2021 FDA Science Forum
Universal Influenza Vaccine Based on Conserved Antigens Induces Broad, Long-Term Immunity and Protection
- Authors:
- Center:
-
Contributing OfficeCenter for Biologics Evaluation and Research
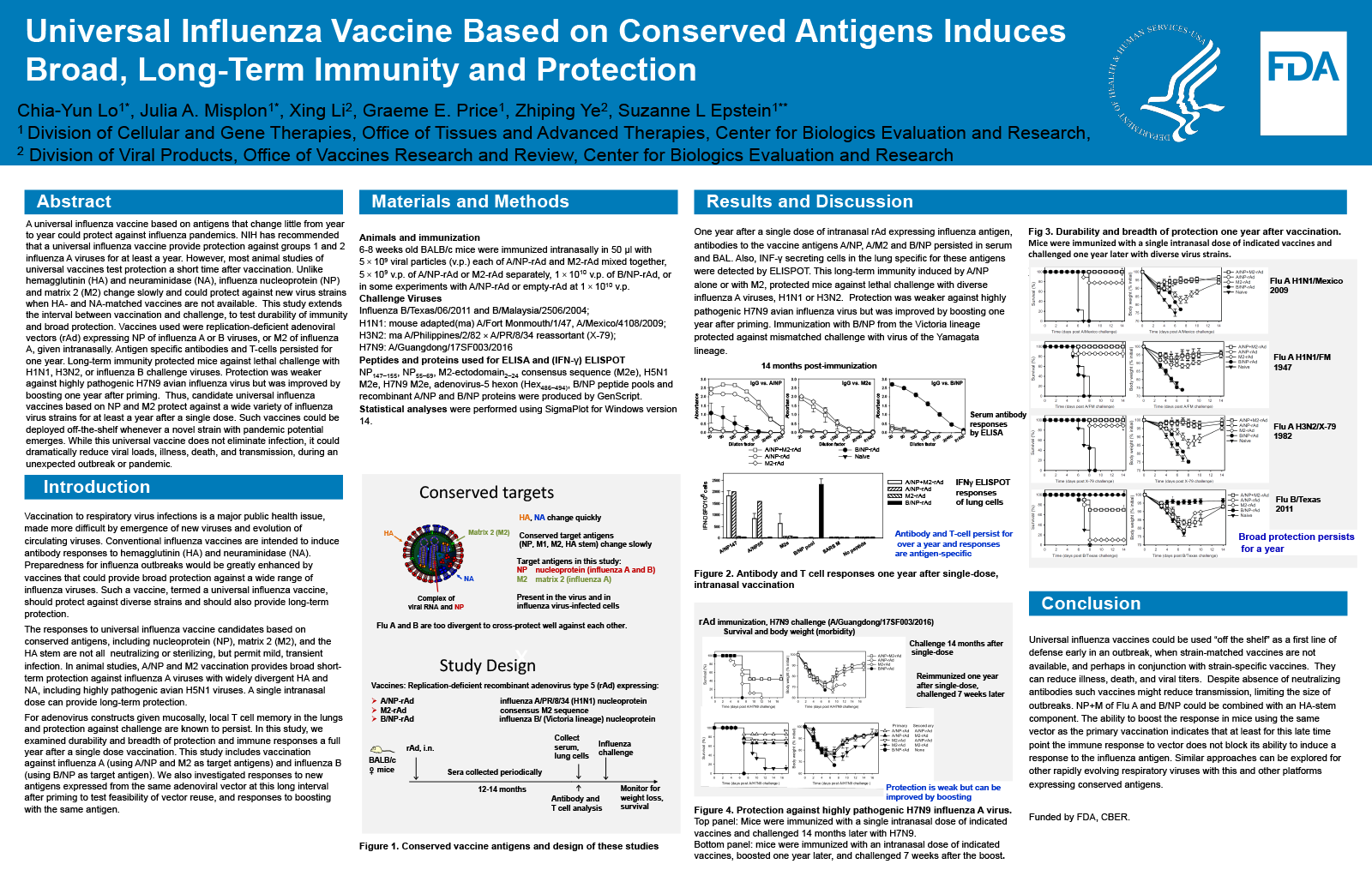
Abstract
Background
Respiratory viruses can spread rapidly and pose a threat of pandemics, as shown by influenza and SARS-CoV-2. When a new respiratory virus emerges, development of a vaccine takes months, and during that time the population is vulnerable. NIAID/NIH has recommended that a universal influenza vaccine should provide protection against groups 1 and 2 influenza A viruses for at least a year. However, most animal studies of universal vaccine candidates involve challenge a short time after vaccination.
Purpose
Unlike hemagglutinin (HA) and neuraminidase (NA), which mutate frequently, influenza nucleoprotein (NP) and matrix 2 (M2) change slowly. As target antigens they could protect against new virus strains when HA- and NA-matched vaccines are not available. We have shown that immunity to NP and M2 protects animals against diverse influenza strains. This study extends the interval between vaccination and challenge, to test durability of immunity and broad protection.
Methodology
Vaccines used were replication-deficient adenoviral vectors (rAd) expressing nucleoprotein (NP) of influenza A or B viruses, or matrix 2 (M2) of influenza A. Mice were immunized intranasally to focus immunity in the respiratory tract. Groups received A/NP+M2-rAd, A/NP-rAd alone, A/M2-rAd alone, or B/NP-rAd prior to intranasal challenge one year later with diverse influenza A or B strains. Antibodies in serum and bronchoalveolar lavage were analyzed by ELISA. Lung T cell responses were analyzed by interferon-gamma ELISPOT.
Results
One year after single-dose intranasal rAd, antibodies to the vaccine antigens persisted in serum. Antigen-specific T cell responses persisted in the lungs. Long-term immunity protected mice against lethal challenge with H1N1, H3N2, or influenza B challenge viruses. Protection was weaker against highly pathogenic H7N9 avian influenza virus but was improved by boosting one year after priming.
Conclusion
Candidate universal influenza vaccines based on NP and M2 provide protection against a wide variety of influenza virus strains that lasts at least a year after a single dose. Such vaccines could be deployed off-the-shelf whenever a novel strain with pandemic potential emerges. While this universal vaccine does not eliminate infection, it could dramatically reduce viral loads and thus illness, death, and transmission, during an unexpected outbreak or pandemic.