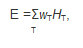Response to University of California - San Francisco Regarding Their Letter of Concern, October 12, 2010
October 12, 2010
Dr. John P. Holdren
Assistant to the President for Science and Technology
Director, Office of Science and Technology Policy
Executive Office of the President
New Executive Office Building
725 17th St. NW
Washington, DC 20502
Dear Dr. Holdren,
Thank you for sharing the April 6 letter you received regarding general-use full-body x-ray screening systems used for airport security. As with all x-ray security products, justified application demands a balancing act: The radiation dose delivered must be sufficient to do the job—in this case to identify security threats—while presenting no more than a miniscule risk to people being scanned, including special populations.
The overriding concern expressed in the letter is the extent to which the safety of the security devices has been adequately demonstrated. Since 1990, the Food and Drug Administration (FDA) has regulated manufacturers to ensure the radiation safety of full-body x-ray security screening systems. The FDA consulted its Technical Electronic Product Radiation Safety Standards Committee (TEPRSSC) about these products during several meetings from 1998 through 2003. TEPRSSC is the independent advisory committee to FDA with expertise in electronic product radiation issues. This expert committee raised several issues during these meetings and FDA responded by initiating work on a consensus radiation safety standard through the American National Standards Institute (ANSI) and Health Physics Society (HPS). FDA assembled a working group of experts that included representatives from manufacturers, security agencies, and other regulatory agencies. The working group produced a national standard, Radiation Safety for Personnel Security Screening Systems Using X-rays1, which was published in July 2002. The 2002 standard required facilities to ensure that no individual scanned received an effective dose in excess of 0.25 mSv (25 mrem) in any 12-month period. The standard also provided other guidelines specific to the radiation safety aspects of the design and operation of these systems. This annual dose limit is based on the National Council of Radiation Protection and Measurements2 (NCRP) recommendations for the annual effective dose limit for individual members of the general public3. NCRP’s dose limitation recommendations for the general public were made with the understanding that the general public includes special populations that are more sensitive to radiation, such as children.
In September 2002 FDA asked the NCRP to undertake a study that led to NCRP Commentary No. 16 (2003), Screening of Humans for Security Purposes Using Ionizing Radiation Scanning Systems. The committee that prepared this commentary included representatives from the Milton S. Hershey Medical Center, Columbia University, FDA’s Center for Devices and Radiological Health, the U.S. Environmental Protection Agency, and the NCRP Secretariat consulting staff. This commentary introduced the concept of general-use and limited-use systems. Commentary No. 16 recommended g eneral-use systems should not exceed the dose limit set in the 2002 standard and can be used mostly4 without regard to the number of individuals scanned or the number of scans per individual in a year.
FDA brought the issue of consistent federal evaluation and justification of security screening practices that used ionizing radiation to the Interagency Steering Committee on Radiation Standards (ISCORS)5. ISCORS was established to foster early resolution and coordination of regulatory issues associated with radiation standards and guidelines. ISCORS published Guidance for Security Screening of Humans Utilizing Ionizing Radiation (2008) to assist Federal agencies in determining when the use of ionizing radiation for security screening of humans is warranted and to provide guidelines for establishing a radiation safety program. The Transportation Security Administration (TSA), FDA, Occupational Safety and Health Administration, National Institute of Standards and Technology (NIST), U.S. Army Center for Health Promotion and Preventive Medicine, Nuclear Regulatory Commission, Environmental Protection Agency, Federal Bureau of Prisons, Department of Energy, Customs and Border Protection, Central Intelligence Agency, Maryland Department of the Environment, and Pennsylvania Bureau of Radiation Protection collaborated in developing this federal guidance.
Since publication of the original 2002 standard, a number of new system designs have been developed, including portal systems, multi-source systems, vehicle scanners meant for screening occupied vehicles, scanners for inspecting casts and prosthetic devices, and scanners using a radioisotope as the source of radiation. New uses for these systems include the use of vehicle and cargo scanners to inspect people and the limited use of higher-dose systems as defined in NCRP Commentary No. 16. Consequently, FDA and NIST chaired a working group to revise the national standard6. The revised standard, Radiation Safety for Personnel Security Screening Systems Using X-Ray or Gamma Radiation7, was published November 2009. It includes requirements that cover these new designs and uses. This standard applies to security screening systems in which people are intentionally exposed to primary beam x-rays and provides guidelines specific to radiation safety in the design and operation of these systems.The standard covers doses to individuals scanned, safety systems, operational procedures, information to provide to screened individuals, training for operators, and other issues. The revised standard retained the annual effective dose limit for members of the public of 0.25 mSv (25 mrem). This standard defines a general-use x-ray screening system as one that delivers less than 1/1000 of this dose per screening (0.25 µSv (25 µrem)). The rationale for the annual and per screening dose limits is presented in the standard8.
TSA requires that the full-body x-ray security systems approved for deployment (Advanced Imaging Technology (AIT)) conform to the requirements in the 2009 standard for general-use systems. Surveys of the recently deployed backscatter x-ray personnel security screening systems have been performed by an independent party to confirm compliance with the radiation dose-per-screening limits for general-use of the 2009 standard. All systems surveyed to date have been found to comply with the general-use dose-per-screening limit in that standard. In addition, our independent survey teams are gathering area radiation dose data by mounting dosimeters on (within the inspection zone) select systems.
Regarding the specific “Red Flag” issues raised in the letter:
First, the letter is correct to note that the TSA-deployed product is a recent model. However, the specification for the x-ray tube for the deployed model is almost identical to the original 1991 product. The stated concern was, “The majority of their energy is delivered to the skin and the underlying tissue.” We agree. However, the concern that “the dose to the skin may be dangerously high” is not supported. The recommended limit for annual dose to the skin for the general public is 50,000 µSv9. The dose to the skin from one screening would be approximately 0.56 µSv10 when the effective dose for that same screening would be 0.25 µSv11. Therefore the dose to skin for the example screening is at least 89,000 times lower than the annual limit.
Second, radiation safety protection quantities are stated as ‘effective dose’12. NCRP Commentary No. 16 says, “The purpose of effective dose13 is to place on a common scale the radiation doses: (1) from different types of ionizing radiation that have different biological effectiveness, and (2) in different organs or tissues that have different radiation sensitivities.” Comparing effective doses from different sources is appropriate. The comparison between the effective dose from cosmic ray exposure or a medical diagnostic chest x-ray and the effective dose from a security screening is intended to be a clear means of risk communication.
The third point relates to a concern “that real independent safety data do not exist.” In fact, independent safety data do exist. Independent measurements have been made on various versions of this product and all results are consistent with the dose specified by the manufacturer. Examples include:
- Sandia National Laboratories, measurements made July 1991. Published as Sandia Report: Evaluation Tests of the SECURE 1000 Scanning System (1992), National Technical Information Service, DE92013773
- FDA, dose measurements re-verified via computational evaluation, September 15, 1998
- N43.17 working group, measurements made at Folsom State Prison on November 15, 1999
- FDA & NIST, Assessment for TSA, July 21, 2006
- Johns Hopkins University Applied Physics Laboratory (JHU APL), Assessment for TSA, October 2009
Fourth is the concern that “the relevant radiation quantity, the Flux [photons per unit area and time (because this is a scanning device)] has not been characterized.” We disagree that flux is the appropriate quantity. The air kerma (or skin entrance exposure) for one screening can be determined by a direct measurement of the total charge produced in the air contained in an ion chamber during one complete screening when the meter is correctly calibrated14. Additionally, measurements to determine the amount of material required to reduce the intensity of the x-ray exposure by half16 are necessary to convert air kerma (or exposure) to effective dose15. These measurements can most practically be made —and indeed have been repeatedly made— at locations where these products are installed and can be made without altering a scanner’s normal operation. These are the same sorts of measurements made to characterize the output of medical x-ray systems17.
Fifth is the assertion that “if the key data (flux-integrated photons per unit values) were available, it would be straightforward to accurately model the dose being deposited in the skin and adjacent tissues using available computer codes [. . .]” In fact, we have done better. FDA and NIST used software called PCXMC18 to estimate the individual organ doses and to calculate effective dose. This analysis was part of an evaluation performed under contract for TSA. The input information required by the PCXMC program required considerably more information than simply the x-ray flux. Its parameters include 1) the x-ray tube anode angle, 2) anode voltage, 3) total filtration, 4) x-ray field size, 5) location of the field on the body, 6) focus-to-skin distance (FSD), and 7) entrance skin exposure. Every parameter was measured, calculated, or verified by indirect measurement. The modeling results revealed that the dose to the skin is approximately twice the effective dose19.
The letter continues: “[. . .] which would resolve the potential concerns over radiation damage.” Direct measurements of the exposure or air kerma from one screening combined with measurements to determine the half-value layer provide sufficient information to adequately estimate the effective dose. There are a number of available publications by groups of recognized experts regarding the biological effects of ionizing radiation and the risk of detriment related to the effective dose. These documents include Health Risks from Exposure to Low Levels of Ionizing Radiation : BEIR VII Phase 2 (2006) and NCRP report no. 115 Risk Estimates for Radiation Protection, as well as the documents that specifically address security screening of people with ionizing radiation mentioned in this letter.
Other specific concerns expressed in the letter are based on the assumption that a screening results in skin or other organ doses that are orders of magnitude higher than the effective dose per screening. The dose to other organs is less than, equal to, or at most approximately three times the effective dose20 for the deployed product. The annual dose limit for security screening is the same as the NCRP recommendations for the annual effective dose limit for the general public including special populations21. An individual would have to receive more than 1000 screenings to begin to approach the annual limit.
With regard to concerns about the hardware itself, the standard requires that products have safety systems to terminate emission of the primary beam in the event of any system problem that could result in abnormal or unintended radiation emission. The scan motion cannot be adjusted. If the scan motion were intentionally redesigned and changed to scan the groin at a slower rate than the rest of the body, the point of measurement to determine the dose per screening would also change. The dose per screening measurement must be made at the point of maximum exposure in order to comply with the standard. Manufacturers are required to report changes to a product’s performance specifications when those changes can affect radiation safety, as would be the case with any change to dose per screening.
These products have been available commercially in the United States since 1992. Manufacturers of any type of electronic product that emits radiation -- including full-body x-ray security systems -- are required to notify FDA immediately upon discovery of any accidental radiation occurrence or radiation safety defect. TSA policy is to require a survey of x-ray systems annually, after any maintenance that could affect radiation shielding, and after any impacts that could affect radiation shielding. FDA regulations require notifications if the manufacturer or FDA determines that an electronic product emits radiation unnecessary to the accomplishment of its primary purpose creating a risk of injury, including genetic injury, to any person. Such a product is then considered to have a radiation safety defect. Unless a manufacturer can provide evidence that a significant risk to public health is not created by a defect, the manufacturer is required to repair, repurchase, or replace its products. Raising the dose delivered without gaining a commensurate increase in safety could be grounds to declare that a product emits radiation unnecessary to the accomplishment of its primary purpose and thus has a radiation safety defect. Products and practices that comply with the American national radiation safety standard22 do not present a significant risk to public health.
In summary, the potential health risks from a full-body screening with a general-use x-ray security system are miniscule. Several groups of recognized experts have been assembled and have analyzed the radiation safety issues associated with this technology. These experts have published recommendations, commentaries, technical reports, and an American national radiation safety standard as a result of their analyses. This technology has been available for nearly two decades and we have based our evaluation on scientific evidence and on the recommendations of recognized experts. Public meetings were held to discuss these products with FDA’s advisory panel (TEPRSSC), and the American national radiation safety standard was available for public comment both before its initial publication and before its recently published revision. There are numerous publications regarding the biological effects of radiation and the appropriate protection limits for the general public that apply to these products. As a result of these evidence-based, responsible actions, we are confident that full-body x-ray security products and practices do not pose a significant risk to the public health.
We enclose a list of references to some of the relevant reports, commentaries, and the current safety standard. If you have any further questions or concerns, please contact either of the individuals listed below.
Sincerely yours,
John L. McCrohan
Deputy Director for Technical and Radiological Initiatives
Office of Communication, Education, and Radiation Programs
Center for Devices and Radiological Health
Food and Drug Administration
Karen R. Shelton Waters
Deputy Assistant Administrator / Chief Administrative Officer
Designated Safety and Health Official
Transportation Security Administration
Enclosure
REFERENCES
- ANSI/HPS N43.17-2009 Radiation Safety for Personnel Security Screening Systems Using X-Ray or Gamma Radiation (http://hps.org/hpssc/)
- More information on the ANSI standards setting process is available on the ANSI website (http://www.ansi.org/standards_activities/overview/overview.aspx?menuid=3 )
- Interagency Steering Committee on Radiation Standards (ISCORS), Guidance on Security Screening of Humans Using Ionizing Radiation (GSSHUIR) Report (http://www.iscors.org/doc/GSSHUIR%20July%202008.pdf)
- NCRP commentary 16, Screening of humans for security purposes using ionizing radiation scanning systems (2003)
- Press release regarding commentary no. 16 (May 26, 2010) (http://www.ncrponline.org/Press_Rel/Commentaries/Comm_16_Press_Release.pdf)
- NCRP Statement 10, Recent Applications of the NCRP Public Dose Limit Recommendation for Ionizing Radiation (2004)
- NCRP report no. 115 Risk Estimates for Radiation Protection (1993)
- NCRP report no. 116 Limitation of Exposure to Ionizing Radiation (1993)
- NCRP report no. 160, Ionizing Radiation Exposure of the Population of the United States (2009)
- HPS Position Statement Use of Ionizing Radiation for Security Screening Individuals (http://hps.org/documents/securityscreening_ps017-1.pdf)
- American College of Radiology (ACR) Statement on Airport Full-body Scanners and Radiation
- U.S. Transportation Security Administration’s (TSA) web site regarding advanced imaging technology (http://www.tsa.gov/approach/tech/imaging_technology.shtm).
- Information on laws and regulations applicable to manufacturers of people screening security systems that use x-rays is available on FDA’s X-Ray & Particulate Products other than Medical Diagnostic or Cabinet page
- The concept of justification based on a societal benefit appears in the International Commission on Radiological Protection (ICRP) report 60 (see paragraph S14).
- Health Physics Society fact sheet on Environmental radiation (http://hps.org/documents/environmental_radiation_fact_sheet.pdf)
- Security screening with x-rays was discussed at several Technical Electronic Product Radiation Safety Standards Committee (TEPRSSC) meetings (http://www.fda.gov/AdvisoryCommittees/CommitteesMeetingMaterials/Radiation-EmittingProducts/TechnicalElectronicProductRadiationSafetyStandardsCommittee/default.htm). The last discussion took place during the October 1, 2003 meeting.
- Sandia Report: Evaluation Tests of the SECURE 1000 Scanning System (http://www.ntis.gov/search/product.aspx?ABBR=DE92013773)
- Health Risks from Exposure to Low Levels of Ionizing Radiation: BEIR VII Phase 2 (2006) (http://www.nap.edu/openbook.php?record_id=11340&pag)
- Assessment of the Rapiscan Secure 1000 ® Body Scanner for Conformance with Radiological Safety Standards. July 21, 2006, produced for TSA, measurements made at FDA, report completed at NIST (http://www.tsa.gov/research/reading/index.shtm)
- Radiation Safety Engineering Assessment Report for the Rapiscan Secure 1000 in Single Pose Configuration ,Johns Hopkins University Applied Physics Laboratory, Assessment for TSA, October 2009 and revised August 2010 (http://www.tsa.gov/research/reading/index.shtm)
1 ANSI/HPS N43.17-2002 Radiation Safety For Personnel Security Screening Systems Using X-rays
2 NCRP was founded in 1964 by the U.S. Congress to “cooperate with the International Commission on Radiological Protection, the Federal Radiation Council, the International Commission on Radiation Units and Measurements, and other national and international organizations, governmental and private, concerned with radiation quantities, units and measurements and with radiation protection.”
3 NCRP report no. 116 Limitation of exposure to ionizing radiation (1993); pages 45-47, 56
4 “Mostly” refers to the unlikely situation where individuals are routinely screened so many times in one 12-month period that the annual dose limit would be exceeded. For example, an individual can be screened 19 times each week and would not receive more than the annual dose limit.
5 Initial presentation October 20, 2003
6 1st work group meeting May 11, 2006
7 ANSI/HPS N43.17-2009 Radiation Safety for Personnel Security Screening Systems Using X-Ray or Gamma Radiation
8 Ibid (ANSI/HPS N43.17-2009): “Various organizations have studied the biological effects of ionizing radiation exposure. The National Council on Radiation Protection and Measurements (NCRP) reviewed two independent studies, one by the United Nations Scientific Committee on the Effects of Atomic Radiation (UNSCEAR 1988) and the other by the National Academy of Sciences/National Research Council, Committee on the Biological Effects of Ionizing Radiation, known as BEIR V (NAS/NRC 1990). Based on this review, the NCRP recommends that, for radiation protection purposes, an incremental lifetime risk of fatal cancer of 5% per sievert be used for the general population (NCRP 1993). The 5% per sievert risk is also consistent with the more recent BEIR VII report (NAS/NRC 2006). Application of this risk estimate means that each 0.01 μSv (1 μrem) of effective dose received is considered to contribute 5 × 10-10 (one chance in two billion) to an individual's risk of contracting a fatal cancer during his or her lifetime. These low-dose estimates assume a “linear no-threshold” relationship between radiation exposure and health effects.
Both the NCRP and the International Commission on Radiological Protection (ICRP) recommend that members of the general population who are frequently exposed to ionizing radiation not exceed an annual effective dose of 1 mSv (100 mrem) from all man-made, non-medical sources (NCRP 1993; ICRP 2007). Further, the NCRP recommends that institutions ensure that the individuals they expose do not repeatedly exceed the 1 mSv yearly limit from all non-medical sources. Information relating to other sources of radiation exposure may be difficult to obtain, so institutions have the option to ensure that the radiation sources under their own control do not contribute to an individual more than an annual effective dose of 0.25 mSv (25 mrem).
General-use systems operating in accordance with this standard produce a maximum reference effective dose of 0.25 μSv (25 μrem) per screening. Therefore, an individual may be screened up to 1,000 times each year without exceeding the annual 0.25 mSv (25 mrem) limit. The associated incremental risk of death is 1 in 80,000,000 per screening.”
9 NCRP report no. 116 Limitation of exposure to ionizing radiation (1993), page 56
10 FDA & NIST Assessment of the Rapiscan Secure 1000® Body Scanner for Conformance with Radiological Safety Standards. July 21, 2006, produced for TSA. This skin dose is an estimate based on dose modeling. This estimate is only for products with very similar x-ray output. The difference between skin and effective dose is smaller for products that use higher energy or more filtration.
11 The actual dose per screening specification is 0.05 µSv or less. The JHU APL assessment report confirms that the product meets this specification.
12 NCRP report no. 158 Uncertainties in the Measurement and Dosimetry of External Radiation, page 22
13 Radiation doses from exposures that may result in delayed stochastic effects are expressed in the quantity effective dose (E):
where HT is the equivalent dose in an organ or tissue T, and wT is the tissue weighting factor that accounts for the radiation sensitivity of organ or tissue T.
14 ANSI/HPS N43.17-2009 Radiation Safety for Personnel Security Screening Systems Using X-Ray or Gamma Radiation, section ‘C.3.2 Calibration’
15 This quantity is called the half-value layer (HVL). HVL is often expressed in terms of the thickness of aluminum required.
16 ANSI/HPS N43.17-2009 Radiation Safety for Personnel Security Screening Systems Using X-Ray or Gamma Radiation, section ‘6.0 Dose Limitation’ and ‘A.1 Reference Effective Dose’
17 Nationwide Evaluation of X-Ray Trends (NEXT) (http://www.fda.gov/Radiation-EmittingProducts/RadiationSafety/ NationwideEvaluationofX-RayTrendsNEXT/default.htm)
18 Servomaa, A. and Tapiovaara, M. Organ dose Calculation in Medical X Ray Examinations by the Program PCXMC. Radiation Protection Dosimetry 80, 213-219 (1998).
19 FDA & NIST Assessment of the Rapiscan Secure 1000® Body Scanner for Conformance with Radiological Safety Standards. July 21, 2006, produced for TSA.
20 FDA & NIST Assessment (2006); Our dose modeling reveals that a screening that delivers 0.25 µSv effective dose would deliver approximately 0.12 µSv to the uterus or 0.69 µSv to the testes. This estimate applies to products with very similar x-ray output.
21 NCRP Statement 10, Recent Applications of the NCRP Public Dose Limit Recommendation for Ionizing Radiation (2004) and NCRP report no. 116 Limitation of exposure to ionizing radiation (1993).
22 ANSI/HPS N43.17-2009 Radiation Safety for Personnel Security Screening Systems Using X-Ray or Gamma Radiation