2021 FDA Science Forum
Continuous Manufacturing of Liposomes and Lipid Nanoparticles
- Authors:
- Center:
-
Contributing OfficeCenter for Drug Evaluation and Research
Abstract
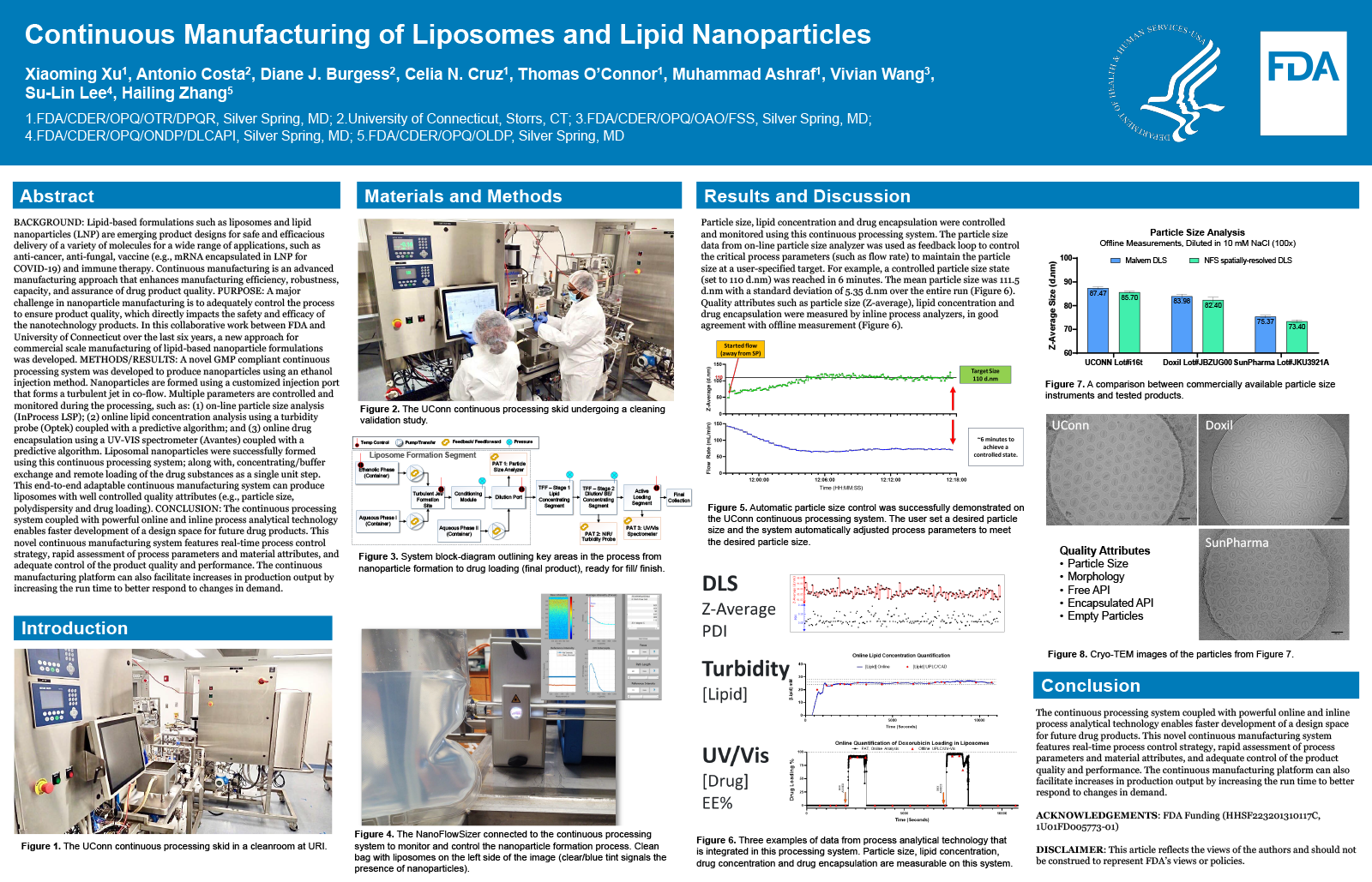
BACKGROUND
Lipid-based formulations such as liposomes and lipid nanoparticles (LNP) are emerging product designs for safe and efficacious delivery of a variety of molecules for a wide range of applications, such as anti-cancer, anti-fungal, vaccine (e.g., mRNA encapsulated in LNP for COVID-19) and immune therapy. Continuous manufacturing is an advanced manufacturing approach that enhances manufacturing efficiency, robustness, capacity, and assurance of drug product quality.
PURPOSE
A major challenge in nanoparticle manufacturing is to adequately control the process to ensure product quality, which directly impacts the safety and efficacy of the nanotechnology products. In this collaborative work between FDA and University of Connecticut over the last six years, a new approach for commercial scale manufacturing of lipid-based nanoparticle formulations was developed.
METHODS/RESULTS
A novel GMP compliant continuous processing system was developed to produce nanoparticles using an ethanol injection method. Nanoparticles are formed using a customized injection port that forms a turbulent jet in co-flow. Multiple parameters are controlled and monitored during the processing, such as: (1) on-line particle size analysis (InProcess LSP); (2) online lipid concentration analysis using a turbidity probe (Optek) coupled with a predictive algorithm; and (3) online drug encapsulation using a UV-VIS spectrometer (Avantes) coupled with a predictive algorithm. Liposomal nanoparticles were successfully formed using this continuous processing system; along with, concentrating/buffer exchange and remote loading of the drug substances as a single unit step. This end-to-end adaptable continuous manufacturing system can produce liposomes with well controlled quality attributes (e.g. particle size, polydispersity and drug loading).
CONCLUSION
The continuous processing system coupled with powerful online and inline process analytical technology enables faster development of a design space for future drug products. This novel continuous manufacturing system features real-time process control strategy, rapid assessment of process parameters and material attributes, and adequate control of the product quality and performance. The continuous manufacturing platform can also facilitate increases in production output by increasing the run time to better respond to changes in demand.
ACKNOWLEDGEMENTS
FDA Funding Support# HHSF223201310117C, 1U01FD005773-01.
DISCLAIMER
This article reflects the views of the authors and should not be construed to represent FDA’s views or policies.