2023 FDA Science Forum
Multi-cardiac ion channel block profiles for lopinavir, ritonavir, chloroquine and vanoxerine align with clinical ECG changes and proarrhythmia: two case studies in support of the CiPA initiative.
- Authors:
- Center:
-
Contributing OfficeCenter for Drug Evaluation and Research
Abstract
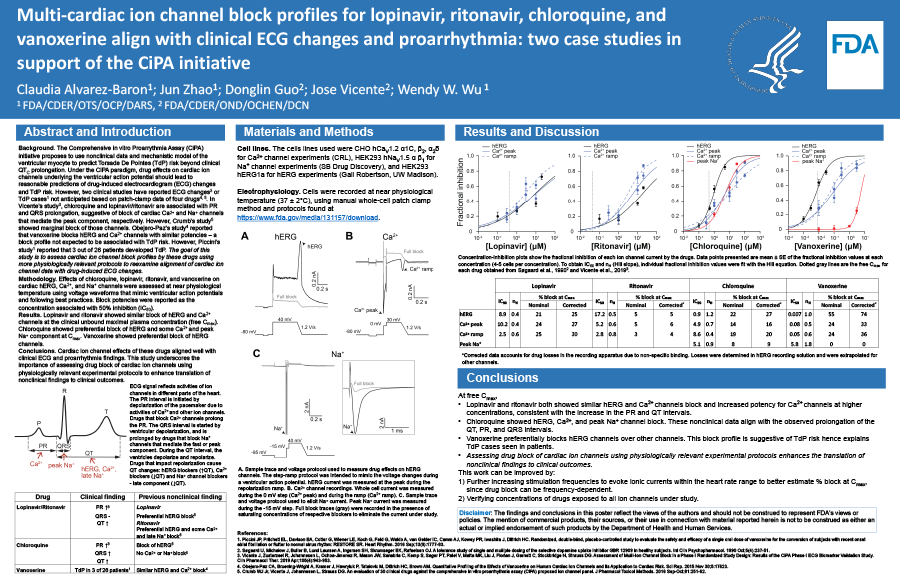
Background
The Comprehensive in vitro Proarrythmia Assay (CiPA) initiative proposes to use nonclinical data and mechanistic model of the ventricular myocyte to predict Torsade De Pointes (TdP) risk beyond clinical QTC prolongation. Under the CiPA paradigm, drug effects on cardiac ion channels underlying the ventricular action potential should lead to reasonable predictions of drug-induced ECG changes and TdP risk. However, two clinical studies have reported ECG changes (Vicente et al., 2019) or TdP cases (Piccini et al., 2016) not anticipated based on patch-clamp data of four drugs (Crumb et al., 2016; Obejero-Paz et al., 2015). In Vicente’s study, chloroquine and lopinavir/ritonavir are associated with PR and QRS prolongation, suggestive of CaV1.2 and NaV1.5 block, respectively. However, Crumb’s study showed marginal block of those channels. Obejero-Paz’s study reported that vanoxerine blocks hERG and CaV1.2 with similar potencies – a profile not expected to associate with TdP. However, Piccini’s study reported that 3 out of 26 patients developed TdP.
Methodology
Effects of chloroquine, lopinavir, ritonavir, and vanoxerine on cardiac hERG, Ca2+, and Na+ channels were assessed at near physiological temperature using voltage waveforms that mimic ventricular action potentials and following current best practices. Block potencies were reported as the concentration associated with 50% inhibition (IC50).
Results
Chloroquine’s IC50s: hERG, 0.9 µM; CaV1.2, 3.9 µM; and NaV1.5 peak, 5.1 µM. At free Cmax (0.3 µM), chloroquine blocked hERG by 22%, CaV1.2 by 17%, and NaV1.5 peak by 8%. Lopinavir’s IC50s: hERG, 8.5 µM; and CaV1.2, 10.2 µM. At free Cmax (387.3 nM), lopinavir blocked hERG by 4% and CaV1.2 by 24%. Ritonavir’s IC50s: hERG, 15.4 µM; and CaV1.2, 5.2 µM. At free Cmax (63.5 nM), ritonavir blocked hERG by 2% and CaV1.2 by 7%. Vanoxerine’s IC50s: hERG, 6.7 nM; CaV1.2, 81.5 nM; and NaV1.5 peak, 5.8 µM. At free Cmax (8.3 nM), vanoxerine blocked hERG by 55% and CaV1.2 by 23%.
Conclusions
Cardiac ion channel effects of these drugs aligned well with clinical ECG and proarrhythmia findings. This study underscores the importance of assessing drug block of cardiac ion channels using physiologically relevant experimental protocols to enhance translation of nonclinical findings to clinical outcomes.