2021 FDA Science Forum
Descriptive Analysis of COVID-19 Clinical Trials Curated on the CURE ID Platform
- Authors:
- Center:
-
Contributing OfficeCenter for Drug Evaluation and Research
Abstract
Background:
CURE ID is an internet-based data repository (https://cure.ncats.io/explore), developed collaboratively by FDA and NCATS/NIH. It is designed to capture real-world clinical outcome data to advance drug repurposing and inform future studies and clinical trials for infectious diseases with high unmet medical need. It also serves as a repository of clinical trials curated from https://www.clinicaltrials.gov with the intention of keeping the infectious diseases community updated on the various clinical trials underway.
Purpose:
The current study is a descriptive analysis of various therapeutics in clinical trials against COVID-19 on the CURE ID platform. Having a panoramic view of the various drugs in clinical trials and the efforts being undertaken will keep the academic community informed and help prevent duplication of efforts amidst the current pandemic.
Methods:
Using clinicaltrials.gov we selected those trials addressing therapeutics for COVID-19 and reviewed the drugs used, their current status and the phases of development.
Results:
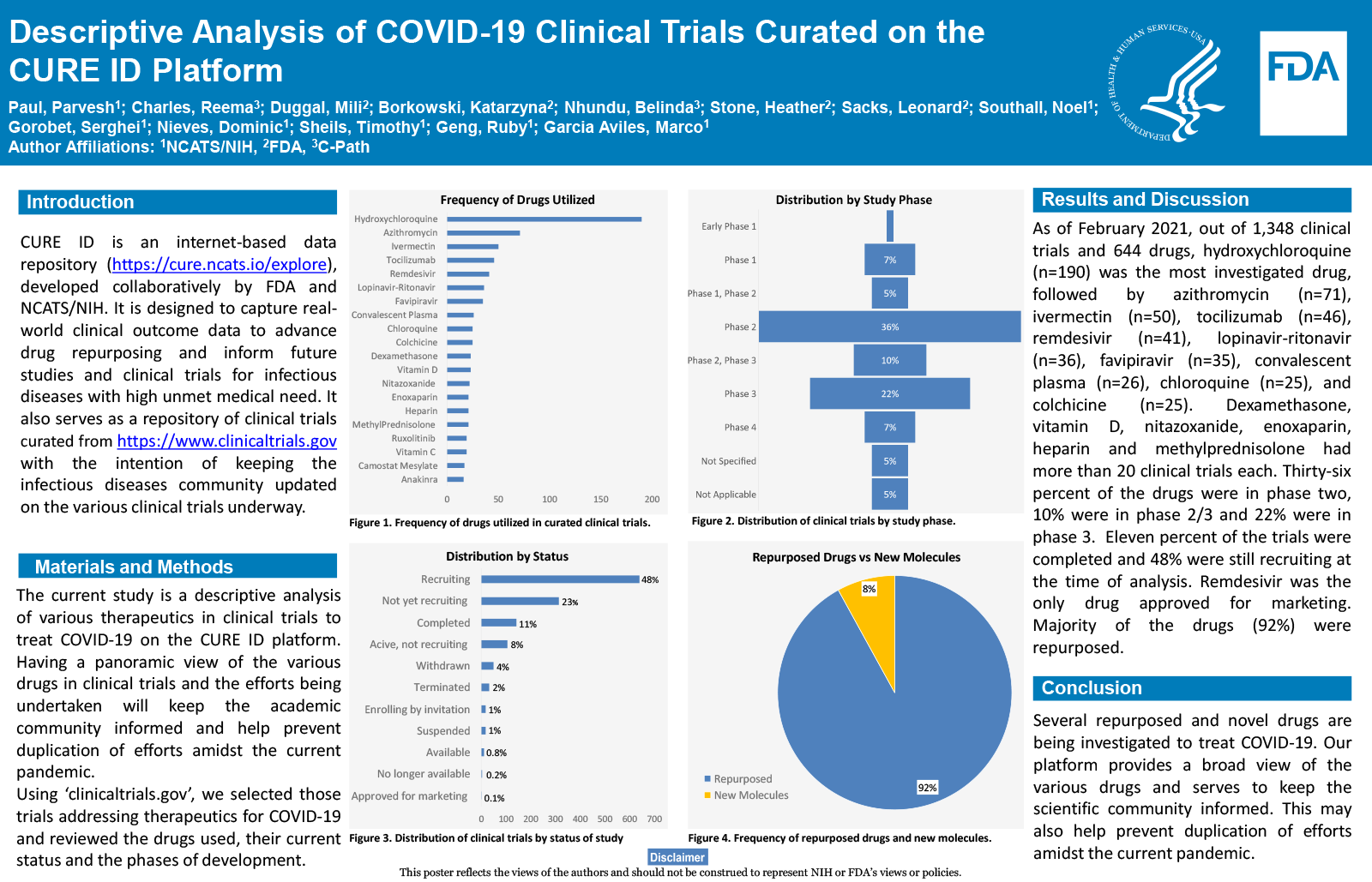
Out of 1,348 clinical trials and 644 drugs, Hydroxychloroquine (n=190) was the most commonly investigated drug, followed by Azithromycin (n=71), Ivermectin (n=50), Tocilizumab (n=46), Remdesivir (n=41), Lopinavir-Ritonavir (n=36), Favipiravir (n=35), Convalescent Plasma (n=26), Chloroquine (n=25), and Colchicine (n=25). Dexamethasone, Vitamin D, Nitazoxanide, Enoxaparin, Heparin and Methylprednisolone had more than 20 clinical trials each. Eleven percent of the trials were completed and 48% were still recruiting at the time of analysis. Remdesivir was the only drug approved for marketing. Thirty-six percent of the drugs were in phase two,10% were in phase 2/3 and 22% were in phase 3.
Conclusion:
Several repurposed and novel drugs are being investigated to treat COVID-19. Our platform provides a broad view of the various drugs and serves to keep the scientific community informed. This may also help prevent duplication of efforts amidst the current pandemic.