2021 FDA Science Forum
Serious Cutaneous Adverse Reaction Related Labeling Changes: An Evaluation of Supporting Evidence and Labeling Features
- Authors:
- Center:
-
Contributing OfficeCenter for Drug Evaluation and Research
Abstract
Background
Stevens-Johnson Syndrome (SJS) and Toxic Epidermal Necrolysis (TEN) are serious, rare, and life-threatening dermatologic adverse reactions that are rarely detected in pre-market drug development programs. The lack of diagnostic testing, terminology changes over time, and infrequent occurrence can create challenges for consistent management of SJS/TEN postmarket safety signals. Understanding the evidence and other factors associated with safety-related labeling changes (SrLCs) may provide information to enhance current postmarketing pharmacovigilance practices.
Purpose
To characterize the evidence supporting SJS/TEN SrLCs and language used to convey the risk in the Prescribing Information (PI).
Methodology
FDALabel and FDA’s SrLC database were utilized to identify all approved drug and therapeutic biological products in which SJS/TEN terms were added to the labeling between January 1, 2016 and December 31, 2020. Attributes of each SrLC were collected for analysis including the labeling section modified and terminology utilized. Two reviewers retrospectively reviewed FDA’s internal document repositories to identify the initiator of the SrLC (i.e., applicant or FDA) and evidence source supporting each SrLC.
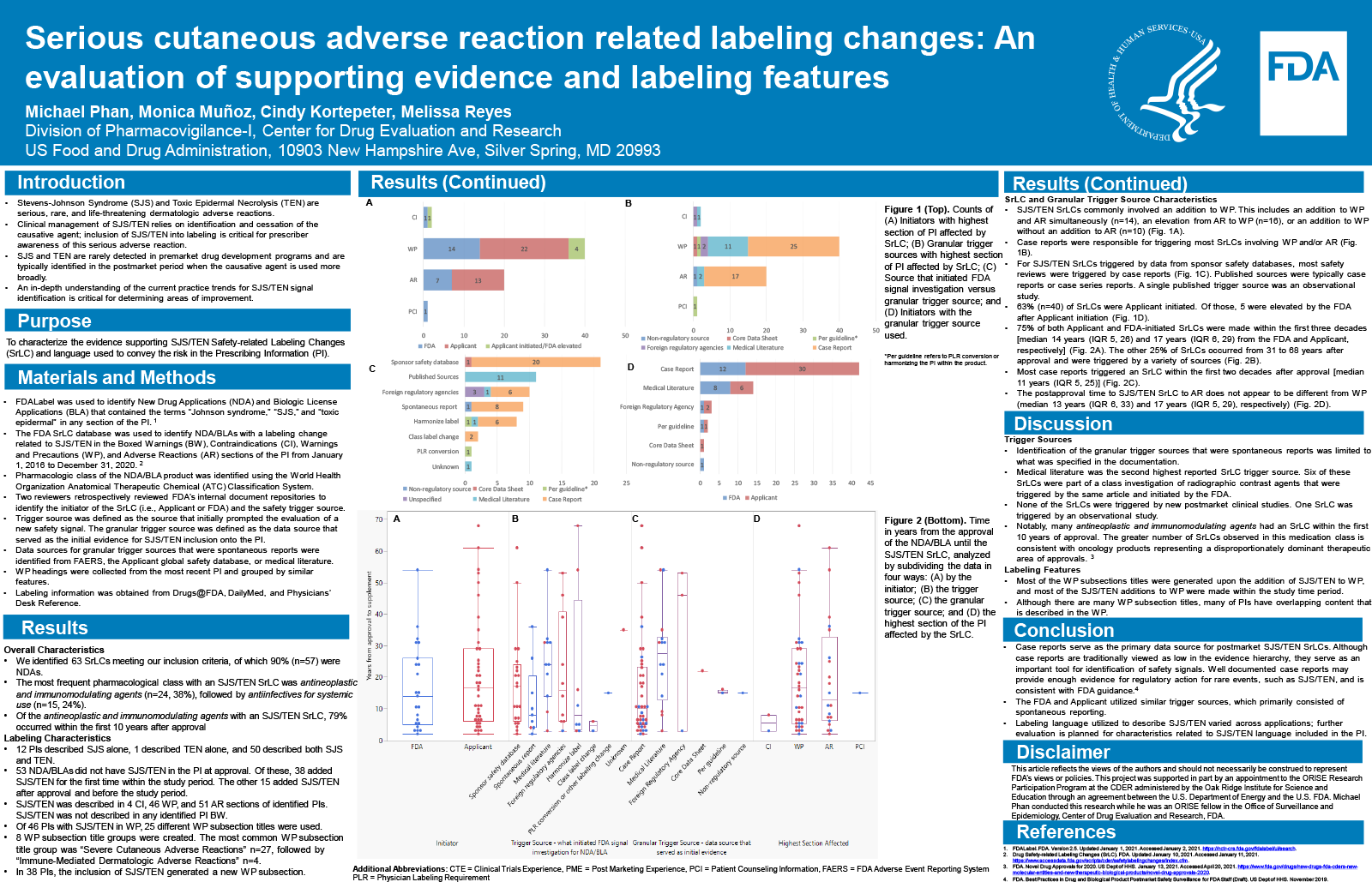
Results
In the study period, 61 products (54 drugs, 7 biologics) had an SJS/TEN related SrLC. New additions to the Adverse Reaction (AR) section alone (n=18, 30%), elevations from AR to Warnings and Precautions (WP) (n=13, 21%), and new additions to both the AR and WP (n=11, 18%) were the most common labeling changes. Most PI included both SJS and TEN terminology (n=51, 84%). Of the 43 labels that included SJS/TEN in the WP, 22 different section titles were used. More SrLCs were applicant initiated than FDA initiated (59% vs 39%), and the initiator was unknown for one SrLC. The applicant’s safety database (n=21, 34%), medical literature (n=11, 18%), foreign regulatory agencies (n=8, 13%), and spontaneous reports (n=8, 13%) were the most frequent sources triggering the SrLC.
Conclusion
Case reports serve as the primary data source for postmarket SJS/TEN SrLCs. While variation in SrLCs for a particular risk is not unexpected given the context of each change may be unique (e.g., magnitude of risk, evidence quality, therapeutic area), the variability observed in SJS/TEN SrLCs provides an opportunity to further evaluate factors influencing safety labeling decisions.