2023 FDA Science Forum
Interlaboratory comparison exercise (ILC) of SARS-CoV-2 molecular detection method used by veterinary diagnostic laboratories
- Authors:
- Center:
-
Contributing OfficeCenter for Veterinary Medicine
Abstract
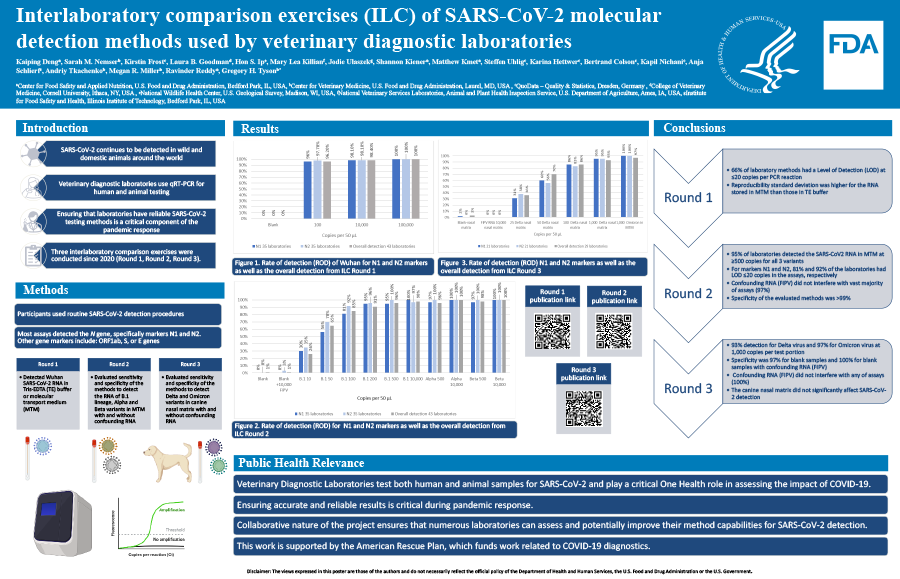
Ensuring reliable detection methods for SARS-CoV-2 is critical as veterinary diagnostic labs are testing both human and animal samples. The US Food and Drug Administration (FDA) has previously organized two collaborative inter-laboratory comparisons (ILC) exercises to veterinary diagnostic laboratories for evaluating their RT-qPCR methods routinely used for clinical testing. The results showed that the participants’ methods were sensitive and specific for detecting SARS-CoV-2 RNA spiked in viral transport medium. To further assess the RT-qPCR methods routinely used to detect emerging SARS-CoV-2 variants, the current ILC3 was conducted to detect the Delta and Omicron variants spiked in canine nasal matrix or viral transport medium.
Samples included inactivated Delta variant at levels ranging 25 to 1,000 copies per 50 mL of nasal matrix and Omicron variant at 1,000 copies per 50 mL of transport medium. Feline infectious peritonitis virus (FIPV) RNA was used as a confounder for specificity assessment. Participants used their routine diagnostic procedures for RNA extraction and RT-qPCR. The overall results showed 93% detection for Delta and 97% for Omicron at 1,000 copies per 50 uL (22-200 copies per reaction).
The overall specificity was 97% for blank samples and 100% for blank with FIPV. There were no significant differences in Ct values for samples at the same virus levels between N1 and N2 markers, nor between the two variants. The results indicated that all ILC3 participating laboratories were able to detect both Delta and Omicron variants. The canine nasal matrix did not significantly impact SARS-CoV-2 detection. This knowledge will lead to higher confidence in laboratory detection of current and new SARS-CoV-2 variants and aid in establishing reasonable cutoff parameters for these diagnostic methods. This work plays a critical role in public health as part of the continued pandemic response of these laboratories.