2021 FDA Science Forum
Assessing Quality of Pluripotent Stem Cells (PSCs) Expanded in 3D Suspension Culture
- Authors:
- Center:
-
Contributing OfficeCenter for Biologics Evaluation and Research
Abstract
Background
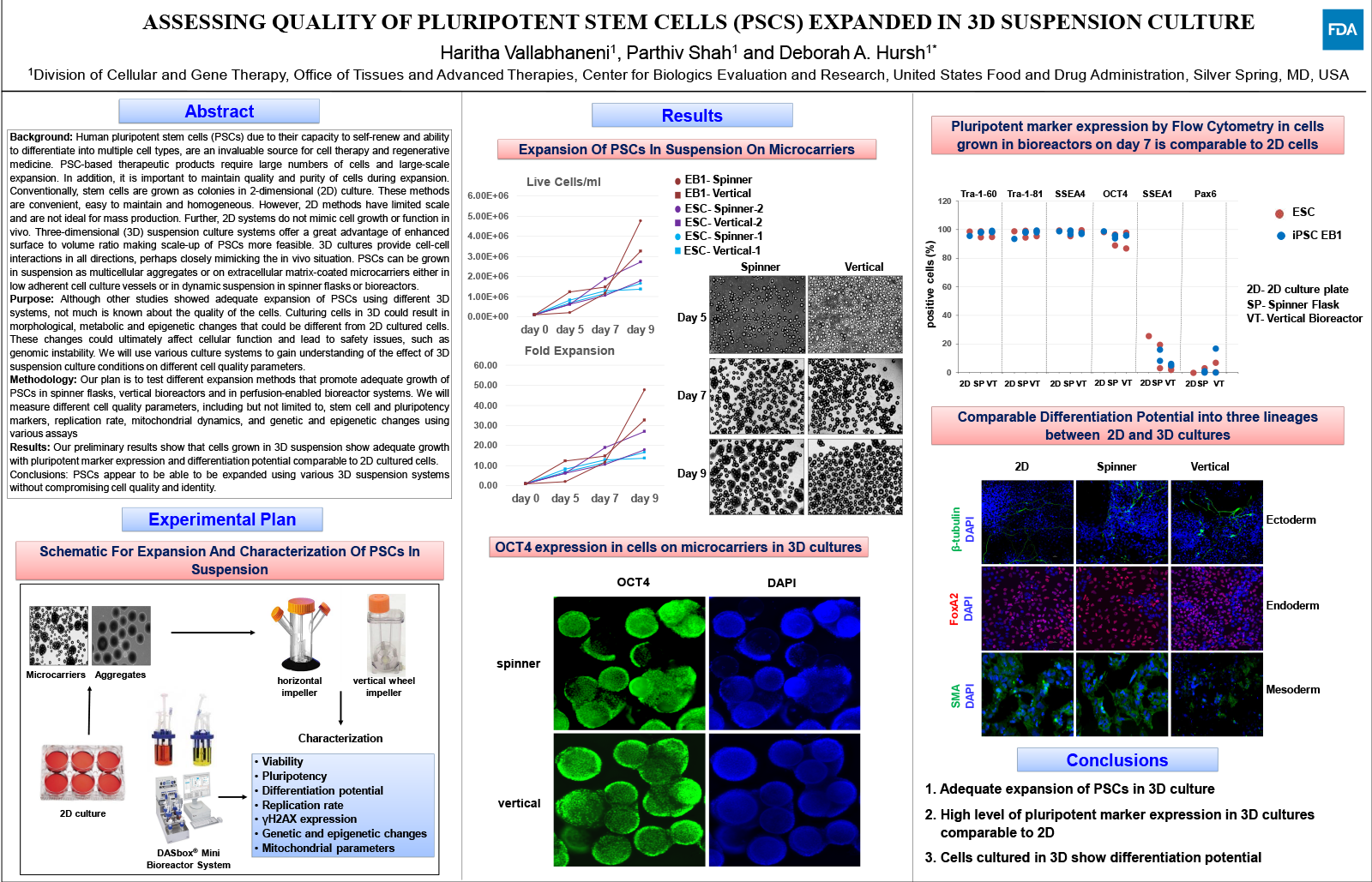
Human pluripotent stem cells (PSCs) due to their capacity to self-renew and ability to differentiate into multiple cell types, are an invaluable source for cell therapy and regenerative medicine. PSC-based therapeutic products require large numbers of cells and large-scale expansion. In addition, it is important to maintain quality and purity of cells during expansion. Conventionally, stem cells are grown as colonies in 2-dimensional (2D) culture. These methods are convenient, easy to maintain and homogeneous. However, 2D methods have limited scale and are not ideal for mass production. Further, 2D systems do not mimic cell growth or function in vivo. Three-dimensional (3D) suspension culture systems offer a great advantage of enhanced surface to volume ratio making scale-up of PSCs more feasible. 3D cultures provide cell-cell interactions in all directions, perhaps closely mimicking the in vivo situation. PSCs can be grown in suspension as multicellular aggregates or on extracellular matrix-coated microcarriers either in low adherent cell culture vessels or in dynamic suspension in spinner flasks or bioreactors.
Purpose
Although other studies showed adequate expansion of PSCs using different 3D systems, not much is known about the quality of the cells. Culturing cells in 3D could result in morphological, metabolic and epigenetic changes that could be different from 2D cultured cells. These changes could ultimately affect cellular function and lead to safety issues, such as genomic instability. We will use various culture systems to gain understanding of the effect of 3D suspension culture conditions on different cell quality parameters.
Methodology
Our plan is to test different expansion methods that promote adequate growth of PSCs in spinner flasks, vertical bioreactors and in perfusion-enabled bioreactor systems. We will measure different cell quality parameters, including but not limited to, stem cell and pluripotency markers, replication rate, mitochondrial dynamics, and genetic and epigenetic changes using various assays
Results
Our preliminary results show that cells grown in 3D suspension show adequate growth with pluripotent marker expression and differentiation potential comparable to 2D cultured cells.
Conclusions
PSCs appear to be able to be expanded using various 3D suspension systems without compromising cell quality and identity.