2021 FDA Science Forum
Profiling In-Vitro Release of Verteporfin from VISUDYNE Liposomal Formulation and Investigating the Kinetics of Human Serum Albumin (HSA) - Verteporfin Complex Formation
- Authors:
- Center:
-
Contributing OfficeOffice of Regulatory Affairs
Abstract
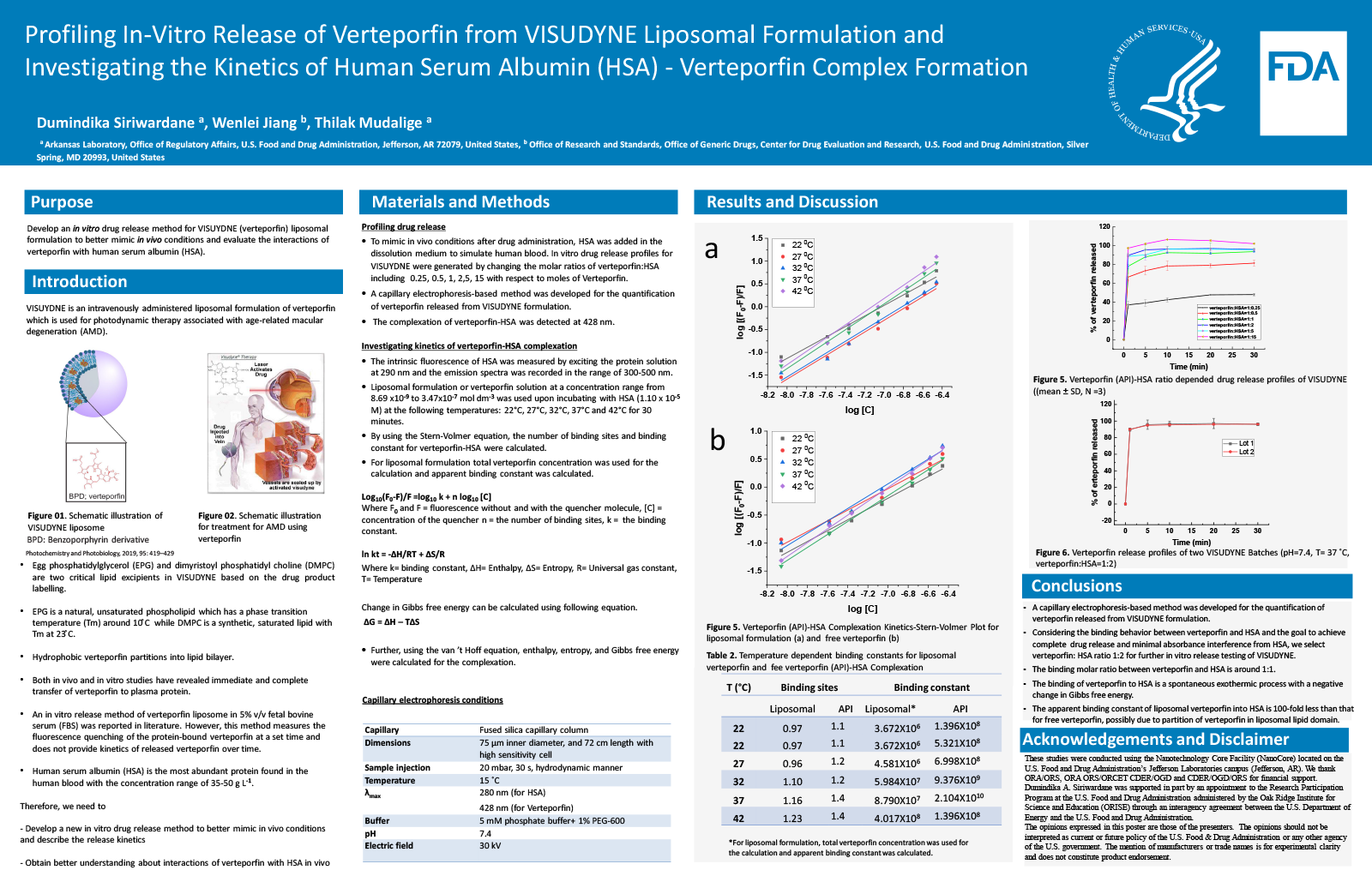
Purpose
VISUDYNE is an intravenously administered liposomal formulation of verteporfin used for photodynamic therapy of age-related macular degeneration. In this study, we sought to develop an in-vitro method to quantify drug release of verteporfin from VISUDYNE liposomal formulations in order to (1) better mimic in-vivo conditions, and (2) evaluate the interactions of verteporfin with human serum albumin (HSA).
Methods
The in-vitro release of verteporfin from VISUDYNE suspended in solution was quantified using a capillary electrophoresis-based method. HSA was added in the dissolution medium to simulate the protein content of human blood, and mimic in vivo conditions. In-vitro drug release profiles for verteporfin were generated by changing the amount of HSA, thus altering the ratios of verteporfin:HSA. Different experiments were conducted to evaluate the thermodynamic parameters of verteporfin binding to HSA and determine the number of verteporfin binding sites on HSA. Drug binding was quantified using drug-induced quenching of the intrinsic fluorescence of HSA as measured by exciting the protein solution at 290 nm and recording the emission spectra in the range of 300-500 nm. Verteporfins solutions ranging from 8.69 x10-9 to 3.47x10-7 mol L-1 were used with HSA (1.10 x10-5 mol L-1) at the following temperatures: 22 °C, 27 °C, 32 °C, 37 °C and 42 °C. The Stern-Volmer equation was used to calculate the number of binding sites and binding constant for verteporfin-HSA. The Van’t Hoff equation was used to calculate enthalpy, entropy, and Gibbs free energy for the verteporfin-HSA complex.
Results
HSA is the most abundant protein found in human blood with the concentration range of 35-50 g L-1 (0.51-0.73 mM). Upon administration of VISUDYNE formulation, the ratio of HSA:verteporfin is estimated to be about 27:1. When incubating with HSA, verteporfin can form verteporfin-HSA complex via an exothermic process involving hydrophobic interactions. According to Van’t Hoff equation, the thermodynamic parameters including enthalpy change, free energy change, and entropy changed for the verteporfin-HSA complex were calculated to be 190.51 kJ/mol, -40.10 kJ/mol (at 300 K), 768.71 J/mol K, respectively. With the increase of temperature, the binding constant between HSA and verteporfin increases. Verteprofin binding to HSA involves one albumin binding site and the binding molar ratio between verteporfin and HSA is approximately 1:1. When the release media contain higher levels of HSA (verteporfin:HSA molar ratios 1:1, 1:2, 1:5 and 1:15), the higher the levels of HSA resulting in increasing amounts of verteporfin released, achieving more than 90% of drug immediately released within 10 min. The immediate verteporfin release can be explained by the leaky nature of VISUDYNE. In addition, the incomplete release at lower verteporfin:HSA molar ratio (1:0.5, and 1:0.25) is possibly due to insufficient amount of HSA for complexation. When verteporfin:HSA ratio is 1:15, over 100% drug release was observed, possibly because HSA at higher concentration led to absorbance interference.
Conclusion
The binding of verteporfin released from VISUDYNE to HSA is a spontaneous reaction with a negative change in Gibbs free energy. The binding molar ratio between verteporfin and HSA is around 1:1. When the verteporfin:HSA ratio is 1:1 or above, more than 90% of API is released within 10 min. Considering the binding behavior between verteporfin and HSA and the goal to achieve complete drug release and minimal absorbance interference from HSA, we select verteporfin: HSA ratio 1:1 for further in vitro release testing of verteporfin from VISUDYNE.