2021 FDA Science Forum
Analyzing the Role of Gut Microbiota in the Onset of Autoimmune Diseases Using the TNFΔARE Murine Model
- Authors:
- Center:
-
Contributing OfficeCenter for Drug Evaluation and Research
Abstract
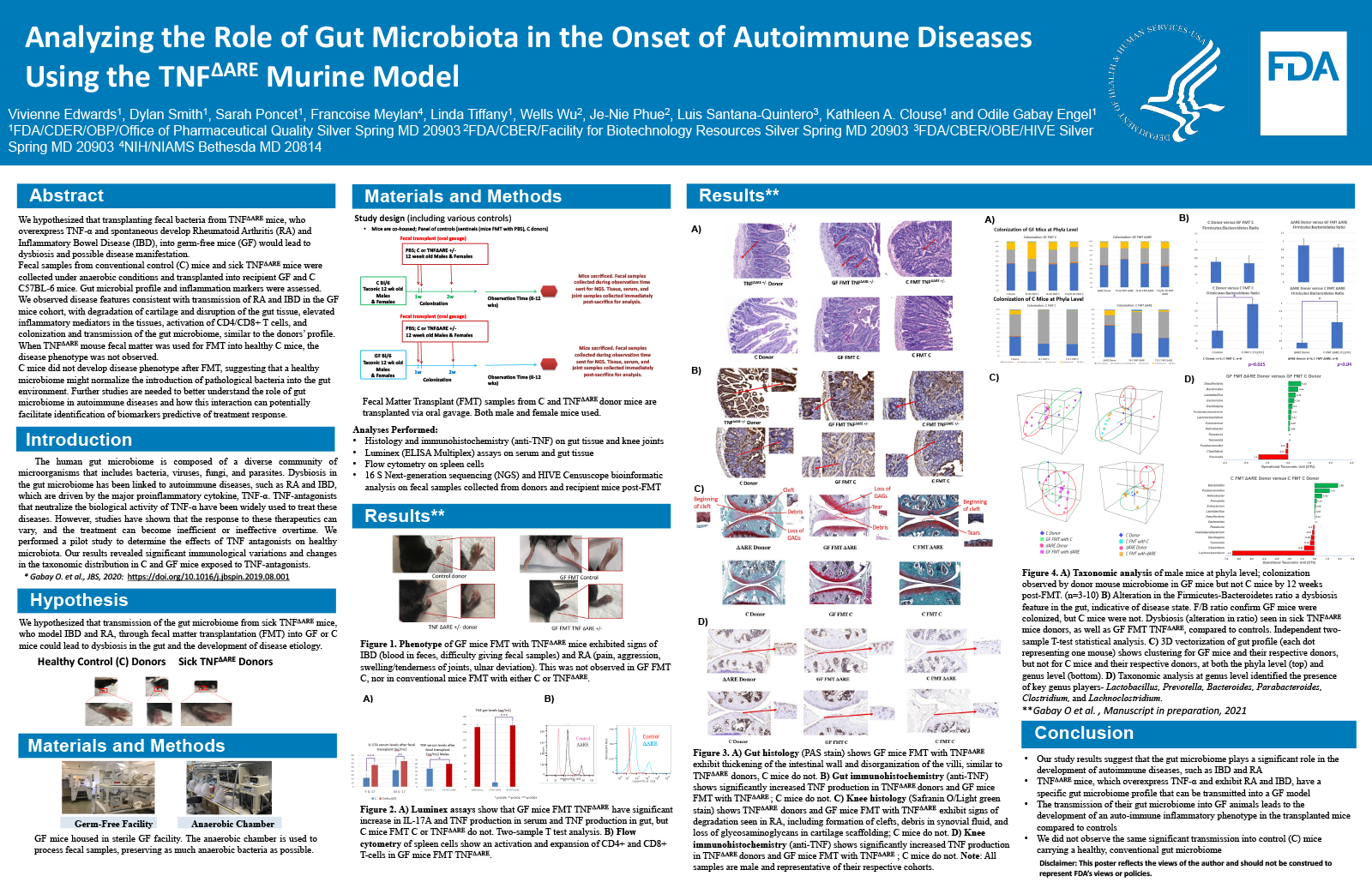
Background
Biologic therapeutics have been used to treat autoimmune diseases, but have shown variable efficacy and responses in patients. Dysbiosis in the gut microbial community is associated with inflammatory bowel disease (IBD) and rheumatoid arthritis (RA). In a previous study, we found taxonomic differences between healthy mice and those treated with distinct TNF-antagonists.
Purpose/Objectives>
Utilizing our established experimental study design, we hypothesized that transplanting fecal bacteria from TNF?ARE mice, a mouse model exhibiting spontaneous IBD and RA, into germ-free (GF) mice would lead to dysbiosis and possible disease manifestation.
Materials/methods
Fecal samples from TNF?ARE mice were collected under anaerobic conditions and transplanted into C57BL-6 male and female WT and GF mice. Microbiome composition of the transplanted mice was assessed before and after Fecal Matter Transplant (FMT), using next generation sequencing (NGS) and FDA HIVE Censuscope bioinformatics analysis platform. Histology, immunohistochemistry, flow cytometry, and ELISA multiplex assays were also used to evaluate inflammatory markers in joints, tissue, and blood.
Results
We observed disease features transmission of RA and IBD in the GF mice cohort, with degradation of cartilage and disruption of the gut tissue, elevated inflammatory mediators in the tissues, activation of CD4/CD8+ T cells, and colonization and transmission of the gut microbiome, similar to the donors’ profile. When TNF?ARE mouse fecal matter was FMT into healthy WT mice, disease phenotype was not observed.
Conclusions
This study highlights the importance of the gut microbiome in the onset of autoimmune diseases, such as RA and IBD, and the variation occurring during treatment with biologic therapeutics. WT mice did not develop disease phenotype after FMT, suggesting that a healthy microbiome might normalize the introduction of pathological bacteria into the gut environment. Further studies are needed to better understand these interactions and to potentially lead to identification of biomarkers predictive of treatment response.