2021 FDA Science Forum
Preliminary Studies on the Effect of Manufacturing Process on Globule Size Distribution of Sunscreen Formulations.
- Authors:
- Center:
-
Contributing OfficeCenter for Drug Evaluation and Research
Abstract
Background and Purpose
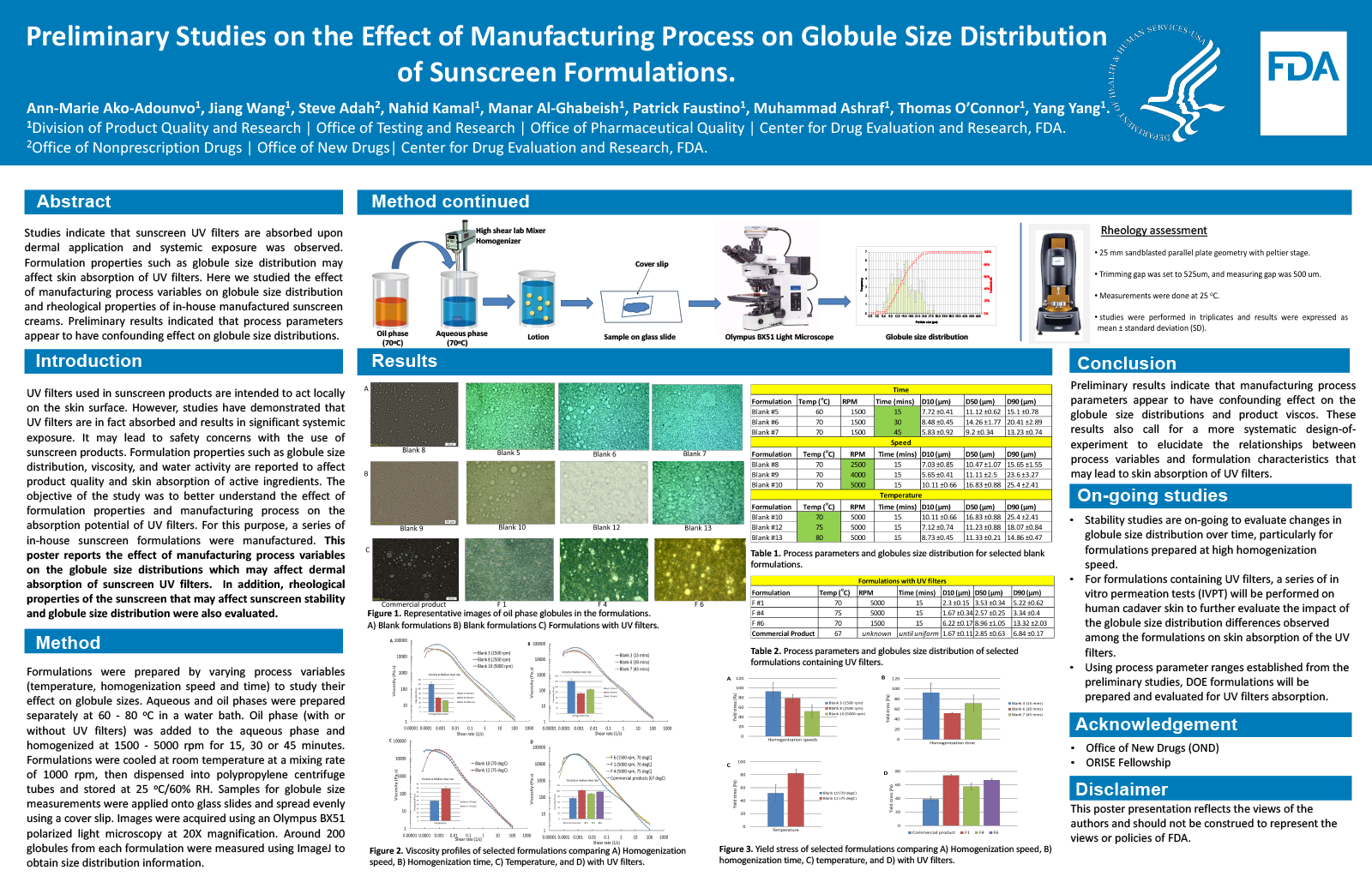
UV filters used in sunscreen products are intended to act locally on the skin surface. However, studies have demonstrated that UV filters are in fact absorbed and results in significant systemic exposure. It may lead to safety concerns with the use of sunscreen products. Formulation properties such as globule size distribution, viscosity, and water activity are believed to affect skin absorption of active ingredients. The objective of the study was to better understand the effect of formulation properties and manufacturing process on the absorption potential of UV filters. For this purpose, a series of in-house sunscreen formulations were manufactured. This abstract report the effect of manufacturing process variables on the globule size distributions which may affect drug release characteristics of sunscreen products.
Method
Formulations were prepared by varying process variables (temperature, homogenization speed and time) to study their effect on globule sizes. Aqueous and oil phases were prepared separately at 60, 70, 75 or 80 oC in a water bath. Oil phase (with or without UV filters) was added to the aqueous phase and homogenized at 1500, 2500, 4000 or 5000 rpm for 15, 30 or 45 minutes. Formulations were cooled at room temperature at a mixing rate of 1000 rpm, then dispensed into polypropylene centrifuge tubes and stored at 25 oC/60% RH. Samples for globule size measurements were applied onto glass slides and spread evenly using a cover slip. Images were acquired using an Olympus BX51 polarized light microscopy at 20X magnification. Around 200 globules from each formulation were measured using ImageJ to obtain size distribution information.
Results
Globule size distribution were noticeably smaller for formulations containing UV filters. The process temperature and homogenization time appeared to have an inverse effect on globule sizes in blank formulations. Increase in homogenization speed appeared to increase globule sizes as observed with the blank formulations. It may be explained by decrease in viscosity which may result in coalescence of globules at higher speeds. The temperature was also found to affect the globule size distribution in formulations containing UV filters.
Conclusion
Preliminary results indicate that manufacturing process parameters appear to have confounding effect on the globule size distributions. These results also call for a more systematic design-of-experiment to elucidate the relationships between process variables and formulation characteristics that may lead to skin absorption of UV filters.