2023 FDA Science Forum
Confirmatory Evidence of Effectiveness Used to Support Non-Oncologic Rare Disease Novel Drug Marketing Application Approvals, CY 2020-2022
- Authors:
- Center:
-
Contributing OfficeCenter for Drug Evaluation and Research
Abstract
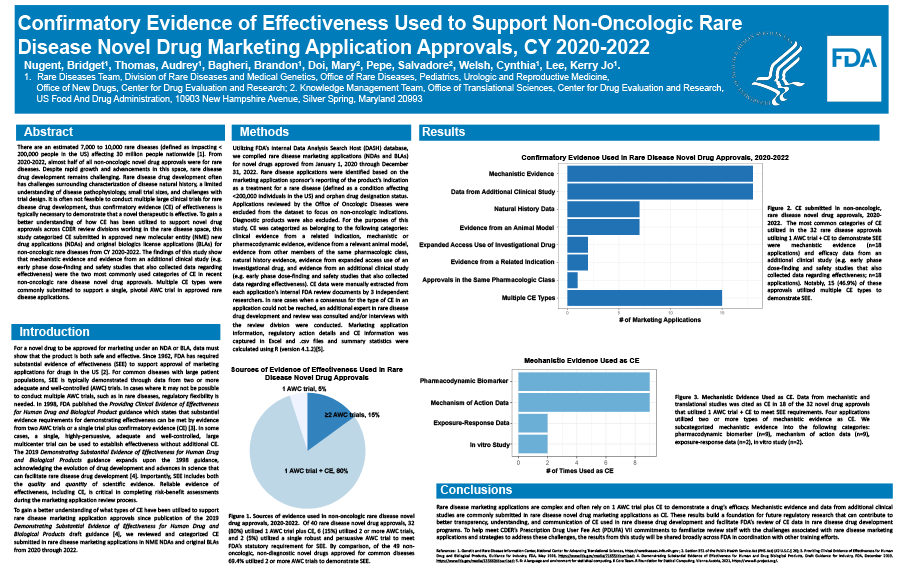
There are over 10,000 rare diseases (defined as impacting < 200,000 people in the US) affecting 30 million people nationwide. From 2020-2022, almost half of all non-oncologic novel drug approvals were for rare diseases. However, rare disease drug development remains challenging often due to limited understanding of disease biology, disease heterogeneity, and small patient populations. Since 1962, FDA has required substantial evidence of effectiveness to support approval of marketing applications, typically consisting of two adequate and well-controlled clinical trials or one adequate and well-controlled trial plus confirmatory evidence (CE). In cases where it may not be possible to conduct multiple adequate and well-controlled trials, such as in rare diseases, CE is often needed to support effectiveness. To gain a better understanding of how CE has been utilized to support novel drug approvals across CDER review divisions working in the rare disease space, this study categorized CE submitted in approved new molecular entity (NME) novel drug applications (NDAs) and original biologics license applications (BLAs) for non-oncologic rare diseases from CY 2020-2022. Categories of CE were based on the 1998 Providing Clinical Evidence of Effectiveness for Human Drug and Biological Product FDA guidance and the 2019 Demonstrating Substantial Evidence of Effectiveness for Human Drug and Biological Products draft guidance. CE data were manually extracted from internal FDA review documents by 3 independent researchers. Summary statistics for marketing application information, review decision, regulatory action details, and CE information were calculated using the R programming language. The findings of this study show that Mechanistic or Pharmacodynamic Evidence and Evidence from an Additional Clinical Study (e.g. early proof of concept, dose-finding, and safety studies) were the most commonly used categories of CE in recent non-oncologic rare disease NME drug approvals. Interestingly, multiple CE types were commonly submitted to support a single, pivotal adequate and well-controlled trial in approved rare disease applications. To help meet CDER’s Prescription Drug User Fee Act (PDUFA) VII commitments to familiarize review staff with the challenges associated with rare disease marketing applications and strategies to address these challenges, the results from this study will be shared broadly across FDA.