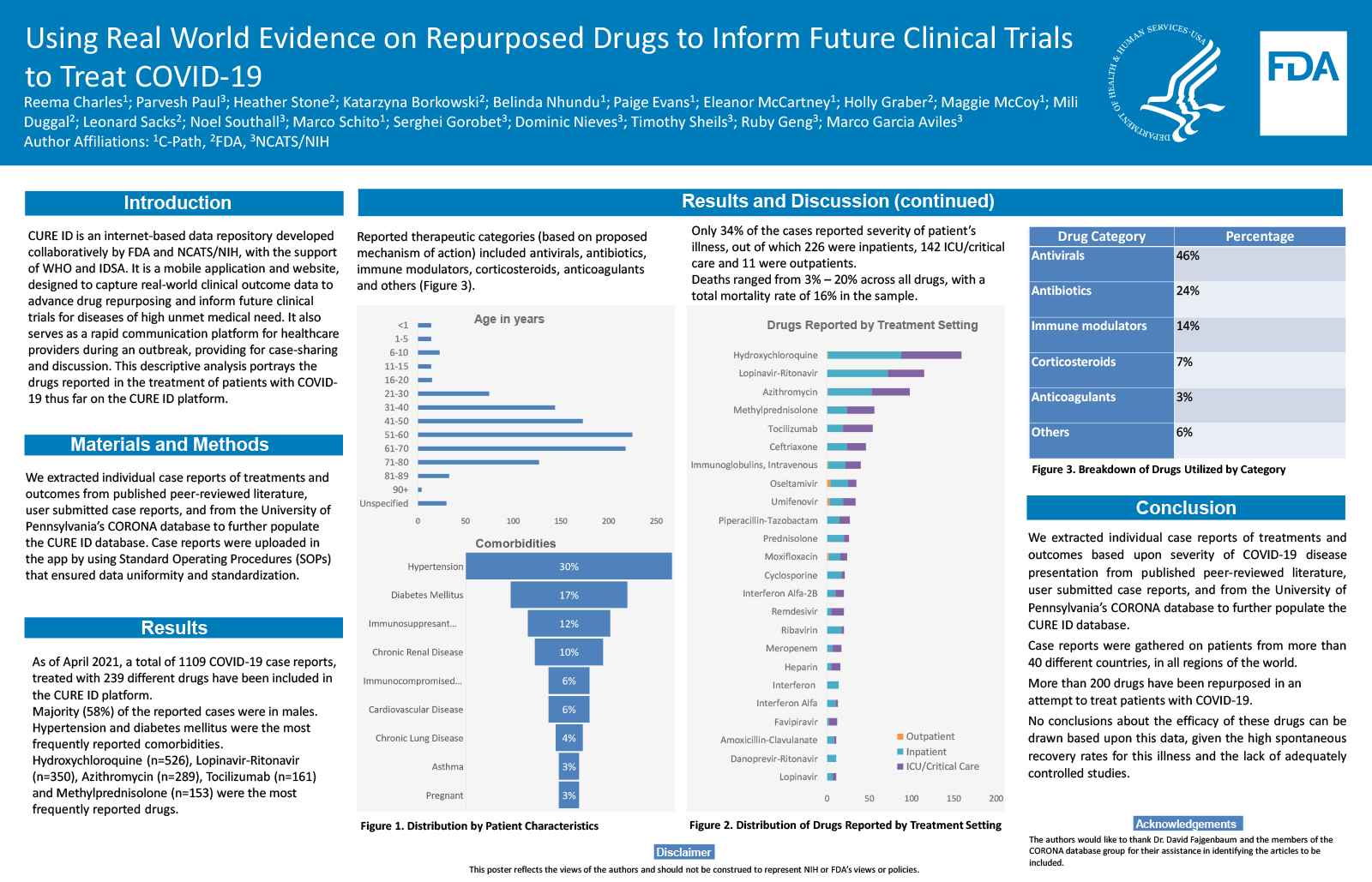
CURE ID is an internet-based repository developed collaboratively by FDA and NCATS/NIH. CURE ID is a mobile application and website (https://cure.ncats.io/explore), designed to capture real-world clinical outcome data to advance drug repurposing and inform future clinical trials for diseases of high unmet medical need. It also serves as a rapid communication platform for healthcare providers during an outbreak, providing for case-sharing and discussion. Latest medical research, disease specific clinical trials curated from https://clinicaltrials.gov/, and a news feed that offers updates related to infectious diseases are the other features of the app.
Purpose
To gather data on the use of various drugs re-purposed to treat COVID-19 from case reports submitted on the CURE ID platform, from the published literature and users, with the intent to capture efficacy signals and generate hypotheses to guide future studies.
Methods
We extracted individual case reports of treatments and outcomes from published literature, user submitted case reports, and from the University of Pennsylvania’s CORONA database to further populate the CURE ID database. Case reports were uploaded in the app by using Standard Operating Procedures (SOPs) that ensured data uniformity and standardization.
Results
Hydroxychloroquine (n=525) was most commonly reported drug, followed by lopinavir-ritonavir (n=350), azithromycin (n=288), tocilizumab (n=160), methylprednisolone (n=153), intravenous immunoglobulins (n=114), ceftriaxone (n=114), oseltamivir (n=110), umifenovir (n=108), remdesevir (n=58), ribavirin (n=56). Piperacillin-Tazobactam, moxifloxacin, meropenem, convalescent plasma, enoxaparin, interferon, prednisolone, interferon alfa, vancomycin, anakinra and interferon Alfa-2B were also reported.
Conclusion
Several drugs are being repurposed to treat COVID-19. By utilizing the real-world data captured in CURE ID, along with published observational studies, hypotheses can be generated to inform future clinical trials.